Case Study: The Amazonian Road Decision
The proposed Pucallpa–Cruzeiro do Sul will connect the Amazon’s interior to urban centers and export markets in Peru and Brazil. However, critics are worried that the road will also create new opportunities for illegal logging and infringe on the territory of indigenous communities and wildlife.
Biology, Geography, Human Geography

Loading ...

On the western edge of the Amazon River, there is a proposal to construct a road. This road would connect the remote town of Cruzeiro do Sul, Brazil, with the larger city of Pucallpa, Peru. The construction of the road has become a subject of contentious debate. Proponents of the road claimed that it would provide an efficient way for rural farmers and tradesmen to get their goods to city markets. They claimed it would also allow loggers to more easily transport timber from the depths of the Amazon rainforest to sawmills. From the sawmills in Pucallpa, goods could be transported to Peru’s Pacific coast and shipped to international buyers. Critics of the Pucallpa-Cruzeiro do Sul road, however, argue that it would cut right through traditional territories of the Ashéninka, an indigenous people of eastern Peru. Many leaders fear the road will increase access to previously undeveloped rainforest, threatening the ecosystem and the Ashéninka way of life. Large trees, such as mahogany, for example, will catch the eye of illegal loggers because of their high market value. The great mahogany trees also serve as protection to the Ashéninka from the outside world and are essential for the health of the Amazonian rainforest. The trees provide shelter, food, and nesting grounds that sustain the vast biodiversity within the ecosystem, an ecosystem the Ashéninka have come to depend on for their own food, shelter, and life sustenance. Geography The Amazon Basin is located in South America, covering an area of seven million square kilometers (2.7 million square miles). Nearly 70 percent of the basin falls within Brazil with remaining areas stretching into parts of Peru, Ecuador, Bolivia, Colombia, Venezuela, and Guyana. The Amazon’s massive drainage basin is made of dozens of smaller watersheds , including the Tamaya. Its watershed lies at the headwaters of the Purus and Juruá Rivers, near the border of Peru and Brazil. The Ashéninka people have lived in this region for centuries, surviving on game, fish, and cultivated crops, such as yucca roots, sweet potato, corn, coffee, and sugar cane. Background The rainforest surrounding the Amazon is the largest on the entire planet. In addition to 33 million human inhabitants, including 385 distinct Indigenous groups, it hosts the greatest diversity of plant and animal life in the world. More than two million species of insects are native to the region, including many tree-living species and hundreds of spiders and butterflies. Primates are abundant—including howler, spider, and capuchin monkeys—along with sloths, snakes, and iguanas. Brightly colored parrots, toucans, and parakeets are just some of the region’s native birds. Many of these species are unique to the Amazon rainforest, which means they cannot be found anywhere else in the world. At a global level, the Amazon rainforest helps to regulate climate and acts as a carbon sink for greenhouse gases . At a national level, the Amazon is considered a source of energy and income, based on production and commercialization of raw materials. Some of the most valued tree species in the world thrive in the rainforest. Mahogany is one of the most valuable resources from the Amazon forest. The tree’s rich, red grain and durability make it one of the most coveted building materials in the world. A single mahogany tree can fetch thousands of U.S. dollars on the international market. Even though logging is prohibited in much of the Amazon River, it is legal in some areas in large part because the sale of the wood is so lucrative. The high demand for mahogany has left many of Peru’s watersheds—such as the Tamaya—stripped of their most valuable trees. Without large trees, and their roots, the watershed risks heavy flooding and soil erosion. Conflict The Pucallpa-Cruzeiro do Sul road is part of a larger development plan to link South America’s remote, isolated economies through new transportation, energy, and telecommunications projects. Tension exists between communities that favor developing the rural economies of the Amazon Basin and those who favor preserving its forested areas and diversity of life. The Initiative for the Integration of the Regional Infrastructure of South America (IIRSA) is a proposal for the construction of several highways throughout the continent, five of them within the western Amazon Basin. The Pucallpa-Cruzeiro do Sul road is one such proposed highway. Supporters of the Pucallpa-Cruzeiro do Sul road say international demand for Amazonian resources could help develop the rural economies that are scattered throughout the basin. In addition to providing a route of access for rural goods to enter the global market, the road will allow members of rural communities to access better health care, education, and welfare. This could lead to improved living conditions, healthier lifestyles, and longer life spans. Conservationists are concerned that infrastructure such as the Pucallpa-Cruzeiro do Sul road will devastate an already weakened Amazonian ecosystem, as road access is highly correlated with deforestation . In Brazil, for instance, 80 percent of deforestation occurs within 48.28 kilometers (30 miles) of a road. Critics argue that the construction of a road along the Brazil-Peru corridor will provide easier access for loggers to reach mahogany and other trees. Indigenous communities like the Ashéninka will also be affected. These communities have largely chosen to maintain a traditional way of life, and conservationists are concerned that the Pucallpa-Cruzeiro do Sul road may expose them to disease and land theft. Identification of Stakeholders Indigenous Communities: Members of the Ashéninka community are trying to protect the forest and their native lands. Yet, like other Indigenous communities in the area, they are in turmoil, largely divided between those favoring conservation and those seeking greater economic opportunities. While the Ashéninka want to preserve their culture and connections to the forest, they also need access to things like clothes, soap, and medicine. The road could establish trade routes that make these goods more accessible. However, isolated peoples could be exposed to disease and land theft. Wildlife: The proposed Pucallpa-Cruzeiro do Sul road runs through Serra do Divisor National Park, Brazil, and other reserves that are home to threatened and rare species, including mammals, reptiles, and birds. For some of these species, such as the spider monkey and red howler monkey, the construction of the road could make their populations vulnerable to fragmentation and more visible to hunters. As mahogany and other canopy giants are removed, any wildlife that relies on the trees for shelter, nesting, or food will need to relocate. Amazonian Ecosystem: In addition to the detrimental effects to the flora and fauna in the area, the construction of the Pucallpa-Cruzeiro do Sul road could accelerate erosion, reduce water quality, and increase deforestation for agriculture and timber extraction. Tropical forest accounts for 40 percent of the global terrestrial carbon sink. A reduced number of trees could exacerbate global warming. Fewer forests means larger amounts of greenhouse gases entering the atmosphere. Logging Companies: If a road is constructed, loggers will have easier access to mahogany and other trees, allowing them to generate more income and provide a higher standard of living for their families and communities. A higher standard of living might include expanded educational opportunities, improved healthcare facilities, and the chance to participate in political debate. Residents of Rural Communities: The Pucallpa-Cruzeiro do Sul road would allow local farmers and business people to transfer goods from the Amazonian interior to Peru’s Pacific coast. Right now, merchants who want to travel between Cruzeiro do Sul and Pucallpa must do so by plane. A reliable road would improve basic infrastructure, transportation, and communication for greater commercial and social integration between Peru and Brazil, which meets part of the larger objective of the Initiative for the Integration of Regional Infrastructure in South America. International Consumers: The global demand for mahogany makes it a multimillion dollar business. Mahogany is used to create bedroom sets, cabinets, flooring, and patio decks throughout the world, mostly in the United States and Europe. Conflict Mitigation Groups are seeking to mitigate conflict in the Pucallpa-Cruzeiro do Sul road conflict through dialogue and alternate infrastructure plans. Environmental conservation groups have suggested that the Pucallpa-Cruzeiro do Sul road be removed from the list of approved projects until the community engages in greater communication surrounding two aspects of the project. First, conservationists are seeking more information on the environmental impact of the construction. This discussion involves local environmental groups, government representatives, and businesses. Second, conservationists are seeking full consent to the project from indigenous communities. Some critics of the Pucallpa-Cruzeiro do Sul road argue that roads are not the only option for the Pucallpa business community to extend its commerce. Traditional river systems are already in place. These critics think the fluvial network should be explored as a viable alternative to road construction. The Upper Amazon Conservancy is working with indigenous peoples to help protect their native territories. One initiative involves organizing community “vigilance committees” that consist of members of indigenous peoples who help park services by patrolling the edges of national parks and keeping illegal loggers out.
Media Credits
The audio, illustrations, photos, and videos are credited beneath the media asset, except for promotional images, which generally link to another page that contains the media credit. The Rights Holder for media is the person or group credited.
Research Manager
Educator reviewer, expert reviewer, last updated.
October 19, 2023
User Permissions
For information on user permissions, please read our Terms of Service. If you have questions about how to cite anything on our website in your project or classroom presentation, please contact your teacher. They will best know the preferred format. When you reach out to them, you will need the page title, URL, and the date you accessed the resource.
If a media asset is downloadable, a download button appears in the corner of the media viewer. If no button appears, you cannot download or save the media.
Text on this page is printable and can be used according to our Terms of Service .
Interactives
Any interactives on this page can only be played while you are visiting our website. You cannot download interactives.
Related Resources

- Life in the Amazon basin
Human beings are the most brained creatures. As humans, we excel in exploring the mother Earth and using it for our best purposes. This skill of ours has given us the advantage of interacting with the environment, and Amazon basin is the best living example of that. Amazon basin is the result of the many tributaries joining the Amazon river. Let’s explore the human capacity to interact with the environment.
Suggested Videos
Life in amazon basin.
We humans, are dependent on nature and interact with it for various reasons. Our interaction with the environment is endless and so is our ability to use it. Being the best example of human interaction with the environment, the Amazon basin has been exploited since time immemorial. Amazon basin is located in South America at 10° N and 10° S of the tropical region.
This region is also referred as the equatorial region. The Amazon river flows through the region and reaches the Atlantic Ocean through the mountains in the west.

Before reading further about Amazon basin it is important that we know the following definitions:
- River’s Mouth: The place where a river flows into another water body is called the river’s mouth.
- Tributaries: When a river or a stream flows into a larger river or lake then that river or lake is called the tributary of the larger river.

The Climate
Amazon basin is situated in the equatorial region which is hot and humid all through the year. Days and nights both are equally hot and wet. Nights are comparatively less hot but the humidity level remains unchanged. Due to the humid conditions here, it rains almost every day.
Browse more Topics under Human Environment Interactions
- Life in the Ganga – Brahmaputra Basin
Read more about Life in Ganga Brahmaputra Basin here in detail.
The Rainforest
As it rains throughout the year the forests here are dense. The trees form a dense roof of leaves that do not even let sunlight penetrate into the forest area. Also, the surface of the earth is damp and dark. So shade tolerant vegetation is present in abundance here. Prominent plant parasites found here are Bromeliads and Orchids. The rainforests of Amazon basin are flocked with a variety of fauna as well. As a result, you can find the rarest of species loitering in the forests here.
Hummingbird, Toucans, Amazon Kingfisher, Hyacinth Macaw, Blue-fronted Amazon are some of the bird species that are present here. And animals like Sloth, Capybara, monkeys, ant-eating tapirs, poison dart frogs are present all over the rainforests of Amazon. The list does not end here! You get a glimpse of the grandest of the reptiles as well.
Crocodiles and snakes like Pythons and Anacondas are common here. Apart from these aquatic animals like the Piranha and Giant Otter can be sighted in the river basin. The list of fauna and flora in Amazon Basin is endless.

People here cut a few trees and cultivate the land according to their needs and requirements. Men have occupations like fishing and hunting, while it’s the women who take care of the crops and fields. The land being near the Amazon river is very fertile which makes it a good for farming. People here generally grow crops like Pineapple, Tapioca, Sweet Potato, Cassava (manioc), Coffee, maize, and cocoa. We call them the Cash crops .
As already said, men practice fishing and hunting which are uncertain means of living. It is the women of Amazon basin who are the major bread earners of the family. From taking care of fields to feeding their families with the vegetables that they cultivate, women are responsible for their families well-being.
They practice slash and burn technique of agriculture. In slash and burn agriculture system, we clear the required forest land for farming. We slash or cut down the trees and bushes. As soon as the fertility of land degrades, women proceed to clear a new piece of land.
The old land eventually gains back its fertility with trees and bushes growing back on them. Manioc or Cassava is the staple food while queen ants and egg sacs are the other savouries of people near the Amazon river.

Settlements of People
People live in special kind of houses called the Maloca here. These houses have steep slant roofs and are large and apartment like in shape. People also reside in houses that are identical to beehives and have thatched roofs over them. Since the settlements here are in close proximity of forests, people find wood in abundance for their personal use.

With pacing technology and modernism, the life of people is slowly changing here. As it is the human nature to evolve and enjoy facilities to their best the advent of transportation has helped in easy navigation through the basin. An area which once was navigable only through the Amazon river can today be explored through Trans-Amazon Highway. Moreover, with aeroplanes and helicopters, it has become easier and faster to reach various places around the basin.
Human Interaction with environment benefits humans, but after an extent depletes the environment. The regular felling of trees in the Amazon basin has resulted in a change of ecology here. The developmental activities near the Amazon river, eventually, have resulted in the destruction of the rainforests. The effects may not be visible today, but in the near future, they surely will. The map below signifies the change human interaction has brought to the environment:

Amazon Basin is the richest example of human interaction with the environment. To let it stay rich with vegetation it has now become imminent to use the resources from the basin intelligently. We humans interact with the environment for our benefits but doing so at the cost of ecology can be fatal for our generations to come.
Solved Questions For You
Q1: Define the major difference between a hamlet and a village.
A) Population B) Group of Houses C) Pollution D) None of the Above
Solution: A) Villages are larger than hamlets. They have a greater population than hamlets. Hamlets are small towns and community while as villages are mostly large suburbs and crossroads. Hence, Hamlet settlements are also less polluted as a result of a smaller population.
Q2: From where the Urban settlements have evolved?
A) Rural Settlements B) Scattered Settlements C) Nucleated Settlements D) None of the Above.
Solution: A) Rural settlements were the first step towards a stable life in the human history. Urban settlements have evolved through the growth and expansion of rural settlements. Increase in population of the rural settlements marks the beginning of urbanisation.
Customize your course in 30 seconds
Which class are you in.

Human Environment Interactions
- Life in Ganga Brahmaputra Basin
Leave a Reply Cancel reply
Your email address will not be published. Required fields are marked *
Download the App

- 0 Shopping Cart

Sustainable Management of the Amazon Rainforest

The Amazon rainforest is located in the north of South America, spanning an area of around 8 million km2 including parts of Brazil, Columbia, Peru, Venezuela, Ecuador Bolivia, Suriname, Guyana and French Guyana.
In some areas of the Amazon rainforest, sustainable management strategies are in place to ensure people today can get the resources they need in a way that ensures future generations can also benefit from the ecosystem .
Sustainable management strategies are affected by political and economic factors .
Governance
Governance relates to control of rainforests and who has a say in how rainforests are used. In some areas, rainforests are protected by national and international laws.
In Brazil, the largest protected area of rainforest is the Central Amazon Conservation Complex (CACC) . The CACC covers 60000 km2 as is classified as a World Heritage Site by the United Nations, which means it is protected by international treaties. Limits are placed on hunting , logging and fishing and access is limited.

Central Amazon Conservation Complex (CACC)
In other areas local communities, with the help of NGOs, are involved in rainforest governance. In Columbia, an organisation known as Natütama is working with the local community in Puerto Nariño to protect river species such as the Amazon River dolphin. Local people are employed to teach members of the community on how to protect habitats and endangered river species. Local fishermen collect information about the number and distribution of species and report illegal hunting.
Commodity Value
Commodity value means assigning a value to different good and services in a rainforest. Sustainable management ensures rainforests are worth. more than the value of the timber and other resources that can be extracted, such as gold. An example of this is sustainable foresty, which balances the removal of trees to sell with the conservation of the forest.
Selective logging involves only removing a small number of trees, allowing the forest to regenerate naturally. This saves money in the long run as logging companies do not need to replace felled trees.
Sustainable logging companies such as Precious Woods Amazon place limits on the number of trees being cut down so the rainforest can recover. They also use a range of species so that none are over-exploited.
International agreements try to reduce illegal logging and ensure timber comes from sustainable sources. The Forestry Stewardship Council allows the use of its logo by companies that operate in a sustainable way so consumers know they are buying sustainable timber.

FSC certified wood
Ecotourism is a type of tourism that minimises damage to the environment and benefits local people.
An example of an ecotourism project is the Yachana Lodge in Equador. It is located in a remote area of the Amazon Rainforest where local people rely on subsistence farming.

Yachana Lodge
The project employs local people. This provides a reliable source of income and a better quality of life. The project encourages local people to use the rainforest in a sustainable way so tourists continue to visit.
Volunteers work with local Amazon youth who study at the Yachana Technical High School where learning is focused on five main areas:
- Rainforest conservation
- Sustainable agriculture
- Renewable energy
- Animal husbandry
- Micro-enterprise development .
Tourists are only allowed to visit in small groups, minimising their impact on the environment. Tourists take part in activities that help raise awareness of conservation issues.
Entrance fees are paid by the tourists which are invested in conservation and education projects.

Premium Resources
Please support internet geography.
If you've found the resources on this page useful please consider making a secure donation via PayPal to support the development of the site. The site is self-funded and your support is really appreciated.
Related Topics
Use the images below to explore related GeoTopics.
Sustainable Management of the Rainforest
Topic home, share this:.
- Click to share on Twitter (Opens in new window)
- Click to share on Facebook (Opens in new window)
- Click to share on Pinterest (Opens in new window)
- Click to email a link to a friend (Opens in new window)
- Click to share on WhatsApp (Opens in new window)
- Click to print (Opens in new window)
If you've found the resources on this site useful please consider making a secure donation via PayPal to support the development of the site. The site is self-funded and your support is really appreciated.
Search Internet Geography
Top posts and pages.
Latest Blog Entries
Pin It on Pinterest
- Click to share
- Print Friendly
NTRS - NASA Technical Reports Server
Available downloads, related records.

Effective Geography
Dolly e sequeira, p s lathika, beeta publications.
- Representation of Geographical Features
- Major Landforms
- Water Bodies
- Agriculture
- North America: Location and Physical Features
- Case Study: Lumbering in Canada
- South America: Location and Physical Features
Case Study: Life in the Amazon River Basin
Available answers.
Fill in the blanks:
- The Amazon Basin is spread over ________ countries.
- The Amazon Basin is covered by equatorial _________.
- The native Americans are called ________.
- _______ is a deadly fish found in the waters of Amazon Basin.
- _______ is a port on the river Amazon in Peru.
Match the following:
| Column A | Column B |
| 1. Manioc | (a) Port of Amazon River |
| 2. Maloca | (b) Equatorial Rainforests |
| 3. Belem | (c) Venomous Snake |
| 4. Selvas | (d) Staple food of Amerindians |
| 5. Anaconda | (e) Long house of Amerindians |
Where is Amazon Basin located? How many countries are part of the Amazon Basin?
- 4. What type of climate is found in the Amazon Basin?
- 5. Why does the Amazon Basin have dense forests?
- 6. How do the native Americans obtain their food?
| GEOG 30N Environment and Society in a Changing World |

- ORIENTATION
- ASSIGNMENTS
- Program Home Page
- LIBRARY RESOURCES
- Getting Help
- Engaging Course Concepts
Case Study: The Amazon Rainforest
The Amazon in context
Tropical rainforests are often considered to be the “cradles of biodiversity.” Though they cover only about 6% of the Earth’s land surface, they are home to over 50% of global biodiversity. Rainforests also take in massive amounts of carbon dioxide and release oxygen through photosynthesis, which has also given them the nickname “lungs of the planet.” They also store very large amounts of carbon, and so cutting and burning their biomass contributes to global climate change. Many modern medicines are derived from rainforest plants, and several very important food crops originated in the rainforest, including bananas, mangos, chocolate, coffee, and sugar cane.

In order to qualify as a tropical rainforest, an area must receive over 250 centimeters of rainfall each year and have an average temperature above 24 degrees centigrade, as well as never experience frosts. The Amazon rainforest in South America is the largest in the world. The second largest is the Congo in central Africa, and other important rainforests can be found in Central America, the Caribbean, and Southeast Asia. Brazil contains about 40% of the world’s remaining tropical rainforest. Its rainforest covers an area of land about 2/3 the size of the continental United States.
There are countless reasons, both anthropocentric and ecocentric, to value rainforests. But they are one of the most threatened types of ecosystems in the world today. It’s somewhat difficult to estimate how quickly rainforests are being cut down, but estimates range from between 50,000 and 170,000 square kilometers per year. Even the most conservative estimates project that if we keep cutting down rainforests as we are today, within about 100 years there will be none left.
How does a rainforest work?
Rainforests are incredibly complex ecosystems, but understanding a few basics about their ecology will help us understand why clear-cutting and fragmentation are such destructive activities for rainforest biodiversity.

High biodiversity in tropical rainforests means that the interrelationships between organisms are very complex. A single tree may house more than 40 different ant species, each of which has a different ecological function and may alter the habitat in distinct and important ways. Ecologists debate about whether systems that have high biodiversity are stable and resilient, like a spider web composed of many strong individual strands, or fragile, like a house of cards. Both metaphors are likely appropriate in some cases. One thing we can be certain of is that it is very difficult in a rainforest system, as in most other ecosystems, to affect just one type of organism. Also, clear cutting one small area may damage hundreds or thousands of established species interactions that reach beyond the cleared area.
Pollination is a challenge for rainforest trees because there are so many different species, unlike forests in the temperate regions that are often dominated by less than a dozen tree species. One solution is for individual trees to grow close together, making pollination simpler, but this can make that species vulnerable to extinction if the one area where it lives is clear cut. Another strategy is to develop a mutualistic relationship with a long-distance pollinator, like a specific bee or hummingbird species. These pollinators develop mental maps of where each tree of a particular species is located and then travel between them on a sort of “trap-line” that allows trees to pollinate each other. One problem is that if a forest is fragmented then these trap-line connections can be disrupted, and so trees can fail to be pollinated and reproduce even if they haven’t been cut.
The quality of rainforest soils is perhaps the most surprising aspect of their ecology. We might expect a lush rainforest to grow from incredibly rich, fertile soils, but actually, the opposite is true. While some rainforest soils that are derived from volcanic ash or from river deposits can be quite fertile, generally rainforest soils are very poor in nutrients and organic matter. Rainforests hold most of their nutrients in their live vegetation, not in the soil. Their soils do not maintain nutrients very well either, which means that existing nutrients quickly “leech” out, being carried away by water as it percolates through the soil. Also, soils in rainforests tend to be acidic, which means that it’s difficult for plants to access even the few existing nutrients. The section on slash and burn agriculture in the previous module describes some of the challenges that farmers face when they attempt to grow crops on tropical rainforest soils, but perhaps the most important lesson is that once a rainforest is cut down and cleared away, very little fertility is left to help a forest regrow.
What is driving deforestation in the Amazon?
Many factors contribute to tropical deforestation, but consider this typical set of circumstances and processes that result in rapid and unsustainable rates of deforestation. This story fits well with the historical experience of Brazil and other countries with territory in the Amazon Basin.
Population growth and poverty encourage poor farmers to clear new areas of rainforest, and their efforts are further exacerbated by government policies that permit landless peasants to establish legal title to land that they have cleared.
At the same time, international lending institutions like the World Bank provide money to the national government for large-scale projects like mining, construction of dams, new roads, and other infrastructure that directly reduces the forest or makes it easier for farmers to access new areas to clear.
The activities most often encouraging new road development are timber harvesting and mining. Loggers cut out the best timber for domestic use or export, and in the process knock over many other less valuable trees. Those trees are eventually cleared and used for wood pulp, or burned, and the area is converted into cattle pastures. After a few years, the vegetation is sufficiently degraded to make it not profitable to raise cattle, and the land is sold to poor farmers seeking out a subsistence living.
Regardless of how poor farmers get their land, they often are only able to gain a few years of decent crop yields before the poor quality of the soil overwhelms their efforts, and then they are forced to move on to another plot of land. Small-scale farmers also hunt for meat in the remaining fragmented forest areas, which reduces the biodiversity in those areas as well.
Another important factor not mentioned in the scenario above is the clearing of rainforest for industrial agriculture plantations of bananas, pineapples, and sugar cane. These crops are primarily grown for export, and so an additional driver to consider is consumer demand for these crops in countries like the United States.
These cycles of land use, which are driven by poverty and population growth as well as government policies, have led to the rapid loss of tropical rainforests. What is lost in many cases is not simply biodiversity, but also valuable renewable resources that could sustain many generations of humans to come. Efforts to protect rainforests and other areas of high biodiversity is the topic of the next section.
Living World - Amazon Case Study
The Amazon is the largest tropical rainforest on Earth. It sits within the Amazon River basin, covers some 40% of the South American continent and as you can see on the map below includes parts of eight South American countries: Brazil, Bolivia, Peru, Ecuador, Colombia, Venezuela, Guyana, and Suriname. The actual word “Amazon” comes from river.

Amazing Amazon facts; • It is home to 1000 species of bird and 60,000 species of plants • 10 million species of insects live in the Amazon • It is home to 20 million people, who use the wood, cut down trees for farms and for cattle. • It covers 2.1 million square miles of land • The Amazon is home to almost 20% of species on Earth • The UK and Ireland would fit into the Amazon 17 times!
The Amazon caught the public’s attention in the 1980s when a series of shocking news reports said that an area of rainforest the size of Belgium was being cut down and subsequently burnt every year. This deforestation has continued to the present day according to the Sao Paulo Space Research Centre. In 2005 they had lost 17% of Amazon rainforest or 650000 square kilometres. Their satellite data is also showing increased deforestation in parts of the Amazon. The process of deforestation The Amazon helps a Newly Emerging Economy(NEE), Brazil, to make money. They build roads into the forest, logging firms then go in and take out valuable hard woods such as mahogany and cedar, worth thousands of pounds in richer economies like Europe. Then farmers, often cattle ranchers from big companies, burn the rest to make way for cattle pasture. 75% of cleared areas are used in this way. This is clearly shown on the map on figure 22 in red. Many of the deforested areas follow roads and branch off from there. Deforestation is also worse in the South and South East of the Amazon basin, closer to major centres of population in Brazil.

© WWF Source Used with permission.
The causes of deforestation 1. Subsistence and commercial farming – subsistence farming is where poor farmers occupy plots of the forest to grow food to feed themselves and their families. They clear forest and then burn it, hence the name slash and burn. They grow crops until the soil is exhausted and then move on. This contributes to deforestation but not as much as commercial farming (Farming to sell produce for a profit to retailers or food processing companies). The Brazilian region of Mato Grosso was affected by deforestation in the 1980s and 1990s. 43% of rainforest losses were in this region, and area almost ½ the size of France. It has been replaced by fields for grain and cattle. This has allowed Brazil to overtake Australia as the largest exporter of beef in the world. The land is also flat and easy to farm. It also has high temperatures and lots of rainfall.
2. Logging – This involves cutting down trees for sale as timber or pulp. The timber is used to build homes, furniture, etc. and the pulp is used to make paper and paper products. Logging can be either selective or clear cutting. Selective logging is selective because loggers choose only wood that is highly valued, such as mahogany. Clear-cutting is not selective. Loggers are interested in all types of wood and therefore cut all of the trees down, thus clearing the forest, hence the name- clear-cutting.
3. Road building – trees are also clear for roads. Roads are an essential way for the Brazilian government to allow development of the Amazon rainforest. However, unless they are paved many of the roads are unusable during the wettest periods of the year. The Trans Amazonian Highway has already opened up large parts of the forest and now a new road is going to be paved, the BR163 is a road that runs 1700km from Cuiaba to Santarem. The government planned to tarmac it making it a superhighway. This would make the untouched forest along the route more accessible and under threat from development.
4. Mineral extraction – forests are also cleared to make way for huge mines. The Brazilian part of the Amazon has mines that extract iron, manganese, nickel, tin, bauxite, beryllium, copper, lead, tungsten, zinc and gold!

The Belo Monte dam site under construction, copyright Used with the kind permission of Phil Clarke-Hill - His website is amazing, click here to see it.
5. Energy development – This has focussed mainly on using Hydro Electric Power, and there are 150 new dams planned for the Amazon alone. The dams create electricity as water is passed through huge pipes within them, where it turns a turbine which helps to generate the electricity. The power in the Amazon is often used for mining. Dams displace many people and the reservoirs they create flood large area of land, which would previously have been forest. They also alter the hydrological cycle and trap huge quantities of sediment behind them. The huge Belo Monte dam started operating in April 2016 and will generate over 11,000 Mw of power. A new scheme the 8,000-megawatt São Luiz do Tapajós dam has been held up because of the concerns over the impacts on the local Munduruku people.

6. Settlement & population growth – populations are growing within the Amazon forest and along with them settlements. Many people are migrating to the forest looking for work associated with the natural wealth of this environment. Settlements like Parauapebas, an iron ore mining town, have grown rapidly, destroying forest and replacing it with a swath of shanty towns. The population has grown from 154,000 in 2010 to 220,000 in 2012. The Brazilian Amazon’s population grew by a massive 23% between 2000 and 2010, 11% above the national average.
Impacts of deforestation – economic development, soil erosion, contribution to climate change. • Every time forest is cleared species are lost – so we lose BIODIVERSITY • Climate Change - Burning the forest releases greenhouse gasses like CO2. This contributes to the warming of our planet via climate change and global warming. In addition, the loss of trees prevents CO2 being absorbed, making the problem worse. The Amazon also helps to drive the global atmospheric system. There is a lot of rainfall there and changes to the Amazon could disrupt the global system. • Economic development – Brazil has used the forests as a way to develop their country. The forest has many natural riches that can be exploited. In addition, Brazil has huge foreign debt and lots of poor people to feed, so they want to develop the forest. May Brazilians see deforestation as a way to help develop their country and improve people’s standard of living. • Soil erosion - the soils of the Amazon forest are not fertile and are quickly exhausted once the forest is cleared. The farmers now artificially fertilise the soil when in the past the nutrient cycle would have done this naturally. In addition, the lack of forest cover means that soils are exposed to the rainfall. This washes huge amounts of soil into rivers in the process of soil erosion.
NEXT TOPIC - Living World - Sustainable Forest Management
©2015 Cool Geography
- Copyright Policy
- Privacy & Cookies
- Testimonials
- Feedback & support

Impacts of hydraulic redistribution on eco-hydrological cycles: A case study over the Amazon basin
- Research Paper
- Published: 18 July 2018
- Volume 61 , pages 1330–1340, ( 2018 )
Cite this article

- Yuanyuan Wang 1 , 2 ,
- Binghao Jia 2 &
- Zhenghui Xie 2
172 Accesses
3 Citations
Explore all metrics
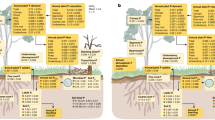
Hydraulic redistribution (HR) refers to the process of soil water transport through the low-resistance pathway provided by plant roots. It has been observed in field studies and proposed to be one of the processes that enable plants to resist water limitations. However, most land-surface models (LSMs) currently do not include this underground root process. In this study, a HR scheme was incorporated into the Community Land Model version 4.5 (CLM4.5) to investigate the effect of HR on the eco-hydrological cycle. Two paired numerical simulations (with and without the new HR scheme) were conducted for the Tapajos National Forest km83 (BRSa3) site and the Amazon. Simulations for the BRSa3 site in the Amazon showed that HR during the wet season was small, <0.1 mm day –1 , transferring water from shallow wet layers to deep dry layers at night; however, HR in the dry season was more obvious, up to 0.3 mm day –1 , transferring water from deep wet layers to shallow dry layers at night. By incorporating HR into CLM4.5, the new model increased gross primary production (GPP) and evapotranspiration (ET) by 10% and 15%, respectively, at the BRSa3 site, partly overcoming the underestimation. For the Amazon, regional analysis also revealed that vegetation responses (including GPP and ET) to seasonal drought and the severe drought of 2005 were better captured with the HR scheme incorporated.
This is a preview of subscription content, log in via an institution to check access.
Access this article
Price includes VAT (Russian Federation)
Instant access to the full article PDF.
Rent this article via DeepDyve
Institutional subscriptions
Similar content being viewed by others
Hydrological Responses to Climate Change and Land-Use Dynamics in Central Asia's Semi-arid Regions: An SWAT Model Analysis of the Tuul River Basin

Spatiotemporal variations of water use efficiency and its driving factors in Inner Mongolia from 2001 to 2020
The response of net primary productivity to climate change and its impact on hydrology in a water-limited agricultural basin
Armas C, Padilla F M, Pugnaire F I, Jackson R B. 2010. Hydraulic lift and tolerance to salinity of semiarid species: Consequences for species interactions. Oecologia, 162: 11–21
Article Google Scholar
Avissar R, Silva Dias P L, Silva Dias M A F, Nobre C. 2002. The largescale biosphere-atmosphere experiment in Amazonia (LBA): Insights and future research needs. J Geophys Res, 107: 8086
Baker I T, Prihodko L, Denning A S, Goulden M, Miller S, Da Rocha H R. 2008. Seasonal drought stress in the Amazon: Reconciling models and observations. J Geophys Res, 113: G00B01
Bauerle T L, Richards J H, Smart D R, Eissenstat D M. 2007. Importance of internal hydraulic redistribution for prolonging the lifespan of roots in dry soil. Plant Cell Environ, 31: 177–186
Google Scholar
Bonan G B, Levis S, Kergoat L, Oleson K W. 2002. Landscapes as patches of plant functional types: An integrating concept for climate and ecosystem models. Glob Biogeochem Cycle, 16: 5-1–5-23
Brooks J R, Meinzer F C, Coulombe R, Gregg J. 2002. Hydraulic redistribution of soil water during summer drought in two contrasting Pacific Northwest coniferous forests. Tree Physiol, 22: 1107–1117
Burgess S S O, Adams M A, Turner N C, Ong C K. 1998. The redistribution of soil water by tree root systems. Oecologia, 115: 306–311
Chen J L, Wilson C R, Tapley B D, Yang Z L, Niu G Y. 2009. 2005 drought event in the Amazon River basin as measured by GRACE and estimated by climate models. J Geophys Res, 114: B05404
Domec J C, Warren J M, Meinzer F C, Brooks J R, Coulombe R. 2004. Native root xylem embolism and stomatal closure in stands of Douglasfir and ponderosa pine: Mitigation by hydraulic redistribution. Oecologia, 141: 7–16
Emerman S H, Dawson T E. 1996. Hydraulic lift and its influence on the water content of the rhizosphere: An example from sugar maple, Acer saccharum. Oecologia, 108: 273–278
Fisher R A, Muszala S, Verteinstein M, Lawrence P, Xu C, McDowell N G, Knox R G, Koven C, Holm J, Rogers B M, Spessa A, Lawrence D, Bonan G. 2015. Taking off the training wheels: The properties of a dynamic vegetation model without climate envelopes, CLM4.5(ED). Geosci Model Dev, 8: 3593–3619
Hao G Y, Jones T J, Luton C, Zhang Y J, Manzane E, Scholz F G, Bucci S J, Cao K F, Goldstein G. 2009. Hydraulic redistribution in dwarf Rhizophora mangle trees driven by interstitial soil water salinity gradients: Impacts on hydraulic architecture and gas exchange. Tree Physiol, 29: 697–705
Hudiburg T W, Law B E, Thornton P E. 2013. Evaluation and improvement of the Community Land Model (CLM4) in Oregon forests. Biogeosciences, 10: 453–470
Hultine K R, Williams D G, Burgess S S O, Keefer T O. 2003. Contrasting patterns of hydraulic redistribution in three desert phreatophytes. Oecologia, 135: 167–175
Hultine K R, Scott R L, Cable W L, Goodrich D C, Williams D G. 2004. Hydraulic redistribution by a dominant, warm-desert phreatophyte: Seasonal patterns and response to precipitation pulses. Funct Ecol, 18: 530–538
Ishikawa C M, Bledsoe C S. 2000. Seasonal and diurnal patterns of soil water potential in the rhizosphere of blue oaks: Evidence for hydraulic lift. Oecologia, 125: 459–465
Jia B H, Xie Z H, Zeng Y J, Wang L Y, Wang Y Y, Xie J B, Xie Z P. 2015. Diurnal and seasonal variations of CO2 fluxes and their climate controlling factors for a subtropical forest in Ningxiang. Adv Atmos Sci, 32: 553–564
Jia B H, Wang Y Y, Xie Z H. 2018. Responses of the terrestrial carbon cycle to drought over China: Modeling sensitivities of the interactive nitrogen and dynamic vegetation. Ecol Model, 368: 52–68
Jung M, Reichstein M, Bondeau A. 2009. Towards global empirical upscaling of FLUXNET eddy covariance observations: Validation of a model tree ensemble approach using a biosphere model. Biogeosciences, 6: 2001–2013
Jung M, Reichstein M, Ciais P, Seneviratne S I, Sheffield J, Goulden M L, Bonan G, Cescatti A, Chen J Q, de Jeu R, Dolman A J, Eugster W, Gerten D, Gianelle D, Gobron N, Heinke J, Kimball J, Law B E, Montagnani L, Mu Q Z, Mueller B, Oleson K, Papale D, Richardson A D, Roupsard O, Running S, Tomelleri E, Viovy N, Weber U, Williams C, Wood E, Zaehle S, Zhang K. 2010. Recent decline in the global land evapotranspiration trend due to limited moisture supply. Nature, 467: 951–954
Jung M, Reichstein M, Margolis H A, Cescatti A, Richardson A D, Arain M A, Arneth A, Bernhofer C, Bonal D, Chen J Q, Gianelle D, Gobron N, Kiely G, Kutsch W, Lasslop G, Law B E, Lindroth A, Merbold L, Montagnani L, Moors E J, Papale D, Sottocornola M, Vaccari F, Williams C. 2011. Global patterns of land-atmosphere fluxes of carbon dioxide, latent heat, and sensible heat derived from eddy covariance, satellite, and meteorological observations. J Geophys Res, 116: G00J07
Lawrence P J, Chase T N. 2007. Representing a new MODIS consistent land surface in the community land model (CLM 3.0). J Geophys Res, 112: G01023
Liu J G, Jia B H, Xie Z H, Shi C X. 2016. Ensemble simulation of land evapotranspiration in China based on a multi-forcing and multi-model approach. Adv Atmos Sci, 33: 673–684
Nadezhdina N, Cermak J, Gasparek J, Nadezhdin V, Prax A. 2006. Vertical and horizontal water redistribution in Norway spruce (Picea abies) roots in the Moravian Upland. Tree Physiol, 26: 1277–1288
Natalia R C, Humberto R R, Lucy R H, Alessandro C A, Laura S B, Bradley C, Osvaldo M C, Plinio B C, Fernando L C, Antonio C C, David R F, Michael L G, Bart K, Jair M M, Yadvinder S M, Antonio O M, Scott D M, Antonio D N, Celso V R, Leonardo S, Ricardo K S, Julio T, Steven C W, Fabricio B Z, Scott R S. 2013. What drives the seasonality of photosynthesis across the Amazon basin? A cross-site analysis of eddy flux tower measurements from the Brasil flux network. Agric For Meteorol, 182–183: 128–144
Neumann R B, Cardon Z G. 2012. The magnitude of hydraulic redistribution by plant roots: A review and synthesis of empirical and modeling studies. New Phytologist, 194: 337–352
Oleson K W, Lawrence D M, Bonan G, Drewniak B, Huang M, Koven C D, Levis S, Li F, Riley W J, Subin Z M, Swenson S C, Thornton P E, Bozbiyik A, Fisher R, Kluzek E, Lamarque J E, Lawrence P J, Leung L R, Lipscomb W, Muszala S, Ricciuto D M, Sacks W, Sun Y, Tang J, Yang Z L. 2013. Technical Description of Version 4.5 of the Community Land Model (CLM), NCAR Tech. Note, NCAR/TN-503+STR, National Center for Atmospheric Research, 434
Oliveira R S, Dawson T E, Burgess S S O. 2005. Evidence for direct water absorption by the shoot of the desiccation-tolerant plant Vellozia flavicans in the savannas of central Brazil. J Trop Ecol, 21: 585–588
Prieto I, Martínez-Tillería K, Martínez-Manchego L, Montecinos S, Pugnaire F I, Squeo F A. 2010. Hydraulic lift through transpiration suppression in shrubs from two arid ecosystems: Patterns and control mechanisms. Oecologia, 163: 855–865
Raczka B, Duarte H F, Koven C D, Ricciuto D, Thornton P E, Lin J C, Bowling D R. 2016. An observational constraint on stomatal function in forests: Evaluating coupled carbon and water vapor exchange with carbon isotopes in the community land model (CLM4.5). Biogeosciences, 13: 5183–5204
Richards J H, Caldwell M M. 1987. Hydraulic lift: Substantial nocturnal water transport between soil layers by Artemisia tridentata roots. Oecologia, 73: 486–489
Ryel R J, Caldwell M M, Yoder C K, Or D, Leffler A. 2002. Hydraulic redistribution in a stand of Artemisia tridentata: Evaluation of benefits to transpiration assessed with a simulation model. Oecologia, 130: 173–184
Saleska S R, Didan K, Huete A R, da Rocha H R. 2007. Amazon forests green-up during 2005 drought. Science, 318: 612–612
Scholz F G, Bucci S J, Hoffmann W A, Meinzer F C, Goldstein G. 2010. Hydraulic lift in a Neotropical savanna: Experimental manipulation and model simulations. Agric For Meteorol, 150: 629–639
Schulze E D, Caldwell M M, Canadell J, Mooney H A, Jackson R B, Parson D, Scholes R, Sala O E, Trimborn P. 1998. Downward flux of water through roots (i.e. inverse hydraulic lift) in dry Kalahari sands. Oecologia, 115: 460–462
Scott R L, Cable W L, Hultine K R. 2008. The ecohydrologic significance of hydraulic redistribution in a semiarid savanna. Water Resour Res, 44: W02440
Shi X Y, Mao J F, Thornton P E, Huang M. 2013. Spatiotemporal patterns of evapotranspiration in response to multiple environmental factors simulated by the Community Land Model. Environ Res Lett, 8: 024012
Smith D M, Jackson N A, Roberts J M, Ong C K. 1999. Reverse flow of sap in tree roots and downward siphoning of water by Grevillea robusta. Funct Ecol, 13: 256–264
Viovy N. 2011. CRUNCEP Dataset, description available at https://doi.org/dods.extra.cea.fr/data/p529viov/cruncep/readme.htm , data available at https://doi.org/dods.extra.cea.fr/store/p529viov/cruncep/V4_1901_2011/
Wang G, Alo C, Mei R, Sun S. 2011. Droughts, hydraulic redistribution, and their impact on vegetation composition in the Amazon forest. Plant Ecol, 212: 663–673
Wang Y Y, Xie Z H, Jia B H. 2016. Incorporation of a dynamic root distribution into CLM4.5: Evaluation of carbon and water fluxes over the Amazon. Adv Atmos Sci, 33: 1047–1060
Wang Y Y, Xie Z H, Jia B H, Yu Y. 2015a. Improving simulation of the terrestrial carbon cycle of China in version 4.5 of the Community Land Model using a revised Vcmax scheme. Atmos Oceanic Sci Lett, 8: 88–94
Wang Y Y, Xie Z H, Jia B H, Yu Y. 2015b. Simulation and evaluation of gross primary productivity in China by using land surface model CLM4 (in Chinese). Clim Environ Res, 20: 97–110
Warren J M, Meinzer F C, Brooks J R, Domec J C, Coulombe R. 2007. Hydraulic redistribution of soil water in two old-growth coniferous forests: Quantifying patterns and controls. New Phytol, 173: 753–765
Warren J M, Hanson P J, Iversen C M, Kumar J, Walker A P, Wullschleger S D. 2015. Root structural and functional dynamics in terrestrial biosphere models-evaluation and recommendations. New Phytol, 205: 59–78
Yan B Y, Dickinson R E. 2014. Modeling hydraulic redistribution and ecosystem response to droughts over the Amazon basin using Community Land Model 4.0 (CLM4). J Geophys Res-Biogeosci, 119: 2130–2143
Zeng N, Yoon J H, Marengo J A, Subramaniam A, Nobre C A, Mariotti A, Neelin J D. 2008. Causes and impacts of the 2005 Amazon drought. Environ Res Lett, 3: 014002
Zeng X. 2001. Global vegetation root distribution for land modeling. J Hydrometeorol, 2: 525–530
Zeng Y J, Xie Z H, Yu Y, Liu S, Wang L Y, Zou J, Qin P H, Jia B H. 2016. Effects of anthropogenic water regulation and groundwater lateral flow on land processes. J Adv Model Earth Syst, 8: 1106–1131
Zheng Z, Wang G. 2007. Modeling the dynamic root water uptake and its hydrological impact at the Reserva Jaru site in Amazonia. J Geophys Res, 112: G04012
Zhu S G, Chen H S, Zhang X X, Wei N, Wei S G, Yuan H, Zhang S P, Wang L L, Zhou L H, Dai Y J. 2017. Incorporating root hydraulic redistribution and compensatory water uptake in the common land model: Effects on site level and global land modeling. J Geophys Res-Atmos, 122: 7308–7322
Download references
Acknowledgements
This work was supported by the National Key Research and Development Program of China (Grant No. 2016YFA0600203), the Key Research Program of Frontier Sciences, Chinese Academy of Sciences (Grant No. QYZDY-SSW-DQC012), and the National Natural Science Foundation of China (Grant No. 41575096).
Author information
Authors and affiliations.
Beijing Meteorological Observatory, Beijing, 100089, China
Yuanyuan Wang
State Key Laboratory of Numerical Modeling for Atmospheric Sciences and Geophysical Fluid Dynamics, Institute of Atmospheric Physics, Chinese Academy of Sciences, Beijing, 100029, China
Yuanyuan Wang, Binghao Jia & Zhenghui Xie
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Binghao Jia .
Rights and permissions
Reprints and permissions
About this article
Wang, Y., Jia, B. & Xie, Z. Impacts of hydraulic redistribution on eco-hydrological cycles: A case study over the Amazon basin. Sci. China Earth Sci. 61 , 1330–1340 (2018). https://doi.org/10.1007/s11430-017-9219-5
Download citation
Received : 06 December 2017
Revised : 03 March 2018
Accepted : 16 May 2018
Published : 18 July 2018
Issue Date : September 2018
DOI : https://doi.org/10.1007/s11430-017-9219-5
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Land-surface model
- Hydraulic redistribution
- Gross primary production
- Evapotranspiration
- Find a journal
- Publish with us
- Track your research
Amazon Basin

Amazon Basin Facts
Basin Area: 5856696 5,856,696 km² 2,260,684.656 mi² km 2 Average Discharge: 193020962.967 193,020,962.967 m³/s 6,816,470,973.293 cfs 6,091,162.528 km³/y m 3 /s Type: watershed or basin , river or creek Includes Riparians: Bolivia ; Brazil ; Colombia ; Ecuador ; Guyana ; French Guiana ; Peru ; Suriname ; Venezuela ; [1] view/browse all article properties
- 1 Case Studies linked to Amazon Basin
- 2 Articles linked to Amazon Basin
- 3 Facts about Included Riparians
The Amazon Basin is located in South America.

Case Studies linked to Amazon Basin [ edit ]
Articles linked to amazon basin [ edit ].
| Riparians | Water Features |
|---|---|
| Located in this basin- Other - |
|
| Projects and Initiatives | Agreements and Treaties |
|---|---|
|
|
|
Facts about Included Riparians
| 0.91 million910,100 people | 366,819 km²141,629.608 mi² | 0 km²0 mi² | 2,820,321 m³/s99,598,696.102 cfs 89,000.87 km³/y | |
| 6.337 million6,337,000 people | 703,423 km²271,593.139 mi² | 0 km²0 mi² | ||
| 0.0017 million1,700 people | 1,566 km²604.636 mi² | 0 km²0 mi² | 95.067 m³/s3,357.259 cfs 3 km³/y | |
| 0 million0 people | 139 km²53.668 mi² | 0 km²0 mi² | 0 m³/s0 cfs 0 km³/y | |
| 1.515 million1,515,000 people | 123,295 km²47,604.466 mi² | 7,400 km²2,857.156 mi² | 95,067 m³/s3,357,259.419 cfs 3,000.029 km³/y | |
| 7.968 million7,968,000 people | 3,654,439 km²1,410,986.786 mi² | 0 km²0 mi² | 184,651,803 m³/s6,520,916,878.499 cfs 5,827,056.947 km³/y | |
| 0.0042 million4,200 people | 40,329 km²15,571.114 mi² | 0 km²0 mi² | 31,689 m³/s1,119,086.473 cfs 1,000.01 km³/y | |
| 9.295 million9,295,000 people | 951,855 km²367,513.27 mi² | 19,000 km²7,335.941 mi² | 5,418,819 m³/s191,363,786.893 cfs 171,001.671 km³/y | |
| 0.0181 million18,100 people | 14,831 km²5,726.281 mi² | 0 km²0 mi² | 3,168.9 m³/s111,908.647 cfs 100.001 km³/y |
- ^ Oregon State University.Transboundary Freshwater Spatial Database Entry for "Amazon Basin" Accessed August 15 2014 online: http://ocid.nacse.org/tfdd/map/result.php?bcode=AMZN
- Water Feature
| 193,020,962.967 m³/s (6,816,470,973.293 cfs, 6,091,162.528 km³/y) | |
| 5,856,696 km² (2,260,684.656 mi²) | |
| South America | |
| , , , , , , , and | |
| and | |
| This property is a special property in this wiki. | , , , , , , , and |
Navigation menu
Personal tools.
- View history
- Browse Case Studies
- Tools for Users
- Tools for Editors
- About AquaPedia
- Contact an Administrator
- What links here
- Related changes
- Special pages
- Printable version
- Permanent link
- Page information
- Browse properties
- Cite this page
- This page was last modified on 10 September 2014, at 15:21.
- This page has been accessed 3,232 times.
- Privacy policy
- About AquaPedia Case Study Database
- Disclaimers


Main page content
- Latin America and Caribbean
- Land and Food
Conflict over Land in the Amazon Basin, Ecuador
As is true for the entire Amazon region, the Ecuadorian Amazon region has undergone different phases of colonisation. This has created land and territorial conflict between settlers from the highlands, and indigenous peoples of the Amazon region. Meanwhile, rising interest for productive land and other natural resources, as well as development plans on the national scale, have further fueled conflict. The tensions between families of the Shuar people and settlers over access to land in the Province of Morona Santiago, Ecuador shows how current infrastructure projects in the Amazon region worsen the conflicts generated during homesteading settlement and reflect the complexity of resolving them in intercultural contexts.

Conceptual Model
Conflict history.
By the early 1970s, approximately 43,000 agricultural colonists from the highlands had moved to the Ecuadorian Amazon Basin, also known as the Oriente, in a state-led effort to integrate the Oriente through settlement. Most migrants settled in Morona Santiago and Zamora Chinchipe in the southern region, where direct routes to Cuenca and Loja in the nearby highlands existed ( Southgate et al. 2009 ). This homesteading changed the social and political structure of the Amazon region and led in some cases to peaceful coexistence, to violence in others, between different ethnic and cultural groups. Meanwhile, the biodiversity of the Amazon Basin - which covers 44% of the land area of the South American continent - is threatened by deforestation, changes in the hydrologic circle associated with changes in the global climate and water pollution ( OAS 2005 ).
Disputed property rights
Two decades ago, farmers settled in the parish of Shaimi, who formed the Puma Association (ASOPUMA, some 80 members) and got property titles. In the parish of Shaimi, up to 2008 the farmers and Shuar people lived peacefully together. The conflict began when, starting in 2011, there were the first irregular Shuar settlements in the urban zone of the parish. The four Shuar families involved were demanding their right to settle that land, because they felt that the land distribution done during colonisation times was illegal, and they never received the payment that had been agreed to at the time. There is no documentary support to confirm or contradict their version.
Rising speculative land values
This conflict developed in the context of rising speculative land values in the urban zone of the parish, because the Méndez – Morona highway linking the parish with the urban centers of the Province was completed. There were also rumors that a port was to be built, with the idea of making a connection by river and overland between Ecuador’s Pacific Ocean port of Manta with Manaus in Brazil, and from there to the Atlantic coast of South America. This strategic project is part of the Initiative to Integrate South American Regional Infrastructure (IIRSA) which includes improving regional transport, energy and telecommunications infrastructure.
Worsening of the conflict
Beginning in 2012, Shuar families made new squatting settlements, denouncing illegal tenure and ownership of the land in the Shaimi sector, arguing that they had ancestral rights to the territory. These families squatted on several farms, leading to verbal and physical aggression and death threats between the two population groups. In May 2012, the conflict increased, resulting in the death of a youth. This caused more mistrust and an environment of insecurity. For the first time, regular police and army stations were set up.
Conflict resolution
Because of the scale of the conflict, an Inter-Ministry Commission intervened, led at the time by the Ministry to Coordinate Policy, and later by the Ministry of the Interior, the National Secretariat to Manage Policy and the Ministry of Agriculture, Livestock, Aquaculture and Fishing (MAGAP) of the Province, to help organise urban planning under the leadership of the Municipality of Tiwintza, to legalise land tenure, help validate their Intercultural Life Plan, and establish lasting order.
Conflict mediation committee
A conflict mediation committee was formed with the Shuar representative (President of the Interprovincial Federation of Shuar Centers - FICSH), the Municipality of Tiwintza, and three societal leaders. Under the mediating committee, the parties prepared different proposals but no agreement was reached among the stakeholders in the conflict. Therefore, the Inter-Ministry Commission and FICSH prepared a proposal for final resolution, which was presented in July and August 2014. This proposal was based on the findings of the report on land tenure in the area, prepared by the MAGAP Conflict Management Unit, which had gathered information since mid-2012 and updated it to June 2014; and proceedings from the dialogues which had lasted nine months (17 December 2013 to 02 September 2014). This proposal recognised titles to 38 properties (26 farmer families and two Shuar families) and proposed to grant the other eight properties to the Shuar people, totaling 1772 hectares, which were basically land with no owner and no one in possession; or lots that the mestizos had allegedly abandoned.
The Shuar families' rejection of the proposed agreement
The farmers accepted the proposal in writing. The Shuar families rejected it and have stated that they would sign a peace and coexistence agreement when the ownership titles by the farmers for properties where the Shuars are currently settled have been canceled. In view of this situation, the Governor's Office, on behalf of the Inter-Ministry Commission, has stated that this proposal had already been analyzed by government agencies and is not sustainable, and anyway it would require a series of legal actions that would require time. In this context, government institutions – members of the Inter-Ministry Commission – have exhausted all administrative options for dialogue, concluded their intervention, and will let any other administrative or judicial bodies take over and resolve these disagreements.
Resilience and Peace Building
Mediation & arbitration.
Several mediation attempts were made by way of a conflict mediation committee, which included representatives from all stakeholder groups, as well as an Inter-Ministry Commission tasked with preparing a proposal for final resolution. However, these interventions have failed to resolve the disagreements between the farmers and the Shuar families. The Inter-Ministry Commission has concluded its mediation efforts.
Social inclusion & empowerment
Shuar families have been represented in dialogues by the President of the Interprovincial Federation of Shuar Centers (FICSH) and have had an opportunity to present their own proposals for review. The latest proposed agreement prepared jointly by the Inter-Ministry Commission and FICSH granted eight properties (1772 hectares) to the Shuar people. Nonetheless, the Shuar families have rejected this proposal and are demanding rights to land where they are currently settled.
Resources and Materials
- Southgate, D., Wasserstrom, R. and Reider, S. (2009) Oil Development, Deforestation, and Indigenous Populations in the Ecuadorian Amazon. Latin American Studies Association in Rio de Janeiro, Brazil, 11 - 14 June 2009
- Organization of American States: Office for sustainable Development & Environment. (2005). Amazon River Basin – Integrated and sustainable management of transboundary water resources in the Amazon River Basin. (Water project series, Number 8 - October 20

Article
- Volume 21, issue 1
- HESS, 21, 117–132, 2017
- Peer review
- Related articles
- Observations and modeling of land surface water and energy...
Assessing the impact of hydrodynamics on large-scale flood wave propagation – a case study for the Amazon Basin
Jannis m. hoch, arjen v. haag, arthur van dam, hessel c. winsemius, ludovicus p. h. van beek, marc f. p. bierkens.
Abstract. Large-scale flood events often show spatial correlation in neighbouring basins, and thus can affect adjacent basins simultaneously, as well as result in superposition of different flood peaks. Such flood events therefore need to be addressed with large-scale modelling approaches to capture these processes. Many approaches currently in place are based on either a hydrologic or a hydrodynamic model. However, the resulting lack of interaction between hydrology and hydrodynamics, for instance, by implementing groundwater infiltration on inundated floodplains, can hamper modelled inundation and discharge results where such interactions are important. In this study, the global hydrologic model PCR-GLOBWB at 30 arcmin spatial resolution was one-directionally and spatially coupled with the hydrodynamic model Delft 3D Flexible Mesh (FM) for the Amazon River basin at a grid-by-grid basis and at a daily time step. The use of a flexible unstructured mesh allows for fine-scale representation of channels and floodplains, while preserving a coarser spatial resolution for less flood-prone areas, thus not unnecessarily increasing computational costs. In addition, we assessed the difference between a 1-D channel/2-D floodplain and a 2-D schematization in Delft 3D FM. Validating modelled discharge results shows that coupling PCR-GLOBWB to a hydrodynamic routing scheme generally increases model performance compared to using a hydrodynamic or hydrologic model only for all validation parameters applied. Closer examination shows that the 1-D/2-D schematization outperforms 2-D for r 2 and root mean square error (RMSE) whilst having a lower Kling–Gupta efficiency (KGE). We also found that spatial coupling has the significant advantage of a better representation of inundation at smaller streams throughout the model domain. A validation of simulated inundation extent revealed that only those set-ups incorporating 1-D channels are capable of representing inundations for reaches below the spatial resolution of the 2-D mesh. Implementing 1-D channels is therefore particularly of advantage for large-scale inundation models, as they are often built upon remotely sensed surface elevation data which often enclose a strong vertical bias, hampering downstream connectivity. Since only a one-directional coupling approach was tested, and therefore important feedback processes are not incorporated, simulated discharge and inundation extent for both coupled set-ups is generally overpredicted. Hence, it will be the subsequent step to extend it to a two-directional coupling scheme to obtain a closed feedback loop between hydrologic and hydrodynamic processes. The current findings demonstrating the potential of one-directionally and spatially coupled models to obtain improved discharge estimates form an important step towards a large-scale inundation model with a full dynamic coupling between hydrology and hydrodynamics.
- Article (PDF, 5975 KB)
- Article (5975 KB)

- Full-text XML
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Published: 19 June 2024
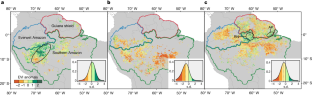
Amazon forest biogeography predicts resilience and vulnerability to drought
- Shuli Chen ORCID: orcid.org/0000-0001-9852-8716 1 ,
- Scott C. Stark ORCID: orcid.org/0000-0002-1579-1648 2 ,
- Antonio Donato Nobre 3 ,
- Luz Adriana Cuartas 4 ,
- Diogo de Jesus Amore 4 ,
- Natalia Restrepo-Coupe ORCID: orcid.org/0000-0003-3921-1772 1 , 5 ,
- Marielle N. Smith ORCID: orcid.org/0000-0003-2323-331X 2 , 6 ,
- Rutuja Chitra-Tarak 7 ,
- Hongseok Ko 1 ,
- Bruce W. Nelson ORCID: orcid.org/0000-0002-0488-6895 8 &
- Scott R. Saleska 1 , 9
Nature ( 2024 ) Cite this article
192 Accesses
86 Altmetric
Metrics details
- Climate-change ecology
- Ecosystem ecology
Amazonia contains the most extensive tropical forests on Earth, but Amazon carbon sinks of atmospheric CO 2 are declining, as deforestation and climate-change-associated droughts 1 , 2 , 3 , 4 threaten to push these forests past a tipping point towards collapse 5 , 6 , 7 , 8 . Forests exhibit complex drought responses, indicating both resilience (photosynthetic greening) and vulnerability (browning and tree mortality), that are difficult to explain by climate variation alone 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 . Here we combine remotely sensed photosynthetic indices with ground-measured tree demography to identify mechanisms underlying drought resilience/vulnerability in different intact forest ecotopes 18 , 19 (defined by water-table depth, soil fertility and texture, and vegetation characteristics). In higher-fertility southern Amazonia, drought response was structured by water-table depth, with resilient greening in shallow-water-table forests (where greater water availability heightened response to excess sunlight), contrasting with vulnerability (browning and excess tree mortality) over deeper water tables. Notably, the resilience of shallow-water-table forest weakened as drought lengthened. By contrast, lower-fertility northern Amazonia, with slower-growing but hardier trees (or, alternatively, tall forests, with deep-rooted water access), supported more-drought-resilient forests independent of water-table depth. This functional biogeography of drought response provides a framework for conservation decisions and improved predictions of heterogeneous forest responses to future climate changes, warning that Amazonia’s most productive forests are also at greatest risk, and that longer/more frequent droughts are undermining multiple ecohydrological strategies and capacities for Amazon forest resilience.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
24,99 € / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
185,98 € per year
only 3,65 € per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others

Meta-analysis reveals the effects of microbial inoculants on the biomass and diversity of soil microbial communities
Microbial competition for phosphorus limits the CO2 response of a mature forest
Increasing numbers of global change stressors reduce soil carbon worldwide
Data availability.
All remote sensing data and products (vegetation/photosynthetic indices ( https://lpdaac.usgs.gov/products/mcd19a3v006/ , http://data.globalecology.unh.edu/data/GOSIF_v2 ), climate variables ( https://disc2.gesdisc.eosdis.nasa.gov/data/TRMM_L3/TRMM_3B43.7/ , https://goldsmr4.gesdisc.eosdis.nasa.gov/data/MERRA2_MONTHLY/M2TMNXRAD.5.12.4/ , https://airs.jpl.nasa.gov/data/get-data/standard-data/ ), land cover ( https://lpdaac.usgs.gov/products/mcd12q1v006/ , https://forobs.jrc.ec.europa.eu/TMF ), tree characteristics (canopy height, https://webmap.ornl.gov/ogc/dataset.jsp?dg_id=10023_1 ) and soil texture ( https://maps.isric.org/ )) are publicly available online. The ground-based demographic validation data are publicly available in refs. 2 , 26 . The ground-based hydraulic trait validation data are publicly available in ref. 50 . The HAND data are from ref. 25 , which derived them from the digital elevation model from the Shuttle Radar Topography Mission. The soil fertility data are available in ref. 43 .
Code availability
Code for reproducing the modelling analysis and figures is posted at Code Ocean ( https://codeocean.com/capsule/2432086/tree ).
Pan, Y. et al. A large and persistent carbon sink in the world’s forests. Science 333 , 988–993 (2011).
Brienen, R. J. W. et al. Long-term decline of the Amazon carbon sink. Nature 519 , 344–348 (2015).
Article ADS CAS Google Scholar
Wigneron, J.-P. et al. Tropical forests did not recover from the strong 2015–2016 El Niño event. Sci. Adv. 6 , eaay4603 (2020).
Gatti, L. V. et al. Amazonia as a carbon source linked to deforestation and climate change. Nature 595 , 388–393 (2021).
Boulton, C. A., Lenton, T. M. & Boers, N. Pronounced loss of Amazon rainforest resilience since the early 2000s. Nat. Clim. Chang. 12 , 271–278 (2022).
Article ADS Google Scholar
Flores, B. M. et al. Critical transitions in the Amazon forest system. Nature 626 , 555–564 (2024).
Article CAS Google Scholar
Oyama, M. D. & Nobre, C. A. A new climate‐vegetation equilibrium state for Tropical South America. Geophys. Res. Lett. 30 , 2199 (2003).
Science Panel for the Amazon. Amazon Assessment Report 2021 (UN SDSN, 2021).
Saleska, S. R., Didan, K., Huete, A. R. & da Rocha, H. R. Amazon forests green-up during 2005 drought. Science 318 , 612 (2007).
Brando, P. M. et al. Seasonal and interannual variability of climate and vegetation indices across the Amazon. Proc. Natl Acad. Sci. USA 107 , 14685–14690 (2010).
Xu, L. et al. Widespread decline in greenness of Amazonian vegetation due to the 2010 drought. Geophys. Res. Lett. https://doi.org/10.1029/2011gl046824 (2011).
Yang, J. et al. Amazon drought and forest response: largely reduced forest photosynthesis but slightly increased canopy greenness during the extreme drought of 2015/2016. Glob. Change Biol. 24 , 1919–1934 (2018).
Anderson, L. O. et al. Vulnerability of Amazonian forests to repeated droughts. Philos. Trans. R. Soc. Lond. B 373 , 20170411 (2018).
Anderegg, W. R. L., Trugman, A. T., Badgley, G., Konings, A. G. & Shaw, J. Divergent forest sensitivity to repeated extreme droughts. Nat. Clim. Change 10 , 1091–1095 (2020).
Feldpausch, T. R. et al. Amazon forest response to repeated droughts. Glob. Biogeochem. Cycles 30 , 964–982 (2016).
Phillips, O. L. et al. Drought sensitivity of the Amazon rainforest. Science 323 , 1344–1347 (2009).
Esquivel-Muelbert, A. et al. Tree mode of death and mortality risk factors across Amazon forests. Nat. Commun. 11 , 5515 (2020).
Tansley, A. G. The use and abuse of vegetational concepts and terms. Ecology 16 , 284–307 (1935).
Article Google Scholar
Whittaker, R. H., Levin, S. A. & Root, R. B. Niche, habitat, and ecotope. Am. Nat. 107 , 321–338 (1973).
Jiménez-Muñoz, J. C. et al. Record-breaking warming and extreme drought in the Amazon rainforest during the course of El Niño 2015–2016. Sci. Rep. 6 , 33130 (2016).
Marengo, J. A. & Espinoza, J. C. Extreme seasonal droughts and floods in Amazonia: causes, trends and impacts. Int. J. Climatol. 36 , 1033–1050 (2016).
Longo, M. et al. Ecosystem heterogeneity and diversity mitigate Amazon forest resilience to frequent extreme droughts. N. Phytol. 219 , 914–931 (2018).
Violle, C., Reich, P. B., Pacala, S. W., Enquist, B. J. & Kattge, J. The emergence and promise of functional biogeography. Proc. Natl Acad. Sci. USA 111 , 13690–13696 (2014).
Costa, F. R. C., Schietti, J., Stark, S. C. & Smith, M. N. The other side of tropical forest drought: do shallow water table regions of Amazonia act as large-scale hydrological refugia from drought?. N. Phytol. 237 , 714–733 (2022).
Nobre, A. D. et al. Height above the nearest drainage—a hydrologically relevant new terrain model. J. Hydrol . 404 , 13–29 (2011).
Sousa, T. R. et al. Palms and trees resist extreme drought in Amazon forests with shallow water tables. J. Ecol. 108 , 2070–2082 (2020).
Esteban, E. J. L., Castilho, C. V., Melgaço, K. L. & Costa, F. R. C. The other side of droughts: wet extremes and topography as buffers of drought negative effects in an Amazonian forest. N. Phytol. 229 , 1995–2006 (2020).
Oliveira, R. S. et al. Linking plant hydraulics and the fast-slow continuum to understand resilience to drought in tropical ecosystems. N. Phytol. 230 , 904–923 (2021).
Garcia, M. N., Domingues, T. F., Oliveira, R. S. & Costa, F. R. C. The biogeography of embolism resistance across resource gradients in the Amazon. Glob. Ecol. Biogeogr. 32 , 2199–2211 (2023).
Chitra-Tarak, R. et al. Hydraulically-vulnerable trees survive on deep-water access during droughts in a tropical forest. N. Phytol. 231 , 1798–1813 (2021).
Brum, M. et al. Hydrological niche segregation defines forest structure and drought tolerance strategies in a seasonal Amazon forest. J. Ecol. 107 , 318–333 (2019).
Tumber-Dávila, S. J., Schenk, H. J., Du, E. & Jackson, R. B. Plant sizes and shapes above and belowground and their interactions with climate. N. Phytol. 235 , 1032–1056 (2022).
Giardina, F. et al. Tall Amazonian forests are less sensitive to precipitation variability. Nat. Geosci. 11 , 405–409 (2018).
ter Steege, H. et al. Continental-scale patterns of canopy tree composition and function across Amazonia. Nature 443 , 444–447 (2006).
McDowell, N. et al. Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? N. Phytol. 178 , 719–739 (2008).
Anderegg, W. R. L. et al. Meta-analysis reveals that hydraulic traits explain cross-species patterns of drought-induced tree mortality across the globe. Proc. Natl Acad. Sci. USA 113 , 5024–5029 (2016).
Lyapustin, A. I. et al. Multi-angle implementation of atmospheric correction for MODIS (MAIAC): 3. Atmospheric correction. Remote Sens. Environ. 127 , 385–393 (2012).
Li, X. & Xiao, J. A Global, 0.05-degree product of solar-induced chlorophyll fluorescence derived from OCO-2, MODIS, and reanalysis data. Remote Sens. 11 , 517 (2019).
Aragão, L. E. O. C. et al. Spatial patterns and fire response of recent Amazonian droughts. Geophys. Res. Lett. https://doi.org/10.1029/2006gl028946 (2007).
Hastie, T. J. & Tibshirani, R. J. Generalized Additive Models (CRC, 1990).
Pearl, J. Causal inference in statistics: an overview. Statist. Surv. 3 , 96–146 (2009).
Article MathSciNet Google Scholar
Aragão, L. E. O. C. et al. 21st Century drought-related fires counteract the decline of Amazon deforestation carbon emissions. Nat. Commun. 9 , 536 (2018).
Zuquim, G. et al. Making the most of scarce data: mapping soil gradients in data‐poor areas using species occurrence records. Methods Ecol. Evol. 10 , 788–801 (2019).
Hengl, T. et al. SoilGrids250m: global gridded soil information based on machine learning. PLoS ONE 12 , e0169748 (2017).
Simard, M., Pinto, N., Fisher, J. B. & Baccini, A. Mapping forest canopy height globally with spaceborne lidar. J. Geophys. Res. https://doi.org/10.1029/2011jg001708 (2011).
Christina, M. et al. Almost symmetrical vertical growth rates above and below ground in one of the world’s most productive forests. Ecosphere 2 , 1–10 (2011).
da Costa, A. C. L. et al. Effect of 7 yr of experimental drought on vegetation dynamics and biomass storage of an eastern Amazonian rainforest. N. Phytol. 187 , 579–591 (2010).
Nepstad, D. C., Tohver, I. M., Ray, D., Moutinho, P. & Cardinot, G. Mortality of large trees and lianas following experimental drought in an Amazon forest. Ecology 88 , 2259–2269 (2007).
Phillips, O. L. et al. Drought–mortality relationships for tropical forests. N. Phytol. 187 , 631–646 (2010).
Tavares, J. V. et al. Basin-wide variation in tree hydraulic safety margins predicts the carbon balance of Amazon forests. Nature 617 , 111–117 (2023).
Duffy, P. B., Brando, P., Asner, G. P. & Field, C. B. Projections of future meteorological drought and wet periods in the Amazon. Proc. Natl Acad. Sci. USA 112 , 13172–13177 (2015).
Wunderling, N. et al. Recurrent droughts increase risk of cascading tipping events by outpacing adaptive capacities in the Amazon rainforest. Proc. Natl Acad. Sci. USA 119 , e2120777119 (2022).
Makarieva, A. M. et al. The role of ecosystem transpiration in creating alternate moisture regimes by influencing atmospheric moisture convergence. Glob. Chang. Biol. 29 , 2536–2556 (2023).
Costa, M. H. et al. in Amazon Assessment Report 2021 (eds Nobre, C. et al.) Ch. 7 (UN SDSN, 2021).
Betts, M. G. et al. When are hypotheses useful in ecology and evolution? Ecol. Evol. 11 , 5762–5776 (2021).
Glass, D. J. & Hall, N. A brief history of the hypothesis. Cell 134 , 378–381 (2008).
Huete, A. et al. Overview of the radiometric and biophysical performance of the MODIS vegetation indices. Remote Sens. Environ. 83 , 195–213 (2002).
Santos, V. A. H. F. D. et al. Causes of reduced leaf-level photosynthesis during strong El Niño drought in a Central Amazon forest. Glob. Change Biol. 24 , 4266–4279 (2018).
Wu, J. et al. Partitioning controls on Amazon forest photosynthesis between environmental and biotic factors at hourly to interannual timescales. Glob. Change Biol. 23 , 1240–1257 (2017).
Wu, J. et al. Leaf development and demography explain photosynthetic seasonality in Amazon evergreen forests. Science 351 , 972–976 (2016).
Mohammed, G. H. et al. Remote sensing of solar-induced chlorophyll fluorescence (SIF) in vegetation: 50 years of progress. Remote Sens. Environ. 231 , 111177 (2019).
Sun, Y. et al. OCO-2 advances photosynthesis observation from space via solar-induced chlorophyll fluorescence. Science 358 , eaam5747 (2017).
Shekhar, A., Buchmann, N. & Gharun, M. How well do recently reconstructed solar-induced fluorescence datasets model gross primary productivity? Remote Sens. Environ. 283 , 113282 (2022).
Huffman, G. J., Adler, R. F., Bolvin, D. T. & Nelkin, E. J. in Satellite Rainfall Applications for Surface Hydrology (eds Gebremichael, M. & Hossain, F.) 3–22 (Springer, 2010).
Gelaro, R. et al. The Modern-Era Retrospective Analysis for Research and Applications, version 2 (MERRA-2). J. Clim. 30 , 5419–5454 (2017).
Aumann, H. H. Atmospheric infrared sounder on the Earth observing system. Opt. Eng. 33 , 776 (1994).
Kahn, B. H. et al. The Atmospheric Infrared Sounder version 6 cloud products. Atmos. Chem. Phys. 14 , 399–426 (2014).
Susskind, J., Blaisdell, J. M. & Iredell, L. Improved methodology for surface and atmospheric soundings, error estimates, and quality control procedures: the atmospheric infrared sounder science team version-6 retrieval algorithm. J. Appl. Remote Sens. 8 , 084994 (2014).
Sun, J. et al. Global evaluation of terrestrial near-surface air temperature and specific humidity retrievals from the Atmospheric Infrared Sounder (AIRS). Remote Sens. Environ. 252 , 112146 (2021).
Bastian, O. et al. In Development and Perspectives of Landscape Ecology (eds Bastian, O. & Steinhardt, U.) 49–112 (Springer, 2002).
Rennó, C. D. et al. HAND, a new terrain descriptor using SRTM-DEM: mapping terra-firme rainforest environments in Amazonia. Remote Sens. Environ. 112 , 3469–3481 (2008).
Brunner, I., Herzog, C., Dawes, M. A., Arend, M. & Sperisen, C. How tree roots respond to drought. Front. Plant Sci. 6 , 547 (2015).
Oliveira, R. S. et al. Embolism resistance drives the distribution of Amazonian rainforest tree species along hydro‐topographic gradients. N. Phytol. 221 , 1457–1465 (2019).
Levis, C. et al. Persistent effects of pre-Columbian plant domestication on Amazonian forest composition. Science 355 , 925–931 (2017).
Fan, Y., Li, H. & Miguez-Macho, G. Global patterns of groundwater table depth. Science 339 , 940–943 (2013).
Cunha, H. F. V. et al. Direct evidence for phosphorus limitation on Amazon forest productivity. Nature 608 , 558–562 (2022).
Chadwick, O. A., Derry, L. A., Vitousek, P. M., Huebert, B. J. & Hedin, L. O. Changing sources of nutrients during four million years of ecosystem development. Nature 397 , 491–497 (1999).
Darela-Filho, J. P. et al. Reference maps of soil phosphorus for the pan-Amazon region. Earth Syst. Sci. Data 16 , 715–729 (2024).
Liu, H.-Y., Sun, W.-N., Su, W.-A. & Tang, Z.-C. Co-regulation of water channels and potassium channels in rice. Physiol. Plant. 128 , 58–69 (2006).
Fan, Y., Miguez-Macho, G., Jobbágy, E. G., Jackson, R. B. & Otero-Casal, C. Hydrologic regulation of plant rooting depth. Proc. Natl Acad. Sci. USA 114 , 10572–10577 (2017).
Hasanuzzaman, M., Araújo, S. & Gill, S. S. The Plant Family Fabaceae: Biology and Physiological Responses to Environmental Stresses (Springer, 2021).
Friedl, M. A. et al. MODIS Collection 5 global land cover: algorithm refinements and characterization of new datasets. Remote Sens. Environ. 114 , 168–182 (2010).
Hess, L. L. et al. Wetlands of the lowland Amazon basin: extent, vegetative cover, and dual-season inundated area as mapped with JERS-1 synthetic aperture radar. Wetlands 35 , 745–756 (2015).
Gómez, J., Schobbenhaus, C. & Montes, N. E. Geological Map of South America 2019. Scale 1: 5 000 000. Commission for the Geological Map of the World (CGMW), Colombian Geological Survey and Geological Survey of Brazil (2019); https://doi.org/10.32685/10.143.2019.929 .
Vancutsem, C. et al. Long-term (1990–2019) monitoring of forest cover changes in the humid tropics. Sci. Adv. 7 , eabe1603 (2021).
ForestPlots.net, et al. Taking the pulse of Earth’s tropical forests using networks of highly distributed plots. Biol. Conserv. 260 , 108849 (2021).
Shi, H. et al. Assessing the ability of MODIS EVI to estimate terrestrial ecosystem gross primary production of multiple land cover types. Ecol. Indic. 72 , 153–164 (2017).
Sims, D. A. et al. On the use of MODIS EVI to assess gross primary productivity of North American ecosystems. J. Geophys. Res. 111 , G04015 (2006).
Rahman, A. F., Sims, D. A., Cordova, V. D. & El-Masri, B. Z. Potential of MODIS EVI and surface temperature for directly estimating per-pixel ecosystem C fluxes. Geophys. Res. Lett. 32 , L19404 (2005).
Sims, D. A. et al. A new model of gross primary productivity for North American ecosystems based solely on the enhanced vegetation index and land surface temperature from MODIS. Remote Sens. Environ. 112 , 1633–1646 (2008).
Huete, A. R. et al. Amazon rainforests green-up with sunlight in dry season. Geophys. Res. Lett. 33 , L06405 (2006).
Huete, A. R. et al. Multiple site tower flux and remote sensing comparisons of tropical forest dynamics in Monsoon Asia. Agric. For. Meteorol. 148 , 748–760 (2008).
Huete, A. R. Vegetation indices, remote sensing and forest monitoring. Geogr. compass 6 , 513–532 (2012).
Samanta, A. et al. Amazon forests did not green-up during the 2005 drought. Geophys. Res. Lett. 37 , L05401 (2010).
Morton, D. C. et al. Amazon forests maintain consistent canopy structure and greenness during the dry season. Nature 506 , 221–224 (2014).
Saleska, S. R. et al. Dry-season greening of Amazon forests. Nature 531 , E4–E5 (2016).
Maeda, E. E., Heiskanen, J., Aragão, L. E. O. C. & Rinne, J. Can MODIS EVI monitor ecosystem productivity in the Amazon rainforest? Geophys. Res. Lett. 41 , 7176–7183 (2014).
Magney, T. S., Barnes, M. L. & Yang, X. On the covariation of chlorophyll fluorescence and photosynthesis across scales. Geophys. Res. Lett. 47 , e2020GL091098 (2020).
Chambers, J. Q. et al. Respiration from a tropical forest ecosystem: partitioning of sources and low carbon use efficiency. Ecol. Appl. 14 , 72–88 (2004).
Smith, M. N. et al. Seasonal and drought-related changes in leaf area profiles depend on height and light environment in an Amazon forest. N. Phytol. 222 , 1284–1297 (2019).
Janssen, T. et al. Drought effects on leaf fall, leaf flushing and stem growth in the Amazon forest: reconciling remote sensing data and field observations. Biogeosciences 18 , 4445–4472 (2021).
Quesada, C. A. et al. Basin-wide variations in Amazon forest structure and function are mediated by both soils and climate. Biogeosciences 9 , 2203–2246 (2012).
Lloyd, J. et al. Edaphic, structural and physiological contrasts across Amazon Basin forest–savanna ecotones suggest a role for potassium as a key modulator of tropical woody vegetation structure and function. Biogeosciences 12 , 6529–6571 (2015).
Wold, S., Esbensen, K. & Geladi, P. Principal component analysis. Chemometr. Intellig. Lab. Syst. 2 , 37–52 (1987).
Doan, H. T. X. & Foody, G. M. Reducing the impacts of intra-class spectral variability on soft classification and its implications for super-resolution mapping. In Proc. IEEE International Geoscience and Remote Sensing Symposium 2585–2588 (IEEE, 2007).
Malhi, A. & Gao, R. X. PCA-based feature selection scheme for machine defect classification. IEEE Trans. Instrum. Meas. 53 , 1517–1525 (2004).
Canty, M. J. Image Analysis, Classification and Change Detection in Remote Sensing: With Algorithms for ENVI/IDL and Python, Third Edition (CRC, 2014).
Grafton, R. Q. et al. Realizing resilience for decision-making. Nat. Sustain. 2 , 907–913 (2019).
Nikinmaa, L. et al. Reviewing the use of resilience concepts in forest sciences. Curr. For. Rep. 6 , 61–80 (2020).
Gunderson, L. H. Ecological resilience—in theory and application. Annu. Rev. Ecol. Syst. 31 , 425–439 (2000).
Samanta, A. et al. Amazon forests did not green-up during the 2005 drought. Geophys. Res. Lett. https://doi.org/10.1029/2009gl042154 (2010).
Haining, R. P. & Haining, R. Spatial Data Analysis: Theory and Practice (Cambridge Univ. Press, 2003).
Burnham, K. P., Anderson, D. R. & Huyvaert, K. P. AIC model selection and multimodel inference in behavioral ecology: some background, observations, and comparisons. Behav. Ecol. Sociobiol. 65 , 23–35 (2011).
Clark, J. S. Models for Ecological Data (Princeton Univ. Press, 2020).
Pearl, J. Causality (Cambridge Univ. Press, 2009).
Ankan, A., Wortel, I. M. N. & Textor, J. Testing graphical causal models using the R package ‘dagitty’. Curr. Protoc. 1 , e45 (2021).
Arif, S. & MacNeil, M. A. Applying the structural causal model framework for observational causal inference in ecology. Ecol. Monogr. 93 , e1554 (2023).
Arif, S. & MacNeil, M. A. Predictive models aren’t for causal inference. Ecol. Lett. 25 , 1741–1745 (2022).
Wood, S. N. Generalized Additive Models: An Introduction with R, Second Edition (CRC, 2017).
Quaresma, M., Carpenter, J. & Rachet, B. Flexible Bayesian excess hazard models using low-rank thin plate splines. Stat. Methods Med. Res. 29 , 1700–1714 (2020).
Schempp, W. & Zeller, K. eds. Constructive Theory of Functions of Several Variables: Proceedings of a Conference Held at Oberwolfach, April 25 - May 1, 1976 (Springer, 1977).
Laurance, W. F. et al. Relationship between soils and Amazon forest biomass: a landscape-scale study. For. Ecol. Manage. 118 , 127–138 (1999).
Levine, N. M. et al. Ecosystem heterogeneity determines the ecological resilience of the Amazon to climate change. Proc. Natl Acad. Sci. USA 113 , 793–797 (2016).
Addicott, E. T., Fenichel, E. P., Bradford, M. A., Pinsky, M. L. & Wood, S. A. Toward an improved understanding of causation in the ecological sciences. Front. Ecol. Environ. 20 , 474–480 (2022).
Textor, J., van der Zander, B., Gilthorpe, M. S., Liskiewicz, M. & Ellison, G. T. Robust causal inference using directed acyclic graphs: the R package ‘dagitty’. Int. J. Epidemiol. 45 , 1887–1894 (2016).
Google Scholar
de Castilho, C. V. et al. Variation in aboveground tree live biomass in a central Amazonian Forest: effects of soil and topography. For. Ecol. Manage. 234 , 85–96 (2006).
Velleman, P. F. & Welsch, R. E. Efficient computing of regression diagnostics. Am. Stat. 35 , 234–242 (1981).
Webb, C. O., Cannon, C. H. & Davies, S. J. in Tropical Forest Community Ecology (eds Carson, W. P. & Schnitzer, S. A.) 79–97 (Wiley–Blackwell, 2008).
Humboldt’s legacy. Nat. Ecol. Evol. 3 , 1265–1266 (2019).
Reichstein, M., Bahn, M., Mahecha, M. D., Kattge, J. & Baldocchi, D. D. Linking plant and ecosystem functional biogeography. Proc. Natl Acad. Sci. USA 111 , 13697–13702 (2014).
Nelson, B. W., Gonçalves, N. B., Chen, S. & Saleska, S. Persistent effect of 2015 El Niño drought on NIR reflectance of central Amazon upland forests? In Anais do XX Simpósio Brasileiro de Sensoriamento Remoto 1636–1638 (INPE, 2023).
Oliveira, R. S., Dawson, T. E., Burgess, S. S. O. & Nepstad, D. C. Hydraulic redistribution in three Amazonian trees. Oecologia 145 , 354–363 (2005).
Nunes, M. H. et al. Forest fragmentation impacts the seasonality of Amazonian evergreen canopies. Nat. Commun. 13 , 917 (2022).
Van der Meer, T., Te Grotenhuis, M. & Pelzer, B. Influential cases in multilevel modeling: a methodological comment. Am. Sociol. Rev. 75 , 173–178 (2010).
Boers, N., Marwan, N., Barbosa, H. M. J. & Kurths, J. A deforestation-induced tipping point for the South American monsoon system. Sci. Rep. 7 , 41489 (2017).
Download references
Acknowledgements
We thank J. Schietti for early discussions of the idea that remote sensing might be used to investigate the effect of water-table depth on forest drought response; T. R. Sousa for sharing and discussing plot-based forest demographic data (from along the BR-319 road) 26 ; G. Zuquim for sharing an early version of mapped basin-wide soil fertility data 43 ; H. ter Steege for sharing mapped basin-wide tree characteristics data 34 ; T. R. Sousa and J. Schietti for comments on an earlier version of the manuscript; L. Alves for advice on forest demography plots; R. Palacios for recommending the use of GAM models; M. N. Garcia for discussions about soil fertility; N. Boers for advice on the South American monsoon system; T. C. Taylor and V. Ivanov for discussions; J. Cronin and S. McMahon for detailed advice and comments; and S.C.’s doctoral dissertation committee members W. K. Smith, J. Hu and B. Enquist for constructive criticism and advice on the direction of this work. This work was supported by US National Aeronautics and Space Administration, fellowship 80NSSC19K1376 (S.C.); US National Science Foundation, DEB grant 1950080 (S.C.S. and M.N.S.); US National Science Foundation, DEB grant 2015832 (S.R.S.); US National Science Foundation, DEB grant 1754803 (S.R.S., N.R.-C.), US National Science Foundation, DEB grant 1754357 (S.C.S.); Brazil National Council for Scientific and Technological Development (CNPq) scholarships 371626/2022-6, 372734/2021-9,381711/2020-0 (D.d.J.A.); and US Department of Energy’s Next Generation Ecosystem Experiments-Tropics (R.C.-T.).
Author information
Authors and affiliations.
Department of Ecology and Evolutionary Biology, University of Arizona, Tucson, AZ, USA
Shuli Chen, Natalia Restrepo-Coupe, Hongseok Ko & Scott R. Saleska
Department of Forestry, Michigan State University, East Lansing, MI, USA
Scott C. Stark & Marielle N. Smith
National Institute for Space Research (INPE), São José dos Campos, Brazil
Antonio Donato Nobre
National Center for Monitoring and Early Warning of Natural Disasters (CEMADEN), São José dos Campos, Brazil
Luz Adriana Cuartas & Diogo de Jesus Amore
Cupoazu LLC, Etobicoke, Ontario, Canada
Natalia Restrepo-Coupe
School of Environmental and Natural Sciences, College of Science and Engineering, Bangor University, Bangor, UK
Marielle N. Smith
Los Alamos National Laboratory, Earth and Environmental Sciences, Los Alamos, NM, USA
Rutuja Chitra-Tarak
Brazil’s National Institute for Amazon Research (INPA), Manaus, Brazil
Bruce W. Nelson
Department of Environmental Sciences, University of Arizona, Tucson, AZ, USA
Scott R. Saleska
You can also search for this author in PubMed Google Scholar
Contributions
S.C. and S.R.S. designed the analysis, based on early conception by A.D.N. and S.R.S., and on funded proposals to investigate ‘the other side of tropical forest drought’ led by S.C.S., M.N.S. and S.R.S. (from NSF) and by S.C. and S.R.S. (from NASA). A.D.N., L.A.C. and D.d.J.A. updated their HAND data product and interpreted it for this analysis. B.W.N. and N.R.-C. contributed remote-sensing expertise and analysis. R.C.-T. contributed statistical modelling expertise and analysis. H.K. contributed code, especially for the variogram analysis. S.C. organized the datasets (with assistance from N.R.-C.), conducted the analysis and wrote the initial draft. S.C., S.R.S. and S.C.S. revised the draft. All of the authors contributed to writing the final version.
Corresponding authors
Correspondence to Shuli Chen or Scott R. Saleska .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Peer review
Peer review information.
Nature thanks Christopher Baraloto, David Bauman, Jovan Tadic and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended data fig. 1 drought maps 2005, 2010 and 2015/16 droughts and gosif-based forest responses droughts..
(a)-(c): Maximum cumulative water deficit (MCWD) standardized anomalies. (relative to the long term mean MCWD across years, blue=positive, orange=negative) during drought for ( a ) 2005, ( b ) 2010, and ( c ) 2015 droughts. MCWD is calculated (see Methods, ‘Climate variables’) as the maximum water deficit reached for each hydrologic year (from May of the nominal year to the following April). The “drought region” is defined as pixels whose MCWD anomaly is more than one SD below the mean (light orange to red). (d)-(f): GOSIF-based forest response to droughts . GOSIF anomalies during drought, relative to the long term mean GOSIF (green=positive, orange=negative) in drought regions for the ( d ) 2005, ( e ) 2010 and ( f ) 2015 droughts, respectively. ( g ) EVI (left axis) and GOSIF (right axis) anomalies in the 2005 drought elliptical region (as depicted in Figs. 1a , 2a , and here in Extended Data Fig. 1d ) show consistent patterns versus HAND (bin averages ±95% CI, with N = 6,547 5-km pixels for both EVI and GOSIF); ( h ) GOSIF anomalies (bin averages points ±95% CI and solid regression line) vs. water-table depths (indexed by HAND) support hypothesis 1 (with negative slopes, consistent with EVI in Fig. 3a ) for the 2005 (green, slope = −0.016 ± 0.006 SE m −1 ), 2010 (purple, slope = −0.012 ± 0.003 SE m −1 ), and 2015 (blue, slope = −0.010 ± 0.003 SE m −1 ) droughts, paired with HAND distributions in each drought region (bottom graphs, right axis, with N = 34,980, 30,004, 43,475 5-km pixels for 2005, 2010, and 2015 droughts, respectively).
Extended Data Fig. 2 Ecotope factors of the Amazon basin.
( a ) Height Above Nearest Drainage (HAND), a proxy for water-table depth 25 ; ( b ) Soil fertility, as exchangeable base cation concentrations 43 ; ( c ) Average forest heights as acquired by lidar 45 ; ( d ) Soil sand content 44 ; ( e ) Proportion of trees belonging to the Fabaceae family 34 ; ( f ) MCWD variability (see the ‘Climate anomalies for drought definition and mapping’ section of methods ), in terms of the standard deviation of the long-term MCWD timeseries. High variance in climate and low soil fertility in Guiana shield might contribute to the greatest proportion of trees belonging to the family Fabaceae with the very high wood density; ( g ) Averaged minimum monthly precipitation (low=green, high=orange). The north-west everwet Amazon is distinguished by lacking a dry season (precipitation exceeds evapotranspiration). ( h ) Community-weighted wood density 34 . Panels a-d are used as ecotope predictors in the GAM analysis of Supplementary Table 1 . (Data sources: see the ‘Climate variables’ and ‘Climate anomalies for drought definition and mapping’ sections of methods ).
Extended Data Fig. 3 Spatial distributions of climate dynamics in the 2005, 2010 and 2015 droughts.
a – i , Spatial distributions of climate dynamics in the 2005 (left column), 2010 (middle column) and 2015 (right column) droughts for: (a)-(i): Drought dynamics showing drought onset date (row 1, a-c), drought end date (row 2, d-f), and drought duration (row 3, g-i, end date minus start date). Pixel-by-pixel drought responses (EVI in Figs. 1 – 4 ; or GOSIF in Extended Data Figs. 1 & 5 ) are taken as the standardized anomalies that occur during the pixel-specific drought period defined here. (j)-(r): climatic anomalies of: photosynthetic active radiation (PAR) (row 4, j-l), vapor pressure deficit (VPD) (row 5, m-o), and precipitation (row 6, p-r). precipitation (Data source: see the ‘Climate variables’ section of methods ).
Extended Data Fig. 4 Regions in the Amazon basin.
that emerge from a principal components analysis (PCA) followed by classification: ( a ) PCA of the Amazon basin 0.4° x 0.4° pixel data (coloured according to a supervised classification into three classes identified by variance minimization), projected onto their first two principal components, which are composed mainly of three dimensions, one defined by wood density and proportions of the family Fabaceae (first principal component, horizontal axis), one defined by minimum monthly precipitation and MCWD variability (second principal component, vertical axis), and a third defined mainly by soil fertility; the classes are significantly separated in PCA space (psuedo-F ratio =950, df=2, 3805, p ~ 0, permanova test); ( b ) The Amazon pixels coloured according to their class (corresponding to the colours in a), showing that the classification of (a) maps pixels into distinct, mostly contiguous spatial regions.) ( c ) Standardized values, for each region, of each group of characteristics (ordered by water availability, soil fertility, and tree traits/characteristics), illustrate distinct regional niches: the everwet Amazon is highest in minimum precipitation and lowest (highest negative) in MCWD variability; the Southern Amazon is moderately high in mean fertility, and the Guiana Shield has the tallest mean forest height and greatest wood density. ( d ) scree plot of the eigenvalues (principal components) of the PCA shown in (a), plotted in rank order.
Extended Data Fig. 5 Amazon forest drought responses in different regions using the EVI and GOSIF remote sensing indices.
Amazon forest EVI (top row) and GOSIF (bottom row) responses to multiple droughts in the Guiana shield (left column) and the ever-wet northwest (right column). These generally do not support the “other side of drought” hypothesis 1, because they show generally consistently positive slopes with water-table depth (HAND), in contrast to negative slope responses in the Southern Amazon (Fig. 3a ). Plots show observations (bin average points ±95% CI, and solid regression lines) and unified multi-drought GAM predictions (±95% CI shaded region, for models in Supplementary Table 1b, c ), with climate fixed to region-wide median drought conditions for each drought.) Observations for EVI (a-b): N = 83 and 666 0.4° pixels for 2005 and 2015 droughts respectively, in the Guiana shield (a), and N = 147, 368, and 648 for 2005, 2010 and 2015 droughts respectively in the ever-wet Amazon (b). Observations for GOSIF (c-d): N = 1876, and 25,460 5-km pixels for 2005 and 2015 droughts, respectively, in Guiana shield (c), and N = 1,914, 8,261, and 19,918 for 2005, 2010 and 2015 droughts, respectively, in the ever-wet Amazon (d). Purple points (2010) are not shown in panels a,c, because the 2010 drought did not significantly affect the Guiana shield.
Extended Data Fig. 6 Implementing Structured Causal Modeling (SCM) of Amazon forest drought response using Directed acyclic graphs (DAGs).
a – d , Development of a Directed acyclic graph (DAG) representing the structure of factors influencing tropical forest responses to drought. ( a ) Initially hypothesized DAG characterizing the causal relationships among climatic, environmental, and forest variables (measured variables depicted as blue nodes, unmeasured rooting depth is depicted in grey) leading to forest drought response (other colour node), with arrows representing the hypothesized causal links. ( b ) DAG-data consistency tests for initial DAG , with the largest 20 approximated non-linear correlation coefficients (estimated via root mean square error of approximation, RMSEA) between unlinked variables in (a). (Note: unlinked variables in a DAG are hypothesized to have zero correlation or zero conditional correlation; thus, the second row of panel b tests “DR_ | | _DSL | DL” -- whether DR is independent of DSL conditioned on DL, by estimating the non-linear correlation between DR and the residuals of DSL regressed on DL.) Correlations greater than an acceptability threshold (dashed vertical lines at ±0.30) fail the test of conditional independence, addressed by adding to the DAG either a direct causal link (indicated by a green symbol), or links to a common cause (pink symbol) (such added arrows are included in panel c). ( c ) Final DAG after correcting for conditional independency inconsistencies of the initial DAG in A, in light of ecological considerations. Also illustrates use of the backdoor criterion to determine the causal effect of ‘drought length (DL)’ (the exposed predictor node and associated forward causal paths, in green) on forest drought response (corresponding to the model in Extended Data Fig. 10c ), while blocking the confounding variable dry season length, DSL (hypothesized to itself affect DL) and its associated causal backdoor paths (which are considered non-causal paths with respect to the exposed variable DL) (in pink). ( d ) DAG-Data consistency tests for final DAG (panel c), showing the largest 20 RMSEA values. (e)-(j): GAM regression model predictions (±95% CI shaded region) of causal effects of different variables derived from DAG, employing backdoor criterion, for the Southern Amazon, average across all three droughts: ( e ) of HAND (no backdoor to be blocked) (f) of PAR (adjusting for back door paths through drought length, dry season length) ( g ) of Drought length (adjusting for back door path through dry season length) on EVI responses (adjusted EVI prediction) ; the whole Amazon basin during the 2015 drought: ( h ) of forest height, categorized by shallow (blue, HAND = 0-10 m) and deep (red, HAND = 20–40 m) water tables (adjusting for back door paths through soil fertility, soil texture and dry season length), ( i ) of soil fertility (adjusting for back door path through dry season length) ( j ) of soil texture (no backdoor path to be blocked).
Extended Data Fig. 7 The sensitivities of forest drought response to soil texture and drought timing.
( a ) The sensitivity of forest response to soil texture (sand content) and water- table depth (HAND) in basin-wide GAM analysis. GAM-predicted adjusted EVI anomaly (left axis) versus soil sand content (%), with water table-depth in colour (shallow=blue to deep=red), paired with distributions of mean forest height in each soil texture bin (bottom graph, right axis, with N = 3,318, and 1,142 0.4° pixels for shallow and deep water tables, respectively). ‘Adjusted’ GAM predictions are made by setting non-displayed predictors (climate variables, tree-height, soil fertility) to their median values during the drought. (b)-(d): The sensitivity of forest responses to dry versus wet season drought periods, across the three-droughts: ( b ) distribution of the proportion of drought that was in the dry season (0 = all in the wet season to 1= all in the dry season) for drought-affected pixels in each of the three droughts; ( c ) GAM-predicted EVI anomaly versus PAR, for different proportions of dry season drought (blue=all wet to red=all dry, corresponding to coloured tick marks in the vertical axis of b). ( d ) Adjusted EVI anomaly from GAM prediction versus drought length, for different proportions of dry-season drought (blue to red, as in panel c).
Extended Data Fig. 8 Scale-dependence of Southern Amazon forest responses to drought, showing that detected response patterns are largely invariant across different scales of analysis.
( a ) At 0.4 degree (40-km) scale (across the Southern Amazon. all three droughts): Climate-adjusted EVI responses (standardized anomalies from MODIS) vs. water-table depths (indexed by HAND) for observations (solid points ±95% CI and solid regression line) and for unified multi-drought GAM predictions (model of Supplementary Table 1a , shaded bands and dashed regression line slopes) for the 2005 (green, slope = −0.019 ± 0.001 SE m −1 ), 2010 (purple, slope = −0.020 ± 0.002 SE m −1 ), and 2015 (blue, slope = −0.028 ± 0.002 SE m −1 ) droughts (with N = 1,384, 1,673, and 1,837 0.4° pixels for 2005, 2010, and 2015 droughts, respectively); ( b ) At 1-km scale (across the Southern Amazon, all three droughts), as in (a): climate-adjusted EVI responses vs. HAND for observations (solid points and regression line) and corresponding GAM (with the same Supplementary Table 1a model now fit at 1 km scale, revealing autocorrelation in observations causing too-narrow confidence bands, and slight model underpredictions of the extremes of the 2005 greenup and the 2010 browdown, but maintaining the similar negative dependence on HAND across all droughts); ( c ) At 30 to 180 m scales (for a forest region around Manaus, 2015-2016 drought only): Delta EVI, the fraction change in EVI due to the drought = (after-drought EVI (July 2016) - pre-drought EVI (August 2015))/pre-drought EVI (Landsat OLI8, at 30 m resolution) vs. water-table depths (indexed by HAND) for Landsat observations (solid points ±95% CI and solid regression line) at native (30 m) and aggregated to 90 and 180-m scales (with N = 105,359, 11,901, and 2,999 pixels for 30-m, 90-m, and 180-m scales, respectively). Also shown in the bottom of each panel is the distribution of water-table depth (HAND proxy) at each scale. Aggregations to larger (coarser) scales induce an apparent regression towards the mean in the water-table depth distributions (as more extreme water-table depths at finer scales become diluted by averaging to large scales), while similar dilution of extremes in EVI response (not shown) preserves the overall relation between EVI responses and watertable depth (especially evident in the Landsat analysis where the slopes through data aggregated at different scales do not detectably differ).
Extended Data Fig. 9 Remote sensing validation with forest inventory plot demography.
( a ) Remotely sensed map of MAIAC EVI (1-km resolution), overlaid with aboveground NPP (ANPP) rates from 321 ground-monitored forest plots (red circles, % standing biomass y −1 ) as aggregated to 1 degree grid plots (RAINFOR plots in Brienen et al. 2 ), with both EVI and ANPP taken during the 2000–2011 interval. ANPP rate is calculated as Aboveground Biomass (AGB) gain (Mg/(ha·yr)) (total annual AGB productivity of surviving trees plus recruitment, plus inferred growth of trees that died between censusing intervals) divided by initial AGB (Mg/ha) (standing above ground biomass at the start of the census interval). ( b ) ANPP rates as predicted by EVI (points from (a) plus solid regression line with statistics; Dashed line and associated statistics in grey represent linear regression without the high leverage point, shown in red, defined by Cook’s distances > 4/n, where n=number of points 134 ). EVI is the mean extracted from intervals matching the average census interval of the corresponding plots in Brienen et al. 2 (c)–(e) MAIAC EVI anomalies (1-km pixels) versus ground-monitored tree demography in shallow water table forests during the 2015-2016 drought 26 for : (c) mortality, (d) recruitment, and (e) mortality:recruitment ratios in 1-ha plots. (f)–(h): GOSIF anomalies (5-km pixels) versus ground-monitored (f) mortality, (g) recruitment, and (h) mortality:recruitment ratios; Solid lines and statistics (R 2 and p-values) represent standard linear regression fits to all data. Red points, if they exist, are high leverage, i.e. with Cook’s distances > 4/n, where n=number of points 134 , and dotted lines and associated statistics in grey represent standard linear regressions without such points, showing that remote detection of ground-derived demographic trends is robust. R 2 values reported here are consistent with the expectation that they should be less than for remote detection of tropical forest GPP (R 2 = 0.5-0.7), because GPP contributes only partially to the NPP driver of demography (as discussed in the 'Validation by forest plot metrics of demography and of physiological drought tolerance' section of Methods). Considering multple comparisons (six regressions), the probability, under the null hypothesis, of seeing five or more significant regresssions out of six is p = 0.000002 (Binomial test).
Extended Data Fig. 10 Modeled forest response to the 2015 drought and implications of the derived map of Amazon forest biogeography.
a – c , Forest response to the 2015 drought in drought-affected pixels. ( a ) Observed EVI anomalies (resampled at 0.4 degrees to match model resolution which accounts for spatial autocorrelation (see Supplementary Fig. 1 ). ( b ) GAM-predicted EVI anomalies (model of Supplementary Table 1d ). ( c ) Residual EVI anomalies (panel a observations minus panel b predictions). The GAM well-predicts the pattern of response (Panel b), but under-estimates the extremes of the responses (as evident from residuals in panel c continuing to show greening/browning patterns beyond the predictions). ( d ) Map of Amazon forest biogeography of resilience/vulnerability, overlaid with mean winds (arrows, at height 650 hPa) and location of the arc of deforestation . The most productive as well as the most vulnerable forests (in red) are also those most experiencing deforestation (in the “arc of deforestation”) which is causing local climatic warming/drying 4 , further stressing these vulnerable forests. These “arc of deforestation”/vulnerable forests are often upwind forests 135 (especially when the Intertropical convergence zone, ITCZ, swings to the south) that are critical for hydrological recycling in the Amazon.
Supplementary information
Supplementary information, reporting summary, peer review file, rights and permissions.
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Reprints and permissions
About this article
Cite this article.
Chen, S., Stark, S.C., Nobre, A.D. et al. Amazon forest biogeography predicts resilience and vulnerability to drought. Nature (2024). https://doi.org/10.1038/s41586-024-07568-w
Download citation
Received : 28 November 2022
Accepted : 15 May 2024
Published : 19 June 2024
DOI : https://doi.org/10.1038/s41586-024-07568-w
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
By submitting a comment you agree to abide by our Terms and Community Guidelines . If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.
Inundation mapping using hydraulic modeling with high-resolution remote sensed data: a case study in the Acre River Basin, Brazil
- Antunes da Silva, Larissa
- Rudorff, Conrado
- Ovando, Alex
- Pimentel, Alan
- Cuartas, Luz Adriana
- dos Santos Alvalá, Regina Célia
Considering the impacts of climate change in recent decades that have exacerbated the frequency and intensity of floods worldwide and especially in the Amazon region. The city of Rio Branco, located in the southwest of Brazil's Amazon region, has been severely affected by a combination of urbanization in flood-prone areas and increasing floods in the Acre River. In addressing this challenge, accurate inundation mapping plays a pivotal role in shaping effective flood risk reduction strategies. This study employs hydraulic simulations of the Acre River, utilizing the HEC-RAS 1D model. The modeling process integrates a high-resolution digital terrain model, acquired through Light Detection and Ranging (LiDAR), and a rich dataset encompassing conventional and unconventional information from the three most significant historical floods in Rio Branco in 2012, 2015, and 2023. To ensure the precision of the simulations, calibration of the roughness coefficient was conducted for steady-state scenarios, drawing upon a diverse range of observed stream-gauge data. The evaluation of water elevation in steady state reveals an impressive mean error of just 0.01 m, underscoring the model's accuracy. Moving beyond steady-state simulations, the study evaluates unsteady-state scenarios by calculating the root mean square error (RMSE). Results showcase commendable accuracies of 0.22, 0.25, and 0.26 m for the 2012, 2015, and 2023 floods, respectively. Flooding extent simulation was assessed using the Critical Success Index (C) from two optical aerial survey images recorded during the 2012 and 2015 floods and one optical satellite image recorded during the 2023 flood. The accuracy was 0.88 (2012) and 0.84 (2015) for optical aerial survey images and 0.97 for the optical satellite image (2023). Images from Google Street View recorded after 2012 flood event containing high-water marks were used to evaluate the accuracy of maximum water depth simulation along the floodplain and presented a mean error of 0.17 m.
- Flood events;
- Flood hazard mapping;
- High-water mark;
- Hydraulic model;

IMAGES
VIDEO
COMMENTS
1. Introduction. The Amazon River Basin (ARB) encompasses the entire eastern and central parts of South America, covering over 6,100,000 km 2, which constitutes 44% of the South American continent.Covered mostly by tropical rainforest, it harbors the greatest biodiversity on Earth (constituting 10-15% of global land biodiversity), serves as a sink for greenhouse gasses by absorbing up to 5% ...
Case study of a tropical rainforest setting to illustrate and analyse key themes in water and carbon cycles and their relationship to environmental change and human activity. Amazon Forest The Amazon is the largest tropical rainforest on Earth. It sits within the Amazon River basin, covers some 40% of the South American continent and as you can ...
Case Study: The Amazonian Road Decision. The proposed Pucallpa-Cruzeiro do Sul will connect the Amazon's interior to urban centers and export markets in Peru and Brazil. However, critics are worried that the road will also create new opportunities for illegal logging and infringe on the territory of indigenous communities and wildlife.
Create your personal learning account. Register for FREE at http://www.deltastep.comDeltaStep is a social initiative by graduates of IIM-Ahmedabad, IIM-Banga...
The Amazon river is home to thousands of unique species of flora and fauna. As a result, the Amazon river has been home to many wonderful civilizations like ancient Mayans and the Incas etc. Let's throw a light on the various features of the Amazon basin. The Climate. Amazon basin is situated in the equatorial region which is hot and humid ...
A case study of a sparsely populated area - Himalayan Mountains ... Case Study - Ganges/Brahmaputra River Basin; The Great Floods of 2000; Kerala flood case study; Rocks, Resources and Scenery ... Volunteers work with local Amazon youth who study at the Yachana Technical High School where learning is focused on five main areas:
Amazon River basin, especially on its northern and southern edges. Biome characteristics 1. Climate The Amazonian climate is hot and wet; for example, at Uaupes in Brazil on the equator, mean monthly temperatures range between 25 and 27°C and rainfall averages 2677mm per annum. Most precipitation occurs as heavy convectional storms, and skies
Basin-Scale River Runoff Estimation from GRACE Gravity Satellites, Climate Models and In Situ Observations: a Case Study in the Amazon Basin River runoff is estimated as a water budget residual using Gravity Recovery and Climate Experiment (GRACE) terrestrial water storage time series, ERA5 reanalysis data, and precipitation observations for January 2003 through December 2015 for the Obidos ...
Case Study: Life in the Amazon River Basin. Available Answers. 1. Fill in the blanks: The Amazon Basin is spread over _____ countries. The Amazon Basin is covered by equatorial _____. The native Americans are called _____. _____ is a deadly fish found in the waters of Amazon Basin.
3 Case Study and Data. The Amazon River and its tributaries constitute the largest volume of liquid freshwater on land. The Amazon has the largest rate of freshwater discharge into the ocean, being about 16% of the total water carried to the oceans by rivers. ... We study the Amazon basin and its nine subbasins (numbered 500-508), covering an ...
The Amazon in context. Tropical rainforests are often considered to be the "cradles of biodiversity.". Though they cover only about 6% of the Earth's land surface, they are home to over 50% of global biodiversity. Rainforests also take in massive amounts of carbon dioxide and release oxygen through photosynthesis, which has also given ...
Practices from a Case Study in the Amazon Basin Joshua Fisher, Hannah Stutzman, Mariana Vedoveto, Debora Delgado, Ramon Rivero, Walter Quertehuari Dariquebe, Luis Seclén Contreras, Tamia ... Amazon Basin through building stakeholder capacity in conflict and natural resource man-agement. First, we present the project area and the social and ...
Living World - Amazon Case Study The Amazon is the largest tropical rainforest on Earth. It sits within the Amazon River basin, covers some 40% of the South American continent and as you can see on the map below includes parts of eight South American countries: Brazil, Bolivia, Peru, Ecuador, Colombia, Venezuela, Guyana, and Suriname.
Hydraulic redistribution (HR) refers to the process of soil water transport through the low-resistance pathway provided by plant roots. It has been observed in field studies and proposed to be one of the processes that enable plants to resist water limitations. However, most land-surface models (LSMs) currently do not include this underground root process. In this study, a HR scheme was ...
Introduction. The Amazon River Basin (ARB) encompasses the entire eastern and central parts of South America, covering over 6,100,000 km 2, which constitutes 44% of the South American continent.Covered mostly by tropical rainforest, it harbors the greatest biodiversity on Earth (constituting 10-15% of global land biodiversity), serves as a sink for greenhouse gasses by absorbing up to 5% of ...
From 1999 to 2003, the area annually logged in the Amazon basin was similar in magnitude to the area deforested 28. Logged areas are accessible by logging roads and are likely to be cleared within ...
Hi guys,😊 In this video I've explained the chapter - CASE STUDY - LIFE IN THE AMAZON RIVER BASIN line by line which will help you to learn the chapter and ...
The Amazon Basin is located in South America. Case Studies linked to Amazon Basin [] no linked case studies, yet Articles linked to Amazon Basin []
We restricted the study area to the Amazon Basin, using a refined boundary of the Amazon 6 based on terrestrial ecoregions of the world 51, and only considered fires in areas that were forested ...
A scaling approach to Budyko's framework and the complementary relationship of evapotranspiration in humid environments: case study of the Amazon River basin A. M. Carmona , G. Poveda , M. Sivapalan , S. M. Vallejo-Bernal , and E. Bustamante
Conflict history. By the early 1970s, approximately 43,000 agricultural colonists from the highlands had moved to the Ecuadorian Amazon Basin, also known as the Oriente, in a state-led effort to integrate the Oriente through settlement. Most migrants settled in Morona Santiago and Zamora Chinchipe in the southern region, where direct routes to ...
Assessing the impact of hydrodynamics on large-scale flood wave propagation - a case study for the Amazon Basin . Jannis M. Hoch, Arjen V. Haag, Arthur van Dam, Hessel C. Winsemius, Ludovicus P. H. van Beek, and Marc F. P. Bierkens. ... (FM) for the Amazon River basin at a grid-by-grid basis and at a daily time step. The use of a flexible ...
The proposed study follows the water budget method similar to a previous study (Swann & Koven, 2017), but focuses on a different topic, quantification of R in the Amazon basin. Utilizing the newer generation of GRACE solutions is expected to improve quantification of basin-scale TWS changes as well.
Three 'once in a century' droughts (Extended Data Fig. 1a-c) occurred in the Amazon basin over a single decade—in 2005, 2010 and 2015-2016 20,21 —provoking multiple forest responses ...
The city of Rio Branco, located in the southwest of Brazil's Amazon region, has been severely affected by a combination of urbanization in flood-prone areas and increasing floods in the Acre River. ... Inundation mapping using hydraulic modeling with high-resolution remote sensed data: a case study in the Acre River Basin, Brazil Antunes da ...