TB-Related Articles
Return to Main Menu
2020 DTBE Publications
Agizew T, Boyd R, Mathebula U, Mathoma A, Basotli J, Serumola C, Pals S, Finlay A, Lekone P, Rankgoane-Pono G, Tlhakanelo T, Chihota V, Auld AF. Outcomes of HIV-positive patients with non-tuberculous mycobacteria positive culture who received anti-tuberculous treatment in Botswana: Implications of using diagnostic algorithms without non-tuberculous mycobacteria. external icon PLoS One. 2020 Jun 12;15(6):e0234646. doi: 10.1371/journal.pone.0234646. eCollection 2020. PMID: 32530972.
Agizew T, Surie D, Oeltmann JE, Letebele M, Pals S, Mathebula U, Mathoma A, Kassa M, Hamda S, Pono P, Rankgoane-Pono G, Boyd R, Auld A, Finlay A. Tuberculosis preventive treatment opportunities at antiretroviral therapy initiation and follow-up visits. external icon Public Health Action. 2020 Jun 21;10(2):64-69. doi: 10.5588/pha.19.0056. PMID: 32639479.
Ahmed A, Feng PI, Gaensbauer JT, Reves RR, Khurana R, Salcedo K, Punnoose R, Katz DJ; Tuberculosis Epidemiologic Studies Consortium. Interferon-γ Release Assays in Children <15 Years of Age. external icon Pediatrics. 2020 Jan;145(1):e20191930. doi: 10.1542/peds.2019-1930. PMID: 31892518.
Armstrong LR, Kammerer JS, Haddad MB. Diabetes mellitus among adults with tuberculosis in the USA, 2010-2017. external icon BMJ Open Diabetes Res Care. 2020 Jul;8(1). pii: e001275. doi: 10.1136/bmjdrc-2020-001275. PMID: 32641300.
Auld AF, Agizew T, Mathoma A, Boyd R, Date A, Pals SL, Serumola C, Mathebula U, Alexander H, Ellerbrock TV, Rankgoane-Pono G, Pono P, Shepherd JC, Fielding K, Grant AD, Finlay A. Effect of tuberculosis screening and retention interventions on early antiretroviral therapy mortality in Botswana: a stepped-wedge cluster randomized trial. external icon BMC Med. 2020 Feb 11;18(1):19. doi: 10.1186/s12916-019-1489-0. PMID: 32041583.
Auld AF, Fielding K, Agizew T, Maida A, Mathoma A, Boyd R, Date A, Pals SL, Bicego G, Liu Y, Shiraishi RW, Ehrenkranz P, Serumola C, Mathebula U, Alexander H, Charalambous S, Emerson C, Rankgoane-Pono G, Pono P, Finlay A, Shepherd JC, Holmes C, Ellerbrock TV, Grant AD. Risk scores for predicting early antiretroviral therapy mortality in sub-Saharan Africa to inform who needs intensification of care: a derivation and external validation cohort study. external icon BMC Med. 2020 Nov 9;18(1):311. doi: 10.1186/s12916-020-01775-8. PMID: 33161899.
Beeler Asay GR, Lam CK, Stewart B, Mangan JM, Romo L, Marks SM, Baepubmrah Morris S, Gummo CL, Keh CE, Hill AN, Thomas A, Macaraig M, St John K, J Ampie T, Chuck C, Burzynski J. Cost of tuberculosis therapy directly observed on video for health departments and patients in New York City; San Francisco, California; and Rhode Island (2017–2018) external icon . Am J Public Health. 2020 Nov;110(11):1696-1703. doi: 10.2105/AJPH.2020.305877. Epub 2020 Sep 17. PMID: 32941064.
Bisson GP, Bastos M, Campbell JR, Bang D, Brust JC, Isaakadis P, Lange C, Menzies D, Migliori GB, Pape JW, Palmero D, Baghei P, Tabarsi P, Viiklepp P, Vilbrun S, Walsh J, Marks SM. Mortality in adults with multidrug-resistant tuberculosis and HIV by antiretroviral therapy and tuberculosis drug use: an individual patient data meta-analysis. external icon Lancet. 2020 Aug 8;396(10248):402-411. doi: 10.1016/S0140-6736(20)31316-7. PMID: 32771107.
Bonney W, Price SF; PMP1, Abhyankar S, Merrick R, Hampole V, Halse TA, DiDonato C, Dalton T, Metchock B, Starks AM, Miramontes R. Towards Unified Data Exchange Formats for Reporting Molecular Drug Susceptibility Testing. external icon Online J Public Health Inform. 2020 Dec 8;12(2):e14. doi: 10.5210/ojphi.v12i2.10644. eCollection 2020. PMID: 33381280.
Cegielski JP, Chan PC, Lan Z, Udwadia ZF, Viiklepp P, Yim JJ, Menzies D. Aminoglycosides and Capreomycin in the Treatment of Multidrug-resistant Tuberculosis: Individual Patient Data Meta-analysis of 12 030 Patients From 25 Countries, 2009-2016. external icon Clin Infect Dis. 2020 Oct 30:ciaa621. doi: 10.1093/cid/ciaa621. [Epub ahead of print]. PMID: 33124668.
Cegielski JP, Udwadia ZF, Viiklepp P, Yim JJ, Menzies D. Reply to van Deun and Decroo. external icon Clin Infect Dis. 2020 Dec 20:ciaa1875. doi: 10.1093/cid/ciaa1875. [Epub ahead of print]. PMID: 33341886.
Chen MP, Miramontes R, Kammerer JS. Multidrug-resistant tuberculosis in the United States, 2011-2016: patient characteristics and risk factors. external icon Int J Tuberc Lung Dis. 2020 Jan 1;24(1):92-99. doi: 10.5588/ijtld.19.0173. PMID: 32005311.
Chorba T, Jereb J. Confusion in the Genesis of Art and Disease: Charles Laval, Paul Gauguin, and Tuberculosis external icon . Emerg Infect Dis. 2020 Mar; 26(3):634–635. doi: 10.3201/eid2603.AC2603.
Click ES, Finlay A, Oeltmann JE, Basotli J, Modongo C, Boyd R, Wen XJ, Shepard J, Moonan PK, Zetola N. Phylogenetic diversity of Mycobacterium tuberculosis in two geographically distinct locations in Botswana – The Kopanyo Study. external icon Infect Genet Evol. 2020 Jul;81:104232. doi: 10.1016/j.meegid.2020.104232. Epub 2020 Feb 3. PMID: 32028055.
Click ES, Kurbatova E, Alexander H, Dalton TL, Chen MP, Posey JE, Ershova JJ, Cegielski P. Isoniazid- and Rifampin-Resistance Mutations Associated with Resistance to Second-line Drugs and with Sputum Culture Conversion. external icon J Infect Dis. 2020 Jun 11;221(12):2072-2082. doi: 10.1093/infdis/jiaa042. PMID: 32002554.
Cole B, Nilsen DM, Will L, Etkind SC, Burgos M, Chorba T. Essential Components of a Public Health Tuberculosis Prevention, Control, and Elimination Program: Recommendations of the Advisory Council for the Elimination of Tuberculosis and the National Tuberculosis Controllers Association . MMWR Recomm Rep. 2020 Jul 31; 69(7): 1–27. doi: 10.15585/mmwr.rr6907a1. PMID: 32730235.
Collins JM, Stout JE, Ayers T, Hill AN, Katz DJ, Ho CS, Blumberg HM, Winglee K. Tuberculosis Epidemiologic Studies Consortium. Prevalence of latent tuberculosis infection among non-U.S.-born persons by country of birth—United States, 2012–2017 external icon . Clin Infect Dis. 2020 Nov 2. pii: 5952274. doi: 10.1093/cid/ciaa1662. [Epub ahead of print]. PMID: 33137172.
Cronin AM, Railey S, Fortune D, Hope Wegener D, Davis JB. Notes from the Field: Effects of the COVID-19 Response on Tuberculosis Prevention and Control Efforts – United States, March-April 2020 . MMWR Morb Mortal Wkly Rep. 2020 Jul 24; 69(29):971–972. doi: 10.15585/mmwr.mm6929a4. PMID: 32701944.
DeGruy K, Klein K, Rey Z, Hall P, Kim A, Alexander H. Development of dried tube specimens for Xpert MTB/RIF proficiency testing. external icon Afr J Lab Med. 2020 Sep 29;9(1):1166. doi: 10.4102/ajlm.v9i1.1166. eCollection 2020. PMID: 33102169.
de Perio MA, Kobayashi M, Wortham JM. Occupational Respiratory Infections. external icon Clin Chest Med. 2020 Dec;41(4):739-751. doi: 10.1016/j.ccm.2020.08.003. PMID: 33153691.
Dorman SE, Nahid P, Kurbatova EV, Goldberg SV, Bozeman L, Burman WJ, Chang KC, Chen M, Cotton M, Dooley KE, Engle M, Feng PJ, Fletcher CV, Ha P, Heilig CM, Johnson JL, Lessem E, Metchock B, Miro JM, Nhung NV, Pettit AC, Phillips PPJ, Podany AT, Purfield AE, Robergeau K, Samaneka W, Scott NA, Sizemore E, Vernon A, Weiner M, Swindells S, Chaisson RE; AIDS Clinical Trials Group and the Tuberculosis Trials Consortium. High-dose rifapentine with or without moxifloxacin for shortening treatment of pulmonary tuberculosis: Study protocol for TBTC study 31/ACTG A5349 phase 3 clinical trial. external icon Contemp Clin Trials. 2020 Mar; 90:105938. doi: 10.1016/j.cct.2020.105938. Epub 2020 Jan 22. PMID: 31981713.
Ershova JV, Volchenkov GV, Somova TR, Kuznetsova TA, Kaunetis NV, Kaminski D, Demikhova OV, Chernousova LN, Vasilyeva IA, Kerr EM, Cegielski JP, Kurbatova EV. Impact of GeneXpert MTB/RIF® on treatment initiation and outcomes of RIF-resistant and RIF-susceptible TB patients in Vladimir TB dispensary, Russia. external icon BMC Infect Dis. 2020 Jul 25;20(1):543. doi: 10.1186/s12879-020-05243-9. PMID: 32711457.
Fernando R, McDowell AC, Bhavaraju R, Fraimow H, Wilson JW, Armitige L, Haley C, Goswami ND. A model for bringing TB expertise to HIV providers: Medical consultations to the CDC-funded Regional Tuberculosis Training and Medical Consultation Centers, 2013-2017. external icon PLoS One. 2020 Aug 31;15(8):e0236933. doi: 10.1371/journal.pone.0236933. eCollection 2020. PMID: 32866154.
Figueroa A, Vonnahme L, Burrell K, Vera-García C, Gulati RK. CureTB and continuity of care for globally mobile patients. external icon Int J Tuberc Lung Dis. 2020 Jul 1;24(7):694-699. doi: 10.5588/ijtld.19.0486. PMID: 32718402.
Gobaud AN, Haley CA, Wilson JW, Bhavaraju R, Lardizabal A, Seaworth BJ, Goswami ND. Multidrug-resistant tuberculosis care in the United States. external icon Int J Tuberc Lung Dis. 2020 Apr 1;24(4):409-413. doi: 10.5588/ijtld.19.0515. PMID: 32317065.
Haddad MB, Lash TL, Castro KG, Hill AN, Navin TR, Gandhi NR, Magee MJ. Tuberculosis Infection Among People With Diabetes: United States Population Differences by Race/Ethnicity. external icon Am J Prev Med. 2020 Jun;58(6):858-863. doi: 10.1016/j.amepre.2019.12.010. Epub 2020 Feb 13. PMID: 32061457.
Haley CA, Macias P, Jasuja S, Jones BA, Rowlinson MC, Jaimon R, Onderko P, Darnall E, Gomez ME, Peloquin C, Ashkin D, Goswami ND. Novel 6-Month Treatment for Drug-Resistant Tuberculosis, United States. external icon Emerg Infect Dis. 2021 Jan;27(1):332-334. doi: 10.3201/eid2701.203766. Epub 2020 Nov 23. PMID: 33227229.
Hill AN, Cohen T, Salomon JA, Menzies NA. High-resolution estimates of tuberculosis incidence among non-U.S.-born persons residing in the United States, 2000-2016. external icon Epidemics. 2020 Nov 10;33:100419. doi: 10.1016/j.epidem.2020.100419. [Epub ahead of print]. PMID: 33242759.
Iqbal SA, Isenhour CJ, Mazurek G, Truman BI. Diagnostic code agreement for electronic health records and claims data for tuberculosis. external icon Int J Tuberc Lung Dis. 2020 Jul 1;24(7):706-711. doi: 10.5588/ijtld.19.0792. PMID: 32718404.
Jo Y, Shrestha S, Gomes I, Marks S, Hill A, Asay G, Dowdy D. Model-Based Cost-Effectiveness of State-level Latent Tuberculosis Interventions in California, Florida, New York and Texas. external icon Clin Infect Dis. 2020 Jun 25. pii: ciaa857. doi: 10.1093/cid/ciaa857. [Epub ahead of print]. PMID: 32584968.
Kerr EM, Vonnahme LA, Goswami ND. Impact of Targeted Local Interventions on Tuberculosis Awareness and Screening Among Persons Experiencing Homelessness During a Large Tuberculosis Outbreak in Atlanta, Georgia, 2015-2016. external icon Public Health Rep. 2020. Jul/Aug;135(1_suppl):90S-99S. doi: 10.1177/0033354920932644. PMID: 32735200.
Klein K, DeGruy K, Rey Z, Hall P, Kim A, Gutreuter S, Alexander H. A global proficiency testing programme for Xpert® MTB/RIF using dried tube specimens, 2013-2015. external icon Afr J Lab Med. 2020 Nov 27;9(1):1167. doi: 10.4102/ajlm.v9i1.1167. eCollection 2020. PMID: 33354528.
Klopper M, Heupink TH, Hill-Cawthorne G, Streicher EM, Dippenaar A, de Vos M, Abdallah AM, Limberis J, Merker M, Burns S, Niemann S, Dheda K, Posey J, Pain A, Warren RM. A landscape of genomic alterations at the root of a near-untreatable tuberculosis epidemic. external icon BMC Med. 2020 Feb 4;18(1):24. doi: 10.1186/s12916-019-1487-2. PMID: 32014024.
Lienhardt C, Nunn A, Chaisson R, Vernon AA, Zignol M, Nahid P, Delaporte E, Kasaeva T. Advances in clinical trial design: Weaving tomorrow’s TB treatments. external icon PLoS Med. 2020 Feb 27;17(2):e1003059. doi: 10.1371/journal.pmed.1003059. eCollection 2020 Feb. PMID: 32106220.
Liu Y, Phares CR, Posey DL, Maloney SA, Cain KP, Weinberg MS, Schmit KM, Marano N, Cetron MS. Tuberculosis among Newly Arrived Immigrants and Refugees in the United States. external icon Ann Am Thorac Soc. 2020 Jul 30. doi: 10.1513/AnnalsATS.201908-623OC. [Epub ahead of print]. PMID: 32730094.
Marks SM, Dowdy DW, Menzies NA, Shete PB, Salomon JA, Parriott A, Shrestha S, Flood J, Hill AN. Policy Implications of Mathematical Modeling of Latent Tuberculosis Infection Testing and Treatment Strategies to Accelerate Tuberculosis Elimination. external icon Public Health Rep. 2020 Jul/Aug;135(1_suppl):38S-43S. doi: 10.1177/0033354920912710. PMID: 32735183.
Marks SM, Katz DJ, Davidow AL, Pagaoa MA, Teeter LD, Graviss EA. The Impact of HIV Infection on TB Disparities Among US-Born Black and White Tuberculosis Patients in the United States. external icon J Public Health Manag Pract. 2020 Sep/Oct;26(5):E5-E12. doi: 10.1097/PHH.0000000000000949. PMID: 32732731.
Mathebula U, Emerson C, Agizew T, Pals S, Boyd R, Mathoma A, Basotli J, Rankgoane-Pono G, Serumola C, Date A, Auld AF, Finlay A. Improving sputum collection processes to increase tuberculosis case finding among HIV-positive persons in Botswana. external icon Public Health Action. 2020 Mar 21;10(1):11-16. doi: 10.5588/pha.19.0051. PMID: 32368518.
McDowell A, Haas M, Seaworth B, Wilson JW, Patrawalla A, Haley C, Lauzardo M, de Bruyn M, Goswami ND. Linezolid use for the treatment of multidrug-resistant tuberculosis, TB centers of excellence, United States, 2013-2018. external icon J Clin Tuberc Other Mycobact Dis. 2020 Nov 16;22:100201. doi: 10.1016/j.jctube.2020.100201. eCollection 2021 Feb. PMID: 33336084.
Mehaffy C, Kruh-Garcia NA, Graham B, Jarlsberg LG, Willyerd CE, Borisov A, Sterling TR, Nahid P, Dobos KM. Identification of Mycobacterium tuberculosis peptides in serum extracellular vesicles from persons with latent tuberculosis infection. external icon J Clin Microbiol. 2020 May 26;58(6):e00393-20. doi: 10.1128/JCM.00393-20. PMID: 32245831.
Menzies NA, Bellerose M, Testa C, Swartwood N, Malyuta Y, Cohen T, Marks SM, Hill AN, Date AA, Maloney SA, Bowden SE, Grills AW, Salomon JA. Impact of Effective Global Tuberculosis Control on Health and Economic Outcomes in the United States. external icon Am J Respir Crit Care Med. 2020 Jul 9. doi: 10.1164/rccm.202003-0526OC. [Epub ahead of print]. PMID: 32645277.
Menzies NA, Swartwood N, Testa C, Malyuta Y, Hill AN, Marks SM, Cohen T, Salomon, JA. Time Since Infection and Risks of Future Disease for Individuals with Mycobacterium tuberculosis Infection in the United States external icon . Epidemiology. 2021 Jan; 32(1): 70–78. Epub 2020 Sep 29. doi: 10.1097/EDE.0000000000001271. PMID: 33009253.
Miele K, Bamrah Morris S, Tepper NK. Tuberculosis in Pregnancy. external icon Obstet Gynecol. 2020 Jun;135(6):1444-1453. doi: 10.1097/AOG.0000000000003890. PMID: 32459437.
Muloma E, Stewart R, Townsend H, Koch S, Burkholder S, Railey S, White K, Redington-Noble R, Caine V. Multipronged Approach to Controlling a Tuberculosis Outbreak Among Persons Experiencing Homelessness. external icon J Public Health Manag Pract. 2020 Sep 9. doi: 10.1097/PHH.0000000000001211. [Epub ahead of print]. PMID: 32956287.
Nabity SA, Mponda K, Gutreuter S, Surie D, Williams A, Sharma AJ, Schnaubelt ER, Marshall RE, Kirking HL, Zimba SB, Sunguti JL, Chisuwo L, Chiwaula MJ, Gregory JF, da Silva R, Odo J, Jahn A, Kalua T, Nyirenda R, Girma B, Buono N, Maida A, Kim EJ, Gunde LJ, Mekonnen TK, Auld AF, Muula AS, Oeltmann JE. Protocol for a Case-Control Study to Investigate the Association of Pellagra With Isoniazid Exposure During Tuberculosis Preventive Treatment Scale-Up in Malawi external icon . Front Public Health. 2020 Nov 26; 8:551308. doi: 10.3389/fpubh.2020.551308. eCollection 2020. PMID: 33324593.
Punetha A, Ngo HX, Holbrook SYL, Green KD, Willby MJ, Bonnett SA, Krieger K, Dennis EK, Posey JE, Parish T, Tsodikov OV, Garneau-Tsodikova S. Structure-Guided Optimization of Inhibitors of Acetyltransferase Eis from Mycobacterium tuberculosis. external icon ACS Chem Biol. 2020 Jun 19;15(6):1581-1594. doi: 10.1021/acschembio.0c00184. Epub 2020 May 18. PMID: 32421305.
Reichler MR, Hirsch C, Yuan Y, Khan A, Dorman SE, Schluger N, Sterling TR; Tuberculosis Epidemiologic Studies Consortium Task Order 2 Team. Predictive value of TNF-α, IFN-γ, and IL-10 for tuberculosis among recently exposed contacts in the United States and Canada. external icon BMC Infect Dis. 2020 Jul 31;20(1):553. doi: 10.1186/s12879-020-05185-2. PMID: 32736606.
Reichler MR, Khan A, Yuan Y, Chen B, McAuley J, Mangura B, Sterling TR; Tuberculosis Epidemiologic Studies Consortium Task Order 2 Team. Duration of Exposure Among Close Contacts of Patients With Infectious Tuberculosis and Risk of Latent Tuberculosis Infection. external icon Clin Infect Dis. 2020 Feb 11;ciz1044. doi: 10.1093/cid/ciz1044. [Epub ahead of print]. PMID: 32044987.
Rossetti A. Latent TB Infection: New Guidelines and Preferred Treatments. external icon Medscape 2020.
Schmit KM, Shah N, Kammerer S, Bamrah Morris S, Marks SM. Tuberculosis Transmission or Mortality Among Persons Living with HIV, USA, 2011-2016. external icon J Racial Ethn Health Disparities. 2020 Feb 14. doi: 10.1007/s40615-020-00709-7. [Epub ahead of print]. PMID: 32060748.
Schmit KM, Wortham JM, Ho CS, Powell KM. Analysis of severe adverse events reported among patients receiving isoniazid-rifapentine treatment for latent Mycobacterium tuberculosis infection – United States, 2012-2016. external icon Clin Infect Dis. 2020 Mar 18;ciaa286. doi: 10.1093/cid/ciaa286. [Epub ahead of print]. PMID: 32185390.
Schwartz NG, Price SF, Pratt RH, Langer AJ. Tuberculosis – United States, 2019. external icon MMWR Morb Mortal Wkly Rep. 2020 Mar 20;69(11):286-289. doi: 10.15585/mmwr.mm6911a3. PMID: 32191684.
Shrestha S, Parriott A, Menzies NA, Shete PB, Hill AN, Marks SM, Dowdy DW. Estimated Population-level Impact of Using a Six-Week Regimen of Daily Rifapentine to Treat Latent Tuberculosis Infection in the United States. external icon Ann Am Thorac Soc. 2020 Sep 11. doi: 10.1513/AnnalsATS.202005-574RL. [Epub ahead of print]. PMID: 32916062.
Sotgiu G, Battista Migliori G, Menzies D, Mase S, Chorba T, Seaworth B, Nahid P. Reply to: “ATS/CDC/ERS/IDSA Clinical Practice Guidelines for Treatment of Drug-resistant TB: A Two-edged Sword?”. external icon Am J Respir Crit Care Med Actions. 2020 May 6. doi: 10.1164/rccm.202003-0698LE. [Epub ahead of print]. PMID: 32374628.
Sterling TR, Njie G, Zenner D, Cohn DL, Reves R, Ahmed A, Menzies D, Horsburgh CR Jr, Crane CM, Burgos M, LoBue P, Winston CA, Belknap R. Guidelines for the Treatment of Latent Tuberculosis Infection: Recommendations from the National Tuberculosis Controllers Association and CDC, 2020. external icon MMWR Recomm Rep. 2020 Feb 14;69(1):1-11. doi: 10.15585/mmwr.rr6901a1. PMID: 32053584.
Stewart RJ, Wortham J, Parvez F, Morris SB, Kirking HL, Cameron LH, Cruz AT. Tuberculosis Infection in Children external icon . J Nurse Pract. 2020 Oct; 16(9):673-678. doi: 10.1016/j.nurpra.2020.06.027.
Talwar A, Stewart R, Althomsons SP, Rinsky J, Jackson DA, Galvis ME, Graham P, Huaman MA, Karrer J, Kondapally K, Mitchell S, Wortham J, de Fijter S. Notes from the Field: Multidrug-Resistant Tuberculosis Among Workers at Two Food Processing Facilities — Ohio, 2018–2019 external icon . MMWR Morb Mortal Wkly Rep. 2020 Aug 14; 69(32): 1104–1105. Published online 2020 Aug 14. doi: 10.15585/mmwr.mm6932a6. PMID: 32790660.
Tsang CA, Langer AJ, Kammerer JS, Navin TR. US Tuberculosis Rates among Persons Born Outside the United States Compared with Rates in Their Countries of Birth, 2012-2016 1 . external icon Emerg Infect Dis. 2020 Mar;26(3):533-540. doi: 10.3201/eid2603.190974.Emerg Infect Dis. 2020. PMID: 32091367.
Wang EY, Arrazola RA, Mathema B, Ahluwalia IB, Mase SR. The impact of smoking on tuberculosis treatment outcomes: a meta-analysis. external icon Int J Tuberc Lung Dis. 2020 Feb 1;24(2):170-175. doi: 10.5588/ijtld.19.0002. PMID: 32127100.
Warria K, Nyamthimba P, Chweya A, Agaya J, Achola M, Reichler M, Cowden J, Heilig CM, Borgdorff MW, Cain KP, Yuen C. Tuberculosis disease and infection among household contacts of bacteriologically confirmed and non-confirmed tuberculosis patients. external icon Trop Med Int Health. 2020 Jun;25(6):695-701. doi: 10.1111/tmi.13392. Epub 2020 Apr 6. PMID: 32170771.
Weiner M, Gelfond J, Johnson-Pais TL, Engle M, Johnson JL, Whitworth WC, Bliven-Sizemore E, Nsubuga P, Dorman SE, Savic R; Pharmacokinetics/Pharmacodynamics Group of Tuberculosis Trials Consortium. Decreased plasma rifapentine concentrations associated with AADAC single nucleotide polymorphism in adults with tuberculosis. external icon J Antimicrob Chemother. 2020 Dec 29:dkaa490. doi: 10.1093/jac/dkaa490. [Epub ahead of print]. PMID: 33374006.
Willby M, Chopra P, Lemmer D, Klein K, Dalton TL, Engelthaler DM, Cegielski P, Posey JE; Global PETTS Investigators. Molecular evaluation of fluoroquinolone resistance in serial Mycobacterium tuberculosis isolates from individuals diagnosed with multidrug-resistant tuberculosis external icon . Antimicrob Agents Chemother. 2020 Oct 26:AAC.01663-20. doi: 10.1128/AAC.01663-20. [Epub ahead of print]. PMID: 33106264.
Yelk Woodruff R, Hill A, Marks S, Navin T, Miramontes R. Estimated Latent Tuberculosis Infection Prevalence and Tuberculosis Reactivation Rates Among Non-U.S.-Born Residents in the United States, from the 2011-2012 National Health and Nutrition Examination Survey. external icon J Immigr Minor Health. 2020 Aug 5. doi: 10.1007/s10903-020-01065-8. [Epub ahead of print]. PMID: 32761297.
Last updated March 26, 2021
To receive email updates about this page, enter your email address:

Exit Notification / Disclaimer Policy
- The Centers for Disease Control and Prevention (CDC) cannot attest to the accuracy of a non-federal website.
- Linking to a non-federal website does not constitute an endorsement by CDC or any of its employees of the sponsors or the information and products presented on the website.
- You will be subject to the destination website's privacy policy when you follow the link.
- CDC is not responsible for Section 508 compliance (accessibility) on other federal or private website.
- - Google Chrome
Intended for healthcare professionals
- My email alerts
- BMA member login
- Username * Password * Forgot your log in details? Need to activate BMA Member Log In Log in via OpenAthens Log in via your institution

Search form
- Advanced search
- Search responses
- Search blogs
- Current and future...
Current and future treatments for tuberculosis
- Related content
- Peer review
- Anthony Lee , medical student 1 2 ,
- Yingda Linda Xie , assistant professor 2 3 ,
- Clifton E Barry , senior investigator 2 ,
- Ray Y Chen , associate research clinical 2
- 1 Medical Research Scholars Program, National Institutes of Health, Bethesda, MD, USA
- 2 Tuberculosis Research Section, Laboratory of Clinical Immunology and Microbiology, Division of Intramural Medicine, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD, USA
- 3 Division of Infectious Diseases, Department of Medicine, Rutgers New Jersey Medical School, Newark, NJ, USA
- Correspondence to RY Chen ray.chen{at}nih.gov
Guidelines on the treatment of tuberculosis (TB) have essentially remained the same for the past 35 years, but are now starting to change. Ongoing clinical trials will hopefully transform the landscape for treatment of drug sensitive TB, drug resistant TB, and latent TB infection. Multiple trials are evaluating novel agents, repurposed agents, adjunctive host directed therapies, and novel treatment strategies that will increase the probability of success of future clinical trials. Guidelines for HIV-TB co-infection treatment continue to be updated and drug resistance testing has been revolutionized in recent years with the shift from phenotypic to genotypic testing and the concomitant increased speed of results. These coming changes are long overdue and are sorely needed to address the vast disparities in global TB incidence rates. TB is currently the leading cause of death globally from a single infectious agent, but the work of many researchers and the contributions of many patients in clinical trials will reduce the substantial global morbidity and mortality of the disease.
Introduction
Tuberculosis (TB) is the leading cause of death globally from a single infectious agent, even surpassing HIV. 1 To achieve the World Health Organization’s End TB Strategy (a 90% decrease in TB incidence and 95% decrease in TB mortality by 2035 compared with 2015) requires shorter and more effective treatment regimens for drug sensitive (DS) and multidrug resistant (MDR) TB (disease resistant to the two first line drugs isoniazid and rifampin). Treatment for latent TB infection also needs to be shorter in duration and targeted to individuals at risk of progression to be practical and effective globally. The current poor global control of TB is due in part to the lack of research innovation over the past few decades; current DS-TB treatment guidelines have been essentially unchanged for 35 years and treatment still takes a minimum six months. Randomized controlled clinical trials for TB treatments are often challenging to complete because of the extended duration of treatment (six months for DS-TB and up to 24 months for MDR/extensively drug resistant (XDR)-TB) and follow-up times (often one year after treatment completion) required to confirm cure in standard of care control arms. The good news is that multiple clinical trials for DS-TB, MDR-TB, XDR-TB, and latent TB are ongoing, and early results have led to important changes in the MDR-TB treatment guidelines. Unlike the last 30+ years, the next five years should continue to bring about major changes in the treatment of TB. This review summarizes the current state of TB treatment with an emphasis on the ongoing clinical trials to anticipate how guidelines may change in the coming years.
Incidence/prevalence
According to WHO estimates, in 2018 10 million people developed TB globally, for an incidence of 132/100 000 people. This global average, however, hides the vast disparities between developed and developing countries. Almost all cases are concentrated in South East Asia (44%), Africa (24%), and the western Pacific (18%) regions. The eastern Mediterranean accounts for 8.1%, Americas 2.9%, and Europe 2.6% of cases. The highest income countries have incidence rates of <10/100 000 people, while the countries with the highest rates are >50-fold higher, exceeding 500/100 000 people. Although global rates of DS-TB are slowly decreasing, rates of MDR-TB are decreasing less quickly, affecting 3.4% of new TB cases and 18% of previously treated cases. Even more challenging to treat is XDR-TB: MDR-TB with additional resistance to fluoroquinolones and injectable aminoglycosides. An estimated 6.2% of MDR-TB cases were actually XDR-TB in 2018. 1
Sources and selection criteria
We searched PubMed for clinical trials of treatments for active and latent TB, including host directed therapies (HDT), through September 2019 and included only English language peer reviewed articles. We used terms including “active tuberculosis clinical trial,” “latent tuberculosis clinical trial,” “drug sensitive,” “drug resistant,” and “HDT.” We prioritized randomized controlled trials, but we also included some notable non-controlled interventional trials. Case reports, case series, and observational trials were generally excluded. We also searched the references used in current TB treatment guidelines and relevant systematic review articles, prioritizing the most recent articles. In addition, because ongoing trials do not yet have published results, we searched clinical trials registries, primarily www.clinicaltrials.gov . We conducted a general internet search to look for additional information on registered clinical trials or to identify clinical trials registered with other clinical trials registries.
Active TB treatment
History of tb treatment.
Treatment of tuberculosis has relied on multidrug chemotherapy to achieve three objectives:
To rapidly reduce mycobacterial burden to decrease disease morbidity, mortality, and transmission
To eradicate persistent mycobacterial populations to prevent relapse
To prevent acquisition of drug resistance.
Before the development of effective chemotherapy, TB treatment was essentially palliative care in sanatoriums with fresh air and sunlight. The age of TB chemotherapy began with the discovery of anti-tuberculosis compounds, beginning with streptomycin and para-aminosalicylic acid (PAS) in 1944.
Reviews of historical TB treatment trials date back to the 1940s, including the initial trial of PAS versus streptomycin to treat TB in 1944, which was one of the first randomized controlled chemotherapy trials ever conducted. 2 These landmark studies encompassed dozens of randomized controlled trials of a few hundred participants each across Africa, India, Hong Kong, and the UK, and investigated the most optimal drug doses and combinations. These trials resulted in an initial “triple therapy” combination of isoniazid, PAS, and streptomycin for 24 months in the 1950s that cured 90-95% of patients.
In the 1980s results from subsequent trials led to formation of the currently recommended regimen of isoniazid, rifampin, pyrazinamide, and ethambutol for six months. The duration of treatment was established after increased relapse rates were seen in trials of less than six months’ duration. 3 4 5 6 7 Incredibly, despite the advent of newer drugs, this first line regimen has not changed for more than 35 years.
The coming decade is likely to change this for DS-TB and MDR-TB, with many clinical trials expected to be completed in the next several years ( fig 1 ). However, the factors that allowed testing every combination of drug, dose, and duration viable in the 1940s through 1970s—limited numbers of drugs, abundance of patients, and relatively few concurrent trials competing to enroll—are no longer true today. To optimize the limited resources and increase the chance of clinical trials success, better methodologies are needed, possibly based on optimized biomarkers such as the NexGen EBA trial (described below), to determine the best combinations of drugs to take forward into advanced stage clinical trials.

Timeline of ongoing active TB clinical trials with start dates and projected end dates
- Download figure
- Open in new tab
- Download powerpoint
Trials of shortening treatment for drug sensitive TB
Recent efforts to shorten duration of chemotherapy for DS-TB have focused on incorporating a fluoroquinolone or using higher doses of a rifamycin. The fluoroquinolone based strategies faced a major setback with the failure of three large trials in 2014, 8 9 10 despite higher rates of sputum culture conversion at two months in the fluoroquinolone arms in two of these trials. 8 9 There is only one ongoing trial to our knowledge, conducted by the Beijing Chest Hospital, which is still attempting a fluoroquinolone strategy by continuing all four intensive phase drugs with or without levofloxacin for 4.5 months ( table 1 ). Data from trials conducted in the 1970s and 1980s by the British Medical Research Council suggested that the sterilizing activity of pyrazinamide was limited to the initial eight weeks of treatment, owing to the acidic environment required for pyrazinamide activation. 11 It remains to be seen whether the extension of intensive phase treatment and adding levofloxacin to 4.5 months will be sufficient for successful shortening of treatment.
Ongoing trials of shortening treatment for drug sensitive, multi-drug resistant, and extensively drug resistant TB
- View inline
Two other ongoing trials attempt to shorten treatment by increasing the dose of the rifamycin, rifampin, or rifapentine. There is substantial evidence to support that rifampin dosed at the recommended 10 mg/kg is well below its therapeutic threshold and that much higher doses, even ≥35 mg/kg, may be safe and effective. 12 13 These studies suggest that high dose rifampin may shorten treatment based on faster rates of sputum culture conversion achieved. 14 15 16
The caveat is that earlier sputum culture conversion may not result in earlier cure, as shown by the failure of the fluoroquinolone treatment shortening trials. The RIFASHORT trial takes the surrogate results to a phase III trial, testing a standard of care (SOC) arm with rifampin 600 mg (10 mg/kg) daily against two experimental four month treatment arms using rifampin 1200 mg or 1800 mg. The TBTC Study 31 trial tests a SOC arm against two experimental rifapentine 1200 mg daily arms (instead of rifampin), with the second arm also adding moxifloxacin 400 mg daily. Results are expected in 2020 and, if successful, would be the first successful four month DS-TB trials to date.
In contrast to these trials which evaluate new or modified drug regimens, two trials are investigating treatment shortening strategies—PredictTB and TRUNCATE-TB. The presentation of TB is quite variable, so some individuals will likely need longer therapy than others no matter what regimen is used. Prospectively identifying this higher risk group can help target treatment duration appropriately.
PredictTB is based on a previously conducted trial that shortened therapy to four months among 394 pulmonary TB patients with less severe disease at baseline, defined as no cavity on baseline chest radiograph, and with sufficient early treatment response, defined as conversion of month 2 sputum culture to negative. 17 Although this trial failed to achieve its predefined 5% non-inferiority margin, the researchers’ algorithm for patient stratification increased the treatment success rate in the four month arm from around 80% seen in other non-stratified four month treatment trials to 93% in their trial. The PredictTB trial revises the baseline disease stratification from chest radiograph to FDG-positron emission tomography/computed tomography (PET/CT) scan. Appropriate disease response is also changed from month 2 sputum culture conversion to reduction in PET/CT disease burden at month 1 and sputum GeneXpert cycle threshold increase at month 4. 18 If the trial is successful, this strategy of shortening treatment only among those less severely diseased could also be evaluated among MDR-TB cases. In addition, investigation will focus on blood, sputum, urine, and other imaging biomarkers that correlate with PET/CT results and are more amenable to being scaled up globally.
In contrast to PredictTB, which only uses the four first line TB drugs, TRUNCATE-TB is a multi-arm, multi-stage trial that incorporates high dose rifamycins and various combinations of linezolid, clofazimine, levofloxacin, or bedaquiline into four two month treatment regimens. Patients who relapse are retreated with standard four drug therapy for six months with the hypothesis that this overall strategy will be non-inferior to standard six month treatment outcomes at two years. 19 Both PredictTB and TRUNCATE-TB build on previously gathered data that show most patients are successfully treated before six months. 3 The PredictTB strategy identifies these patients prospectively, then shortens treatment only for this cohort. The TRUNCATE-TB strategy shortens treatment for everyone to two months using a more intense intensive phase regimen, assuming that those who relapse will not develop resistance and can still be cured with subsequent standard six month therapy. In the end, a combination of these two strategies may be the most successful in maximizing cure rates with minimal treatment burden.
Drug resistant TB treatment shortening trials
An estimated 8% of TB patients globally have isoniazid resistant but rifampin susceptible TB. In 2018, WHO updated its treatment guidelines for isoniazid resistant TB to recommend levofloxacin, rifampin, pyrazinamide, and ethambutol for six months. 20 More worrisome, however, is rifampin resistance. WHO estimates that only 55% of the estimated 558 000 incident cases of rifampin resistant (RR-TB) and MDR-TB in 2017 successfully completed treatment. 1
The landscape of RR-TB and MDR-TB treatment was, for many years, defined by prolonged treatment (20-24 months) with older drugs that had substantial side effects and required daily injections for the initial eight months. This began to change with the publication of the “Bangladesh regimen” in 2010, which touted a successful nine month treatment regimen with daily injections only for the initial four months. 21 However, because the trial was an uncontrolled observational study of sequential treatment arms, questions were raised about the validity of these results.
A slightly modified Bangladesh regimen was therefore tested within a rigorous randomized controlled trial (Standard Treatment Regimen of Anti-Tuberculosis Drugs for Patients with MDR-TB [STREAM]) comparing the nine month experimental arm with a 20 month SOC control arm that followed the 2011 WHO guidelines. This trial enrolled 424 participants and showed that the shortened nine month arm was non-inferior (using a 10% margin) to the standard 20 month arm. 22 WHO had recommended the shortened nine month treatment regimen for uncomplicated MDR-TB (no additional resistance to fluoroquinolones or injectable agents) in 2016 and affirmed this recommendation with the results of the STREAM trial. For patients ineligible for the nine month regimen, the recommended treatment duration remains 18-20 months.
WHO 2019 guidelines now recommend an all oral, bedaquiline-containing regimen to replace injectable agents as the preferred treatment regimen. 23 24 WHO’s move toward shorter and all oral regimens for MDR-TB is also reflected in the ongoing MDR-TB clinical trials. Almost all experimental regimens being tested (except for Opti-Q, which began in 2015) are six to nine months long, and most of the experimental arms are completely oral (except for Opti-Q and STREAM stage 2 six month arm intensive phase; table 1 ). The change in the recommended WHO SOC regimen in duration and composition has implications for ongoing trials using the now obsolete 20-24 month regimens for their control arm (Opti-Q, MDR-END, NEXT, endTB, and TB-PRACTECAL). If enrollment is ongoing, these trials face the decision of whether to update their control arm treatment regimen or duration mid-trial.
Even more problematic are the trials that have experimental regimens that are now obsolete, either because of their duration or inclusion of injectable agents. The Opti-Q trial is one of these but, notably, its primary endpoint is not final treatment outcome but the surrogate endpoints of time to culture conversion by month 6 and safety of higher levofloxacin dosing. The only other trial that still uses an injectable agent is the STREAM stage 2 trial six month arm. If the other trials with all oral six month arms are successful, particularly in the XDR-TB trials, then injectable agents will phase out of first line therapy regardless of the treatment duration and level of resistance (eg, MDR-TB plus fluoroquinolone resistance).
Among the MDR/XDR-TB trials, there are two other major differences between the trials: 1) whether the trial incorporates a concurrent control arm compared with a historical control; and 2) the treatment regimen and duration (six versus nine months). The MDR (non-XDR) TB trials all have a WHO standard control arm. Of the three XDR-TB trials, however, only endTB-Q has a concurrent WHO standard control arm. Both Nix-TB and ZeNix only include experimental arms with no concurrent SOC control arm, with the primary outcome being relapse or failure at 12 months after enrollment. The benefits of not including a control arm are a smaller sample size and a much shorter trial, since the WHO SOC control was 20-24 months at the time these trials began. However, these studies also incur considerable risk, because if the treatment success rate in the experimental arm is below 95%, the benefit of the experimental arm may not be clear. In the three contemporaneous fluoroquinolone DS-TB treatment shortening trials 8 9 10 for example, the per protocol favorable outcomes of the control arms, despite using the same regimen, varied from 88.7% (OFLOTUB trial) to 92.0% (REMoxTB trial) to 95.1% (RIFAQUIN trial; table 2 ). If the experimental arm favorable outcome was 85%, whether this was a success or failure may differ depending on which control arm was used. Applying control arm results of a separately conducted study can interject misleading comparisons owing to varying populations and methodologies, and thus comparing with a historical control increases the risk of unrecognized biases and confounding factors in the data. 25
Study control arm favorable outcome
On 14 August 2019, the US Food and Drug Administration approved pretomanid, its second novel anti-tuberculous drug in 40 years (bedaquiline was approved in 2012). While additional agents against drug resistant TB are critically needed, the basis on which pretomanid was approved is somewhat controversial. 26 27
Pretomanid was approved only in combination with bedaquiline and linezolid to treat XDR-TB and non-responsive MDR-TB because this is how it was used in the Nix-TB trial. In that study, 95/107 (89%) patients had successful outcomes at one year and six months after the end of treatment. 28 As previously discussed, the Nix-TB trial was a small single arm trial that relied on a historical control for comparison, which is problematic given that bedaquiline and linezolid were not included in many historical controls. In addition, all patients received the same three drugs, two of which are already known to be very active against drug resistant TB. For example, when single drug linezolid was added to a failing treatment regimen in XDR-TB patients, 34/39 (87%) converted their sputum culture to negative by six months of treatment. 29 Thus, the individual potency of pretomanid against TB is unknown and more studies are needed to determine this.
Unfortunately, it is unlikely that any of the currently ongoing trials using pretomanid (SimpliciTB, TB-PRACTECAL, Nix-TB, ZeNix) will reveal pretomanid’s potency because of the lack of an appropriate comparison arm for this purpose. TB-PRACTECAL, for example, has a WHO SOC control arm but all three experimental arms contain the Nix-TB regimen just with or without moxifloxacin or clofazimine ( table 1 ), leaving the same conundrum where the individual effect of pretomanid cannot be differentiated from that of bedaquiline and linezolid. Pretomanid is a bicyclic 4-nitroimidazole, which is the same class as delamanid, another novel anti-TB agent that is still trying to find its proper place in the landscape of treating drug resistant TB. 30
Adjunctive and host directed therapies (HDT)
Augmentation of the host immune response is an additional strategy of growing interest to improve treatment efficacy. Current HDT candidates are largely repurposed drugs with pre-clinical evidence for counteracting Mycobacterium tuberculosis pathogenesis and survival, augmenting protective immune responses, and/or modulating detrimental inflammatory responses that exacerbate disease. These therapies encompass agents used to treat metabolic and psychiatric conditions, parasitic infections, cancer, and inflammatory conditions, and span various stages of pre-clinical and early clinical development.
Treatment endpoints of culture conversion and durable cure remain relevant to clinical trials of most HDTs. However, further considerations include how and when to utilize HDTs to complement antibacterial treatment, and how to incorporate clinical biomarkers to investigate or validate therapeutic mechanisms. For example, the IMPACT-TB study ( NCT03891901 ) evaluating safety and pharmacokinetics of imatinib at varying doses with and without isoniazid and rifabutin in healthy adults, will measure myelopoietic effects, proposed as a key therapeutic mechanism of imatinib ( table 3 ).
Host directed therapy (HDT) TB treatment trials
Stage 2 of the StAT-TB trial will evaluate pravastatin as an adjunct to HRZE against time to sputum culture conversion as well as the secondary endpoint of improvement of pulmonary function among individuals with drug sensitive pulmonary TB ( NCT03456102 ). Relevant study populations and biomarkers will also depend on the targeted intervention. HDTs targeted to modulate detrimental inflammatory responses, such as dexamethasone ( NCT03092817 ), may include or target individuals with TB meningitis where uncontrolled inflammation is closely associated with morbidity and mortality.
Finally, HDTs that act directly or indirectly via host responses to disrupt M tuberculosis pathogenesis may have the greatest impact in treating patients with high burden disease or DR-TB, where cure rates are lower and disease chronicity and treatment duration are longer.
Several HDT clinical trials, including a completed phase II study of adjunctive ibuprofen ( NCT02781909 ; no results posted) and an ongoing phase II/III study of recombinant human IL-2 ( NCT03069534 ) 31 have targeted sputum culture conversion in MDR/XDR-TB. Vitamin D has been evaluated as an adjunct to standard therapy in multiple trials with conflicting results, suggesting that any effect relative to anti-tuberculous chemotherapy is modest at best. This topic has been reviewed, including the ongoing randomized trials. 32 If results support robust efficacy endpoint data, we may anticipate future trials evaluating HDTs as a complementary treatment shortening strategy.
Capturing heterogeneity of drug responses in early phase clinical evaluation
When viewing the TB trials together ( fig 2 ), it appears that almost all possible combinations of the newer drugs are tested for either six or nine months without a clear rationale for how one combination or duration was selected compared with another. With the number of new compounds and HDTs in development 33 and the more limited number of patients available to enroll in trials, testing every possible combination as done previously is impractical. Improved methodologies are needed to allow rational selection of the optimal combinations and durations to take forward into clinical trials.

Ongoing active TB treatment shortening clinical trials with duration and drug composition of the experimental regimens tested
Current methods rely heavily on early bactericidal activity studies, 34 which measure the decline in serial sputum colony forming units (CFU) over the initial 14 days of treatment as a measure of sterilizing potency. This approach is endorsed by the Global Alliance for TB Drug Development 35 and the US FDA 36 but EBA does not appear to reflect durable cure. 37 Certain drugs, such as rifampin, pyrazinamide, and linezolid, have relatively low EBA but have been shown repeatedly to be instrumental in killing TB.
Other drugs, like ethambutol and the fluoroquinolones, have high EBA but are not assumed to be very potent clinically (ethambutol at currently used doses) or have not been successful in contributing to treatment shortening (fluoroquinolones). One possible explanation for this is that sputum based methodologies, like EBA, only sample changes in bacillary populations on airway or cavity surfaces rather than deep necrotic lesion compartments which harbor recalcitrant TB populations that are killed more slowly.
A drug’s ability to shorten treatment may be more related to how well it penetrates into necrotic, caseous lesions than airway surfaces. 38 39 For example, both pyrazinamide and rifampin diffuse into caseous lesions well whereas moxifloxacin does not. 40 which may explain why pyrazinamide and rifampin have both contributed to DS-TB regimen treatment shortening but moxifloxacin has not. Combining drugs with different lesion pharmacokinetic profiles is therefore an important consideration in rational regimen design. Furthermore, relying only on sputum measurements to reflect the myriad changes occurring throughout the lungs on treatment is likely to be imprecise.
We are conducting a trial called Radiologic and Immunologic Biomarkers of Sterilizing Drug Activity in Tuberculosis (NexGen EBA; NCT02371681 ) that attempts to augment a traditional EBA study with PET/CT imaging biomarker data to additionally capture sterilizing activity of first line drugs and their combinations which have previously been clinically and pharmacokinetically characterized. While sputum based measurements average all changes across the lungs into one result, a PET/CT image of the lungs can quantitate differential changes in different lesions. We hypothesize that, owing to different lesion pharmacokinetics, different treatment effects—including early bactericidal activity important for initial culture conversion and sterilizing activity important for durable cure—can be captured in different lesion types. This trial has been completed and data analyses are ongoing, with results expected in 2020. With this knowledge, rational combinations of drugs and HDTs can be selected and taken forward to later phase clinical trials with better chances of success.
Preventive TB treatment
Updates to treatment of latent tb infection (ltbi).
Unlike DS-TB treatment guidelines, LTBI treatment guidelines have changed in the last decade. The biggest change was the introduction of isoniazid/rifapentine once weekly for 12 weeks as a successful LTBI treatment regimen. 41 42 43 In addition, rifampin dosed daily for four months was confirmed to be as effective as isoniazid for nine months but with better treatment completion rates and fewer side effects. 44
Additional latent TB clinical trials are ongoing and span the scope of DS and MDR latent TB treatment, as well as HIV negative and HIV positive populations ( table 4 , figs 3and 4 ). Three DS latent TB trials are ongoing (2R2, ASTERoiD, and SCRIPT-TB), all of which test shorter, regimens of one to two months of higher dose rifamycins compared with the SOC regimen and vary in target LTBI populations and endpoints of completion/safety versus efficacy. Even if rifapentine based trials are successful in shortening LTBI treatment duration, uptake of rifapentine based regimens is currently limited by cost and availability, 45 with only one manufacturer making this drug globally. This situation will hopefully change as generic manufacturers enter this market. 46 Three ongoing trials are testing LTBI treatment regimens with MDR-TB exposure ( table 4 , fig 3, fig 4 ). Two trials, V-QUIN MDR and TB-CHAMP, both test levofloxacin for six months compared with placebo. The third trial, PHOENIx MDR-TB, compares six months of isoniazid against six months of delamanid.
Ongoing latent TB treatment shortening trials

Ongoing latent TB treatment shortening clinical trials with duration and drug composition of the experimental regimens tested

Timeline of ongoing latent TB clinical trials with start dates and projected end dates
Targeted treatment of incipient TB infection
While TB treatment and control efforts are currently dichotomized around “latent” and “active” TB infection, neither of the two available methods to identify latent TB infection (tuberculin skin test and interferon gamma release assays) distinguish the few asymptomatic individuals who will develop active TB (estimated to be 5-10% lifetime risk) from the majority of individuals who will never develop active TB. Treating all M tuberculosis latently infected individuals for months is not feasible in most areas of the world. Rather, evidence is emerging of a spectrum of TB disease activity, and new approaches attempt to predict asymptomatic individuals who will progress to active TB disease (incipient TB infection).
Many retrospective studies of biomarker signatures, particularly transcriptomic, have been published identifying incipient or active TB in different cohorts 47 but these signatures generally do not work well across populations and more work is needed to identify consistent signatures across cohorts. In addition, retrospective correlations of biomarker signatures ultimately need to be confirmed prospectively.
The Correlate of Risk Targeted Intervention Study (CORTIS; NCT02735590 ) is currently evaluating a host transcriptional signature previously found to have 66% prognostic sensitivity and 81% specificity for incident TB disease within 12 months 48 to see how well treatment with three months of weekly isoniazid/rifapentine reduces the incidence of TB in individuals who test positive. 49 The ability to both shorten and target preventive TB therapy to the few asymptomatic individuals at greatest risk of TB progression will greatly increase the global feasibility of treating LTBI.
Treatment of HIV/TB co-infection
Many of the ongoing TB clinical trials include individuals who are HIV positive ( table 1 , table 4 ). The recent WHO recommendation of nine months of treatment for MDR-TB also applies to individuals infected with HIV. 23 The treatment of active TB in HIV co-infected patients is generally the same as in patients uninfected with HIV, but with additional attention to overlapping toxicities and drug-drug interactions between commonly used HIV and TB drugs, particularly rifampin. Rifampin is a potent inducer of cytochrome P4503A4, uridine diphosphate glucuronosyltransferase 1A1, and P-glycoprotein enzymes, and thus will typically reduce the levels of drugs metabolized by these enzymes. Some drugs should not be used in conjunction with rifampin, while others can be used with dose adjustments. Rifabutin is less of an enzyme inducer than rifampin and can substitute for rifampin to allow certain antiretroviral drugs to be used. Drug-drug interactions are complex, and current guidelines continuously update tables on recommended combinations and dosing. 50 51 The recommended first line treatment is to use a rifampin based TB regimen with an efavirenz based antiretroviral therapy regimen. Options include using raltegravir instead of efavirenz or a ritonavir boosted protease inhibitor in conjunction with rifabutin. More studies are needed to define better drug-drug interactions for the newer anti-TB drugs, such as pretomanid.
The other major consideration in HIV-TB co-infection is the timing of treatment. When HIV is diagnosed first, earlier initiation of antiretroviral therapy and isoniazid preventive therapy independently reduce the development of active TB and death. 52 53 54 When TB is diagnosed first, there is a clear mortality benefit to starting antiretroviral therapy shortly after anti-TB therapy, despite the increased risk of developing immune reconstitution inflammatory syndrome. 55 56 57 According to current guidelines, 58 ART should begin within two weeks for those with CD4 cell counts <50 cells/mm 3 . For patients with higher CD4 cell counts, antiretroviral therapy should begin no later than eight weeks after starting anti-TB therapy because of known benefits of earlier initiation of antiretroviral therapy. 54 59 60
The treatment of LTBI in individuals infected with HIV is also like that of uninfected individuals, with attention paid to rifamycin drug-drug interactions and the possibility of an additional, even shorter one month regimen. Rifapentine is a key component of shortened LTBI treatment regimens, with rifapentine/INH once weekly×12 weeks currently recommended for HIV infected individuals if drug-drug interactions are manageable. The recent BRIEF TB/A5279 trial showed that in HIV infected individuals, weight based rifapentine and isoniazid 300 mg daily for one month was non-inferior in preventing TB compared with the standard isoniazid daily for nine months. 61 A similar trial, HIV-NAT 225, started in Thailand in 2019 ( table 4 ). Rifapentine/INH once daily for one month may become a recommended regimen in guidelines, but few antiretroviral drugs are recommended to be co-administered with rifapentine. 50 Protease inhibitors and the CCR5 inhibitor maraviroc are contraindicated. Among the non-nucleoside reverse transcriptase inhibitors, only efavirenz is approved for co-administration, although a phase I drug interaction study with doravirine is ongoing ( NCT03886701 ). Another trial is testing the steady state pharmacokinetic effects of once-weekly rifapentine with tenofovir alafenamide fumarate, a preferred backbone nucleoside reverse transcriptase inhibitor ( NCT03510468 ). Among the integrase strand transfer inhibitors, only raltegravir is currently recommended to be dosed with rifapentine only at the once weekly dose. Another trial (DOLPHIN) tested the safety and pharmacokinetics of dolutegravir with rifapentine/isoniazid once weekly in HIV infected adults and presented preliminary results showing this combination was well tolerated and that dolutegravir 50 mg daily maintained HIV viral suppression. 62 Additional trials testing similar combinations with rifampin are also ongoing.
Genotypic and phenotypic drug resistance testing
Substantial resources in TB drug development, including most current active and latent TB clinical trials, are directed against drug resistant tuberculosis ( table 1 , table 4 ). 33 The diagnosis of drug resistance traditionally has been done phenotypically, based on laboratory culture of M tuberculosis in the presence of varying concentrations of antibiotics. This technique requires substantial laboratory infrastructure and, most problematically, returns weeks to months after an empiric treatment decision has already been made. Genotypic resistance testing, which is considerably faster, reproducible, and operable within more automated, less biohazardous formats, are more compatible with guiding prompt treatment decisions outside of reference laboratories.
Commercial kits have been available since the mid-2000s but still necessitated considerable laboratory infrastructure until the rollout of the Cepheid GeneXpert MTB/RIF (Xpert; Sunnyvale, CA, USA) in 2010. 63 Xpert was the first fully integrated, automated genotypic test and provided a direct readout of TB and rifampin resistance within two hours. Recent advances to the GeneXpert platform include the Xpert MTB/RIF Ultra that is more sensitive 64 and the Xpert XDR that detects genotypic resistance to isoniazid, fluoroquinolones, and aminoglycosides. 65 However, detection of point mutations in specific target regions does not capture all phenotypic resistance, will continue to lag identification of new genetic resistance mechanisms, and may become irrelevant with the changing landscape of preferred second line drugs.
To that end, whole genome sequencing (WGS) provides more comprehensive information and has high sensitivity and specificity for isoniazid, rifampin, pyrazinamide, and ethambutol compared with phenotypic resistance testing. 66 WGS can also provide useful information to characterize mixed infections, differentiate relapsed infections from re-infections, and contribute to transmission tracing. Governmental programs in England, the Netherlands, and New York are shifting resources from phenotypic testing to WGS to save time and money. 67
Recent advances in highly portable, miniature sequencing platforms may facilitate access to TB WGS in resource limited settings. 68 Although WGS is typically done from a cultured sample of M tuberculosis , which adds two to four weeks to the result time, efforts are under way to perform analyses directly on sputum. Sputum, however, is complicated by human and other bacterial DNA. 69 The interpretation of genotypic results is also not always straightforward and large standardized databases with user friendly software are needed. 68 70 Finally, although overall sequencing costs continue to decline, startup capital costs for the machines are still prohibitive. 71 The notably faster time to results of genotypic over phenotypic testing make it far more clinically relevant and the test of choice for the future. As more advances are made and costs decline, WGS will likely become more and more accessible globally.
The international TB community jointly released DS-TB treatment guidelines in 2016. 72 This effort was sponsored by the American Thoracic Society, the Centers for Disease Control and Prevention (CDC), and the Infectious Diseases Society of America and was endorsed by the European Respiratory Society and the US National Tuberculosis Controllers Association. The American Academy of Pediatrics, the Canadian Thoracic Society, the International Union Against Tuberculosis and Lung Disease, and WHO also participated. These guidelines note that treatment by directly observed therapy (DOT) by trained personnel compared with self-administered therapy (SAT) is the standard of care in most TB programs in the US and Europe. WHO released another version in 2017 with some updates primarily focused on patient care aspects. 73 The WHO guidelines allow for SAT but conditionally recommends either DOT in the community or home by trained providers or video observed therapy over SAT. For drug resistant TB, the primary guidelines are from WHO, which released its latest version in 2019. 23 24 Recent reviews have been published for the management of drug resistant TB. 74 75
For the treatment of LTBI, WHO and the CDC have released updated treatment guidelines in 2020. 76 77 Both guidelines recommend targeted testing and treatment of LTBI with slight differences in the recommended treatment regimens. The regimens recommended by both guidelines are: 1) isoniazid with rifapentine once weekly for three months, and 2) isoniazid with rifampin daily for three months. In addition, WHO also recommends isoniazid daily for six or nine months as a primary regimen, whereas the CDC now lists these as alternative rather than preferred regimens. In contrast, the CDC recommends rifampin daily for four months as a preferred regimen, whereas WHO lists this as an alternative. The WHO guidelines also include two other regimens that the CDC guidelines do not: 1) isoniazid with rifapentine daily for one month, as an alternative regimen; and 2) for HIV infected individuals in high TB transmission settings, isoniazid daily for 36 months.
Emerging treatments
Owing to the number of ongoing TB clinical trials, the next several years will hopefully be a time of marked changes in treatment guidelines to include newer drugs, shorter regimens, and perhaps even HDTs. Future trials being planned for DS-TB will not only include treatment shortening but will also include newer drug such as bedaquiline. One recent result for MDR-TB treatment is the ACTG5343 (DELIBERATE) trial that tested the safety of combining bedaquiline and delamanid to treat MDR-TB, focusing on cardiac QTc prolongation since both drugs are known to have this side effect. Early results showed patients had a mean QTcF prolongation from baseline of 11.9 ms in the bedaquiline arm, 8.6 ms in the delamanid arm, and 20.7 ms in the combined arm. Thus, the combination of bedaquiline and delamanid appears to be safe from a cardiac standpoint, with only a modest additive effect on QTcF prolongation, in patients with normal QTcF interval at baseline. 78
Conclusions
With the recent advances in TB treatment and diagnostics, and many ongoing trials, the next five years will likely see major changes to TB treatment approaches. However, the future trend is clear and is already arriving for drug resistant TB—shorter, all oral regimens incorporating newer drugs. The question is which drugs, how short, and what patients. Will treatment of TB continue to be a one size fits all with the same regimen and duration recommended for all patients? Will patients be stratified into risk categories with treatment regimens and durations tailored for each? How will all the novel TB compounds currently in clinical development be incorporated effectively and efficiently to create optimal treatment combinations? What role will HDTs play in shortening treatment and reducing functional impairment? Is a one month or even a two week treatment regimen for TB realistic in the next 10 years? The results of ongoing and future trials will help answer these questions. However, even with these biomedical advances toward shorter, oral regimens, we continue to face fundamental challenges along the path to comprehensive, accessible, effective, and reliable delivery of treatment. Concerted efforts of community advocates, researchers, drug companies, TB programs, healthcare delivery systems, and innovative technologies around patient centered strategies will be critical to implement these advances and end the TB epidemic. 79 80 81
Research questions
What biomarkers will predict the development of active TB among LTBI patients to enable targeted treatment that is globally scalable?
What biomarkers will predict treatment outcomes early during treatment of active TB that will allow for more personalized treatment algorithms rather than the current one-size-fits-all approach?
What biomarkers will predict the development of relapsed TB after completion of treatment for active TB?
How should novel and existing drugs be combined to determine the best regimens to bring forward into phase III clinical trials testing of shortened treatment regimens for drug sensitive and drug resistant TB?
How patients and the public were involved in the creation of this article
This article was reviewed by two people who had been successfully treated for TB, one from the US and one from South Africa. The patient from the US received treatment by DOT, which “felt both like a hassle (because I had two young children and was busy caring for them at home) and like a good safeguard against forgetfulness (as one prone to getting caught up in caring for the kids).” She was interested in research on how current treatments could be implemented more successfully The patient from South Africa stated, “I don’t have any comments and any questions or comments I thought of while reading were inevitably answered further down.”
Series explanation: State of the Art Reviews are commissioned on the basis of their relevance to academics and specialists in the US and internationally. For this reason they are written predominantly by US authors
Acknowledgment: This research was supported by the Intramural Research Program of the US National Institutes of Health, National Institute of Allergy and Infectious Diseases.
Contributorship: YLX, CEB, and RYC developed the idea for this review. AL, YLX, and RYC performed the literature search. AL, YLX, CEB, and RYC wrote the review. RYC is the guarantor. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Competing interests: The BMJ has judged that there are no disqualifying financial ties to commercial companies. The author declares the following other interests: none.
Further details of The BMJ policy on financial interests are here: https://www.bmj.com/about-bmj/resources-authors/forms-policies-and-checklists/declaration-competing-interests
Provenance and peer review: commissioned; externally peer reviewed.
- ↵ World Health Organization. Global TB Report 2019. 2019. https://www.who.int/tb/publications/global_report/en/
- Ellard GA ,
- Mitchison DA
- Murray JF ,
- Schraufnagel DE ,
- Hopewell PC
- Gillespie SH ,
- McHugh TD ,
- REMoxTB Consortium
- Jindani A ,
- Harrison TS ,
- RIFAQUIN Trial Team
- Fielding K ,
- OFLOTUB/Gatifloxacin for Tuberculosis Project
- Mitchison D
- Abulfathi AA ,
- Decloedt EH ,
- Svensson EM ,
- Diacon AH ,
- Dorman SE ,
- Goldberg S ,
- Tuberculosis Trials Consortium
- Velásquez GE ,
- Brooks MB ,
- Boeree MJ ,
- Heinrich N ,
- Aarnoutse R ,
- PanACEA consortium
- Johnson JL ,
- Predict TB Study Group
- Papineni P ,
- Phillips P ,
- ↵ World Health Organization. WHO treatment guidelines for isoniazid-resistant tuberculosis: Supplement to the WHO treatment guidelines for drug-resistant tuberculosis. 2018. https://apps.who.int/iris/bitstream/handle/10665/260494/9789241550079-eng.pdf;jsessionid=9E2BD49130618683CDA3C987CABB97D1?sequence=1
- Van Deun A ,
- Salim MAH ,
- Phillips PPJ ,
- Meredith SK ,
- STREAM Study Collaborators
- ↵ World Health Organization. Consolidated guidelines on drug-resistant tuberculosis treatment. 2019. https://apps.who.int/iris/bitstream/handle/10665/311389/9789241550529-eng.pdf
- ↵ World Health Organization. Rapid Communication: Key changes to the treatment of drug-resistant tuberculosis. 2019. https://www.who.int/tb/publications/2019/WHO_RapidCommunicationMDR_TB2019.pdf?ua=1
- Chalmers TC ,
- Smith H Jr .
- McKenna L ,
- ↵ US Food and Drug Administration. FDA approves new drug for treatment-resistant forms of tuberculosis that affects the lungs. https://www.fda.gov/news-events/press-announcements/fda-approves-new-drug-treatment-resistant-forms-tuberculosis-affects-lungs
- Carroll MW ,
- Migliori GB
- Brighenti S ,
- Bergman P ,
- Martineau AR
- J Libardo MD ,
- Boshoff HI ,
- Barry CE 3rd .
- O’Brien RJ ,
- Global Alliance for TB Drug Development
- ↵ US Food and Drug Administration. Guidance for industry: pulmonary tuberculosis: developing drugs for treatment. 2013. https://www.fda.gov/media/87194/download
- Bonnett LJ ,
- Ken-Dror G ,
- Wiesner L ,
- Strydom N ,
- Prideaux B ,
- Zimmerman MD ,
- Sterling TR ,
- Tuberculosis Trials Consortium, the AIDS Clinical Trials Group for the PREVENT TB Trial (TBTC Study 26ACTG 5259) The investigators of the TB Trials Consortium and the AIDS Clinical Trials Group for the PREVENT TB Trial are listed in the Supplement, item 17
- Belknap R ,
- Holland D ,
- TB Trials Consortium iAdhere Study Team
- Borisov AS ,
- Bamrah Morris S ,
- Menzies D ,
- Adjobimey M ,
- Ruslami R ,
- Kanouse J ,
- Wegener D ,
- ↵ Frick M. An activist’s guide to rifapentine for the treatment of TB infection. 2019. http://www.treatmentactiongroup.org/sites/default/files/rifapentine_guide_2019_web_final2.pdf
- MacLean E ,
- Yerlikaya S ,
- Fernandez-Carballo BL ,
- Denkinger CM
- Penn-Nicholson A ,
- Scriba TJ ,
- ACS and GC6-74 cohort study groups
- Fiore-Gartland A ,
- ↵ Panel on antiretroviral guidelines for adults and adolescents. Guidelines for the use of antiretroviral agents in adults and adolescents with HIV. 2019. https://aidsinfo.nih.gov/contentfiles/lvguidelines/AdultandAdolescentGL.pdf
- ↵ Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents. Guidelines for the prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: recommendations from the CDC, the NIH, and the IDSA. 2019. https://aidsinfo.nih.gov/contentfiles/lvguidelines/adult_oi.pdf
- Gabillard D ,
- Temprano ANRS 12136 Study Group
- ↵ TEMPRANO ANRS 12136 Study Group, Danel C, Moh R, et al. A trial of early antiretrovirals and isoniazid preventive therapy in Africa. N Engl J Med 2015;373:808-22.
- Lundgren JD ,
- Babiker AG ,
- INSIGHT START Study Group
- Abdool Karim SS ,
- Grobler A ,
- Blanc F-X ,
- Laureillard D ,
- CAMELIA (ANRS 1295–CIPRA KH001) Study Team
- Havlir DV ,
- Kendall MA ,
- AIDS Clinical Trials Group Study A5221
- ↵ US Department of Health and Human Services. Clinical Guidelines. 2020. https://aidsinfo.nih.gov/guidelines
- Mfinanga SG ,
- Kirenga BJ ,
- Chanda DM ,
- Grinsztejn B ,
- Hosseinipour MC ,
- Ribaudo HJ ,
- HPTN 052-ACTG Study Team
- Swindells S ,
- Ramchandani R ,
- BRIEF TB/A5279 Study Team
- ↵ Dooley KE, Churchyard GJ, Savic RM, et al. Safety and PK of weekly rifapentine/isoniazid (3HP) in adults with HIV on dolutegravir. In: Conference on Retroviruses and Opportunistic Infections . Seattle, WA: 2019. http://www.croiconference.org/sessions/safety-pk-weekly-rifapentineisoniazid-3hp-adults-hiv-dolutegravir
- Boehme CC ,
- Hillemann D ,
- Schumacher SG ,
- Chakravorty S ,
- Armstrong DT ,
- Allix-Béguec C ,
- Arandjelovic I ,
- CRyPTIC Consortium and the 100,000 Genomes Project
- Meehan CJ ,
- Mahomed S ,
- Padayatchi N
- Takiff HE ,
- Colman RE ,
- Seifert M ,
- Alipanah N ,
- ↵ World Health Organization. Guidelines for treatment of drug-susceptible tuberculosis and patient care. 2017. https://apps.who.int/iris/bitstream/handle/10665/255052/9789241550000-eng.pdf
- Aarnoutse RE ,
- Alffenaar JWC ,
- Maartens G ,
- Lancet Respiratory Medicine drug-resistant tuberculosis Commission group
- ↵ World Health Organization. WHO consolidated guidelines on tuberculosis: tuberculosis preventive treatment. Geneva: 2020. https://apps.who.int/iris/bitstream/handle/10665/331170/9789240001503-eng.pdf?sequence=1&isAllowed=y .
- ↵ Dooley KE, Rosenkranz SL, Conradie F, et al. QT effects of bedaquiline, delamanid or both in MDR-TB patients: the DELIBERATE trial. Abstract 84. In: Conf Retroviruses Opportun Infect . Seattle, WA. 2019. https://www.croiconference.org/sessions/qt-effects-bedaquiline-delamanid-or-both-mdr-tb-patients-deliberate-trial
- ↵ World Health Organization. A patient-centred approach to TB care. 2018. https://www.who.int/iris/handle/10665/272467
- Roberts B ,
- van den Boom M ,
Tuberculosis
Book editors.
- PMID: 30212088
- Bookshelf ID: NBK525174
- DOI: 10.1596/978-1-4648-0524-0_ch11
Despite 90 years of vaccination and 60 years of chemotherapy, tuberculosis (TB) remains the world’s leading cause of death from an infectious agent, exceeding human immunodeficiency virus/acquired immune deficiency syndrome (HIV/AIDS) for the first time (WHO 2015b, 2016a). The World Health Organization (WHO) estimates that there are about 10.4 million new cases and 1.8 million deaths from TB each year. One-third of these new cases (about 3 million) remain unknown to the health system, and many are not receiving proper treatment.
Tuberculosis is an infectious bacterial disease caused by Mycobacterium tuberculosis (Mtb), which is transmitted between humans through the respiratory route and most commonly affects the lungs, but can damage any tissue. Only about 10 percent of individuals infected with Mtb progress to active TB disease within their lifetime; the remainder of persons infected successfully contain their infection. One of the challenges of TB is that the pathogen persists in many infected individuals in a latent state for many years and can be reactivated to cause disease. The risk of progression to TB disease after infection is highest soon after the initial infection and increases dramatically for persons co-infected with HIV/AIDS or other immune-compromising conditions.
Treatment of TB disease requires multiple drugs for many months. These long drug regimens are challenging for both patients and health care systems, especially in low- and middle-income countries (LMICs), where the disease burden often far outstrips local resources. In some areas, the incidence of drug-resistant TB, requiring even longer treatment regimens with drugs that are more expensive and difficult to tolerate, is increasing.
Diagnosis in LMICs is made primarily by microscopic examination of stained smears of sputum of suspected patients; however, smear microscopy is capable of detecting only 50–60 percent of all cases (smear-positive). More sensitive methods of diagnosing TB and detecting resistance to drugs have recently become available, although they are more expensive. The time between the onset of disease and when diagnosis is made and treatment is initiated is often protracted, and such delays allow the transmission of disease. Although bacille Calmette–Guérin (BCG) remains the world’s most widely used vaccine, its effectiveness is geographically highly variable and incomplete. Modeling suggests that more effective vaccines will likely be needed to drive tuberculosis toward elimination in high-incidence settings.
The basic strategy to combat TB has been, for 40 years, to provide diagnosis and treatment to individuals who are ill and who seek care at a health facility. The premise is that, if patients with active disease are cured, mortality will disappear, prevalence of disease will decline, transmission will decline, and therefore incidence should decline. The reality in many countries is more complex, and overall the decline in incidence (only about 1.5 percent per year) has been unacceptably slow.
Chemotherapy for TB is one of the most cost-effective of all health interventions (McKee and Atun 2006). This evidence has been central to the global promotion of the WHO and Stop TB Partnership policy of directly observed therapy, short course (DOTS) strategy, the package of measures combining best practices in the diagnosis and care of patients with TB (UN General Assembly 2000). The DOTS strategy to control tuberculosis promotes standardized treatment, with supervision and patient support that may include, but is far broader than, direct observation of therapy (DOT), where a health care worker personally observes the patient taking the medication (WHO 2013a).
Thanks in part to these efforts and national and international investments, much progress has been made in TB control over the past several decades. Between 1990 and 2010, absolute global mortality from TB declined 18.7 percent, from 1.47 million to 1.20 million (Lozano and others 2012) and by 22 percent between 2000 and 2015 (WHO 2016a). By 2015, an estimated 49 million lives had been saved (WHO 2016a). The internationally agreed targets for TB, embraced in the United Nations (UN) Millennium Development Goals (MDGs), sought “to halt and reverse the expanding incidence of tuberculosis by 2015,” and this target has been met to some extent in all six WHO regions and in most, but not all, of the world’s 22 high-burden countries (WHO 2014c).
Despite progress, major gaps persist. Although the Sustainable Development Goals (SDGs) seek to end the tuberculosis epidemic altogether (WHO 2015a, 2015c), the decline in incidence has been disappointing. One of every three TB patients remains “unknown to the health system,” many are undiagnosed and untreated, and case detection and treatment success rates remain too low in the high-burden countries. Ominously, rates of multidrug-resistant (MDR) TB—defined as resistance to the two major TB drugs, isoniazid and rifampicin—are rising globally (WHO 2011a) with the emergence of extensively drug-resistant (XDR) TB, resistant to many second-line drugs, as well as strains resistant to all current drugs (Dheda and others 2014; Udwadia and others 2012; Uplekar and others 2015). These are now primarily the result of transmission rather than inadequate treatment (Shah and others 2017).
Moreover, the TB problem has become more pressing because of co-infection with HIV/AIDS. While globally HIV/AIDS and TB co-infection represents only 11 percent of the total TB burden, in some areas of Sub-Saharan Africa with a high burden of TB, as many as three-quarters of TB patients are co-infected with HIV/AIDS. In those countries, efforts to control TB are overwhelmed by the rising number of TB cases occurring in parallel with the HIV/AIDS epidemic. And after decades of steady decline, the incidence of TB is also increasing in some high-income countries (HICs), mainly as the result of outbreaks in vulnerable groups (WHO 2015b).
If the ultimate goal of controlling an infectious disease is to interrupt transmission, turning the tide on TB will require early and accurate case detection, rapid commencement of and adherence to effective treatment that prevents transmission, and, where possible, preventive treatment of latent TB. It is universally understood that new strategies and more effective tools and interventions will be required to reach post-2015 targets (Bloom and Atun 2016; WHO 2015a). These interventions must be not only cost-effective, but also affordable and capable of having an impact on a very large scale.
TB control will need three new advances—development of new point-of-care diagnostics, more effective drug regimens to combat drug-susceptible and drug-resistant TB, and more effective vaccines. As argued in this chapter, these require new strategies and tools that include moving away from the traditional DOTS passive case finding and toward more active case finding in high-burden regions; service delivery that is targeted to the most vulnerable populations and integrated with other services, especially HIV/AIDS services; and care that is based at the primary health care and community levels. Specifically, in high-burden countries, many individuals with TB are asymptomatic, such that waiting for patients to become sick enough to seek care has not been sufficient to reduce transmission and incidence markedly (Bates and others 2012; Mao and others 2014; Willingham and others 2001; Wood and others 2007). A more active and aggressive approach is needed that tackles health system barriers to effective TB control.
The strategies for controlling TB recommended by the WHO have evolved significantly over time. In the early formulations, the central tenets of the global TB control strategy were clinical and programmatic in nature, focusing principally on the delivery of standardized drug regimens; the underlying assumption was that the problem could be solved largely by existing biomedical tools (Atun, McKee, and others 2005; Schouten and others 2011). Yet, in many LMICs, health system weaknesses in governance, financing, health workforce, procurement and supply chain management, and information systems have impeded TB control (Elzinga, Raviglione, and Maher 2004; Marais and others 2010; Travis and others 2004) and not been adequately addressed by TB control efforts. The current global TB strategy, formulated as the End TB Strategy, is the most comprehensive ever, with three major pillars:
Integrated, patient-centered care and prevention
Social and political action to address determinants of disease
Recognition of the urgent need for research to provide new tools (WHO 2015a).
Health systems are important and need to be strengthened. As with other health interventions, the success of tuberculosis treatment and control in a country is often determined by the strength of its health system (McKee and Atun 2006; WHO 2003). A health system can be defined in many ways, perhaps best as “all the activities whose primary purpose is to promote, restore, or maintain health” (WHO 2000, 5).
In a sense, the major risk factor for acquiring TB is breathing. Thus, people of all social and economic statuses are at risk. While TB disproportionately affects the poor, the narrative that TB is a disease only of the poor is misleading and counterproductive, if it leads either to further stigmatization of the disease or to the view that middle- and high-income countries need not worry about the disease. In the case of co-infection with HIV/AIDS, evidence suggests that HIV/AIDS is often more prevalent in better-off populations in Africa that suffer high rates of TB.
The analytical framework underlying this chapter defines key functions of the health system, ultimate goals, and contextual factors that affect the health system (figure 11.1). It builds on the WHO framework (WHO 2000) as well as health system frameworks developed by Frenk (1994), Hsiao and Heller (2007), and Roberts and others (2004), and national accounts (OECD, Eurostat, and WHO 2011). It also draws on earlier studies by Atun (2012); Atun and Coker (2008); Atun, Samyshkin, and others (2006); Samb and others (2009); and Swanson and others (2012).
The four key health system functions represented in the framework are as follows:
Governance and organization. The policy and regulatory environment; stewardship and regulatory functions of the ministry of health and its relation to other levels of the health system; and structural arrangements for insurers and purchasers, health care providers, and market regulators
Financing. The way funds are collected, funds and risks are pooled, finances are allocated, and health care providers are remunerated
Resource management. The way resources—physical, human, and intellectual—are generated and allocated, including their geographic and needs-based allocation
Service delivery. Both population- and individual-level public health interventions and health care services provided in community, primary health care, hospitals, and other health institutions.
Each of these functions is influenced by the economic, demographic, legal, cultural, and political context.
As the framework suggests, health system goals include better health, financial protection, and user satisfaction. Personal health services and public health interventions should be organized to achieve an appropriate balance of equity (including reducing out-of-pocket [OOP] expenditures and impoverishment of individuals and families), efficiency, effectiveness (that is, the extent to which interventions are evidence based and safe), responsiveness, equity, and client satisfaction (as perceived by the users of services).
This chapter is organized as follows. First, we provide a detailed discussion of the global burden of disease and clinical context, followed by a review of approaches to diagnosis, treatment, and prevention. The aim throughout is to approach TB through a health system lens and, in the latter part of the chapter, to provide recommendations for improving delivery strategies and strengthening health systems, including care, supply chain, and information systems. Because the current tools for combating TB are seriously inadequate, we conclude with sections on critical research and development and economic analyses of new interventions for diagnosis, treatment, and vaccines. Throughout, emphasis is placed on data or modeling of the economic costs and benefits, where available, of current or possible future interventions to combat this disease.
The chapter recommends moving toward active case finding in high-burden countries; greater investments in health systems; community-based rather than hospital-based service delivery; and greater support for research on new tools—that is, developing better diagnostics, treatment regimens, and vaccines. Most of these approaches were included in earlier WHO policies, but were not emphasized. They are now part of the WHO’s End TB Strategy, with which this report is fully consistent (WHO 2015a, 2015c).
© 2017 International Bank for Reconstruction and Development / The World Bank.
- Historical Trends, Current Burden, and Global Response
- Infection and Disease in Individuals and Populations
- TB Diagnosis and Screening
- TB Treatment
- TB Prevention
- Turning the Tide Against TB
- Research and Development
- Financing for TB Programs
- Economic Analyses and Cost-Effectiveness
- Extended Cost-Effectiveness Analysis of Universal Public Financing of TB Treatment
- Summary and Recommendations
Publication types
- Open access
- Published: 06 March 2024
Iron-related gene mutations driving global Mycobacterium tuberculosis transmission revealed by whole-genome sequencing
- Yameng Li 1 na1 ,
- Yifan Li 2 na1 ,
- Yao Liu 3 ,
- Xianglong Kong 4 ,
- Ningning Tao 3 ,
- Yawei Hou 5 ,
- Tingting Wang 1 ,
- Qilin Han 6 ,
- Yuzhen Zhang 6 ,
- Fei Long 2 &
- Huaichen Li 1 , 3
BMC Genomics volume 25 , Article number: 249 ( 2024 ) Cite this article
203 Accesses
1 Altmetric
Metrics details
Iron plays a crucial role in the growth of Mycobacterium tuberculosis ( M. tuberculosis ). However, the precise regulatory mechanism governing this system requires further elucidation. Additionally, limited studies have examined the impact of gene mutations related to iron on the transmission of M. tuberculosis globally. This research aims to investigate the correlation between mutations in iron-related genes and the worldwide transmission of M. tuberculosis .
A total of 13,532 isolates of M. tuberculosis were included in this study. Among them, 6,104 (45.11%) were identified as genomic clustered isolates, while 8,395 (62.04%) were classified as genomic clade isolates. Our results showed that a total of 12 single nucleotide polymorphisms (SNPs) showed a positive correlation with clustering, such as Rv1469 ( ctpD , C758T), Rv3703c ( etgB , G1122T), and Rv3743c ( ctpJ , G676C). Additionally, seven SNPs, including Rv0104 (T167G, T478G), Rv0211 ( pckA , A302C), Rv0283 ( eccB3 , C423T), Rv1436 ( gap , G654T), ctpD C758T, and etgB C578A, demonstrated a positive correlation with transmission clades across different countries. Notably, our findings highlighted the positive association of Rv0104 T167G, pckA A302C, eccB3 C423T, ctpD C758T, and etgB C578A with transmission clades across diverse regions. Furthermore, our analysis identified 78 SNPs that exhibited significant associations with clade size.
Conclusions
Our study reveals the link between iron-related gene SNPs and M. tuberculosis transmission, offering insights into crucial factors influencing the pathogenicity of the disease. This research holds promise for targeted strategies in prevention and treatment, advancing research and interventions in this field.
Peer Review reports
Tuberculosis (TB) is an airborne infectious disease caused by Mycobacterium tuberculosis ( M. tuberculosis ) and is the leading cause of death worldwide among infectious diseases. Despite great progress over the past decades, TB remains a major global health problem. In 2022, TB remained the second leading cause of death from a single infectious agent, after coronavirus disease (COVID-19), and caused almost twice as many deaths as HIV/AIDS [ 1 ]. Globally, there were 7.5 million newly diagnosed cases of TB reported in 2022. Additionally, the total number of deaths attributed to TB, including those among individuals with HIV, reached 1.30 million during the same year [ 1 ]. Despite the immense global burden of tuberculosis, our understanding of the factors influencing its transmission remains limited. Therefore, gaining a deeper insight into the mechanisms underlying the transmission of M. tuberculosis is imperative in order to inform and guide effective strategies for tuberculosis control, ultimately leading to a reduction in the societal burden imposed by this disease.
Iron holds paramount importance as an indispensable element for nearly all living organisms due to its involvement in a vast array of metabolic processes, encompassing oxygen transportation, DNA synthesis, and electron conveyance [ 2 ]. In the context of M. tuberculosis , iron emerges as an essential catalyst for growth. The significance of iron in the growth and metabolism of bacteria is elucidated through its acquisition from host reservoirs like transferrin, lactoferrin, and ferritin, followed by subsequent assimilation and utilization within the bacterial framework. Crucial constituents participating in the procurement of iron (in the form of ferric ion) and its preliminary transference into the mycobacterium cell encompass extracellular iron-binding agents, known as siderophores. In pathogenic mycobacteria, carboxymycobactins fulfill this role, while exochelins perform analogous functions in saprophytic mycobacteria [ 3 , 4 ]. Upon successful acquisition, the next imperative step entails transporting iron across the mycobacterium cell membrane. M. tuberculosis employs specialized systems to facilitate this process. Subsequently, inside the mycobacterium cell, iron finds employment in diverse metabolic pathways, functioning as a pivotal cofactor for enzymes engaged in critical processes such as DNA synthesis, respiration, and energy production [ 3 , 5 ]. Furthermore, iron plays a crucial role in regulating gene expression and maintaining redox homeostasis [ 6 ]. M. tuberculosis exploits iron to disrupt host immune responses, thereby enhancing its survival and dissemination. In summary, iron contributes to the establishment and survival of M. tuberculosis within the host. By utilizing the iron resources, the bacterium can better adapt to the host environment and increase its transmission capacity. Iron plays a key role in the growth, pathogenicity, immune evasion, and host adaptation of M. tuberculosis . However, the specific regulatory mechanisms of iron-related genes involved in the dissemination of M. tuberculosis remain unclear. Further research is needed to uncover these mechanisms, providing insights into the pathogenesis of tuberculosis and facilitating the development of more effective treatment strategies.
Whole-genome sequencing (WGS) is progressively being used to investigate the transmission dynamics of M. tuberculosis . In this study, we employed WGS to analyze the impact of mutations in iron-related genes on the global transmission of M. tuberculosis . Specifically, the genome cluster and clade were used to represent the transmission of M. tuberculosis .
Sample Collection
Between 2011 and 2018, a total of 1,550 culture-positive cases of M. tuberculosis were collected from two medical institutions in China, namely the Shandong Public Health Clinical Research Center (SPHCC) and the Weifang Respiratory Clinical Hospital (WRCH). The study did not include cases with positive culture of M. tuberculosis that were previously evaluated and subsequently treated.
DNA extraction and sequencing
A total of 1447 isolates were included in this study, and genomic DNA was extracted from these isolates using the Cetyltrimethylammonium Bromide (CTAB) method. Prior to analysis, quality control (QC) procedures were conducted on the extracted DNA. However, 103 isolates of M. tuberculosis were excluded from further analysis due to issues related to improper handling during DNA extraction and poor quality of the extracted DNA. For the remaining isolates, their genomes were sequenced utilizing the Illumina HiSeq 4000 system. The resulting sequence data were then deposited in the National Center for Biotechnology Information (NCBI) under the BioProject PRJNA1002108. In addition to the aforementioned isolates, this study also included a larger dataset consisting of 13,267 isolates of M. tuberculosis collected from 52 countries and 18 regions worldwide, as reported in previous studies [ 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 ]. To accurately map the reference genome of the standard isolate M. tuberculosis H37Rv, we employed the BWA-MEM (version 0.7.17-r1188). Our analysis focused solely on samples with a coverage rate of 98% or higher and a minimum depth of at least 20× [ 16 ]. In summary, a total of 13,532 genomes were analyzed in this study, please refer to Additional file 1: Tables S14-S15 for the specific sample numbers.
Single nucleotide polymorphism (SNP) analysis
We performed variant calling using Samclip (version 0.4.0) and SAMtools (version 1.15). Following variant calling, we applied additional filtering steps to refine the resulting variants. This involved utilizing Free Bayes (version 1.3.2) and Bcftools (version 1.15.1) for further variant filtering. To ensure the accuracy of our analysis, we excluded SNPs located within repeat regions. This included polymorphic GC-rich sequences found in PE/PPE genes, direct repeat SNPs, and repeat bases identified through the use of Tandem Repeat Finder (version 4.09) and RepeatMask (version 4.1.2-P1) [ 17 , 18 ]. Finally, SNP annotation was conducted using SnpEff v 4.1 l. The resulting output was obtained by utilizing the Python programming language [ 19 ].
Phylogenetic analysis
According to Coll et al [ 20 ] (Additional file 2: Tables S14-S15), the isolates in this study were classified into different lineages. To construct the maximum likelihood phylogenetic tree, we utilized the IQ-TREE software package (version 1.6.12). The JC nucleotide substitution model and gamma model of rate heterogeneity were used, with 100 bootstrap replicates included for statistical support [ 21 ]. During the analysis, Mycobacterium canettii CIPT140010059 was identified as an outlier and was treated accordingly. The resulting phylogenetic tree was visualized using iTOL ( https://itol.embl.de/ ) for better representation and interpretation.
Propagation analysis
We employed cluster and clade analysis to investigate the impact of mutations in iron-related genes on the transmission of M. tuberculosis [ 22 ]. Expanding upon previous studies [ 23 ], clustering techniques were utilized to define transmitted clusters, using a threshold of less than 12 SNPs. Additionally, clade analysis was conducted to identify transmission clades, with a threshold of less than 25 SNPs. To further categorize the transmission clades, we adopted a classification system used by scholars. The clades were classified into three groups based on size: large (above the 75th percentile), medium (between the 25th and 75th percentiles), and small (below the 25th percentile) [ 24 ]. For a comprehensive analysis of the global distribution patterns and transmission dynamics of M. tuberculosis isolates, we classified them into cross-country and within-country clades. Cross-country clades consist of isolates from two or more different countries. Furthermore, based on geographic location, the M. tuberculosis isolates were classified into cross-regional and within-regional clades using the United Nations standard regions (UN M.49). Cross-regional clades include isolates from two or more different regions.
Acquisition of iron-related genes
In our study, we obtained genes related to iron in M. tuberculosis from the NCBI database, which were previously discovered by scholars. These genes encompass various aspects such as iron uptake transporters, iron storage proteins, iron-regulated transcription factors, and enzymes involved in iron-dependent processes. A total of 59 iron-related genes were retrieved from the NCBI database. Python was utilized to detect mutations in genes associated with iron (Additional file 1: Table S16).
Modeling and statistical analysis
The data were presented as percentages. Positions with mutation frequency below 0.01 in the iron-related genes were excluded from the analysis [ 25 ]. For statistical analysis, we employed generalized linear mixed models in the R statistical language (R 4.2.3). To further analyze the data, random forest and gradient boosting decision tree algorithms were implemented using Python 3.7.4 with the Scikit-learn library (Python Software Foundation, USA; Packt Publishing, UK). The dataset was randomly divided into a training set and a test set in a 7:3 ratio. In order to assess the impact of mutations in iron-related genes on clade size, Spearman’s rank correlation analysis was performed using R version 4.2.3. Confounding factors such as lineage and geographical location were taken into account during all analyses. All statistical analyses were conducted using SPSS 26.0. Two-tailed tests were used, and statistical significance was defined as a P -value below 0.05.
Sample description
We included a total of 13,532 isolates of M. tuberculosis from around the world, with 1,445 isolates collected between 2011 and 2018 at the Shandong Public Health Clinical Research Center (SPHCC) and the Weifang Respiratory Clinical Hospital (WRCH). Among these isolates, the highest proportion was observed in Eastern Asia ( n = 3,172, 23.44%), followed by Eastern Africa ( n = 1,728, 12.77%) and Northern America ( n = 1,646, 12.16%), as depicted in Fig. 1 . Additionally, the majority of these isolates ( n = 6,499, 48.03%) belonged to lineage 4, while ( n = 5,135, 37.95%) belonged to lineage 2, aligning with our expectations. Isolates were divided into clusters based on < = 12 single nucleotide polymorphisms (SNPs). Accordingly, a total of 6,104 isolates clustered together, resulting in a clustering rate of 0.45. Within the lineage 4 group, 2,971 (45.71%) isolates formed clusters, while within the lineage 2 group, 2,131 (41.50%) isolates formed clusters. When applying a threshold of 25 SNPs for clades, a total of 8,395 isolates clade together, resulting in a clade rate of 0.62. The M. tuberculosis isolates were further grouped into 2,218 clades, with the number of isolates per clade ranging from 2 to 224 isolates. Within these clades, there were 177 cross-country clades, consisting of 2 to 4 countries, and 171 cross-regional clades, consisting of 2 to 4 regions, as shown in Table 1 . The phylogenetic tree of M. tuberculosis isolates was constructed as described in Fig. 2 .
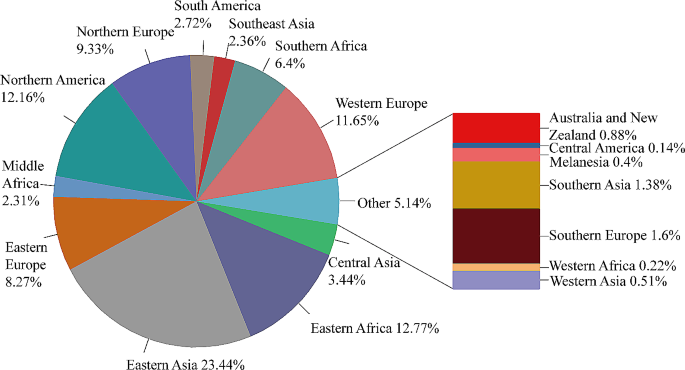
Distribution of Mycobacterium tuberculosis in various regions
Phylogenetic tree for the Mycobacterium tuberculosis isolates from China
Relationship between iron-related gene mutations and transmission clusters
After excluding sites with a mutation frequency below 0.01, we identified and included a total of 90 SNPs for further analysis. Subsequently, we conducted a comparative analysis between clustered and non-clustered isolates, examining the relationship between these 90 SNPs and the occurrence of clustering. The generalized linear mixed model (GLMM) revealed that 21 SNPs were statistically significant for clustering ( P < 0.05) (Additional file 1: Table S1 ). Among these, eight nonsynonymous SNPs and five synonymous SNPs showed a positive correlation with transmission clusters in M. tuberculosis isolates. The specific SNPs included Rv0197 (G344T, T2247G), Rv0252 ( nirB , C2037T, A2058G), Rv0338c C2478T, Rv1229c ( mrp , C649G), Rv1436 ( gap , G654T), Rv1469 (ctpD, C758T), Rv2869c ( rip , C957T, G775T), Rv3703c ( etgB , G1122T), Rv3728 (C2392T), and Rv3743c ( ctpJ , G676C). Two prediction models were established using random forest and gradient boosting decision tree, we found that Rv0197 (G344T, T2247G), nirB (C2037T, A2058G), Rv0338c (C2478T), mrp C649G, ctpD C758T, rip (C957T, G775T), etgB G1122T, Rv3728 C2392T, and ctpJ G676C also contributed most to the random forest and gradient boosting decision tree (Additional file 1: Table S4 , Table S9 and Additional file 2: Fig. S1 ). However, the gap SNP G654T did not contribute significantly to the gradient boosting decision tree model. Overall, our results indicated that Rv0197 (G344T, T2247G), nirB (C2037T, A2058G), Rv0338c (C2478T), mrp C649G, ctpD C758T, rip (C957T, G775T), etgB G1122T, Rv3728 C2392T, and ctpJ G676C were positively correlated with transmission clusters of M. tuberculosis isolates.
Relationship between iron-related gene mutations and transmission clusters of lineages
After excluding the mutation frequency below 0.01, we identified and included a total of 40 SNPs for further analysis. In comparison to non-clustered isolates, we conducted an analysis on the relationship between 40 SNPs and clustered isolates specifically belonging to lineage 2. The GLMM revealed that five SNPs showed statistical significance for clustering ( P < 0.05) (Table 2 ). Among these, two nonsynonymous SNPs and two synonymous SNPs displayed a positive correlation with clustering. These significant SNPs included Rv0197 T2247G, Rv1553 (f rdB , C87T), and Rv2869c ( rip , C957T, G775T). Two prediction models were established using random forest and gradient boosting decision tree algorithms (Additional file 1: Table S5 , Table S10 and Additional file 2: Fig. S2 ). Our findings demonstrated that Rv0197 T2247G, frdB C87T, and rip (C957T, G775T) contributed significantly to both the random forest and gradient boosting decision tree models. Overall, our results indicated that the SNPs Rv0197 T2247G, frdB C87T, and rip (C957T, G775T) were positively correlated with transmission clusters within M. tuberculosis isolates of lineage 2.
After excluding sites with a mutation frequency less than 0.01, we identified and included a total of 68 SNPs for further analysis. In comparison to non-clustered isolates, we conducted an analysis on the relationship between 68 SNPs and clustered isolates specifically belonging to lineage 4. The GLMM showed that 20 SNPs were found to be statistically significant for clustering ( P < 0.05) (Additional file 1: Table S2 ), among which eight nonsynonymous SNPs and five synonymous SNPs were positively correlated with clustering, including Rv0069c ( sdaA , A565G), Rv0197 T2247G, Rv0233 ( nrdB , C97G), Rv0338c C2478T, Rv1207 ( folP2 , C153A), Rv1436 ( gap , G654T), Rv2711 ( ideR , G57A), Rv3025c ( iscS , C1101G), Rv3703c ( etgB , G1122T, C578A), Rv3728 C2392T, Rv3743c ( ctpJ , G676C), Rv3818 G373A. Two prediction models were established using random forest and gradient boosting decision tree (Additional file 1: Table S6 , Table S11 and Additional file 2: Fig. S3 ). We found that Rv0197 T2247G, nrdB C97G, Rv0338c C2478T, folP2 C153A, gap G654T, ideR G57A, etgB (G1122T, C578A), ctpJ G676C, and Rv3818 G373A also contributed most to the random forest and gradient boosting decision tree. Overall, our results indicated that the SNPs Rv0197 T2247G, nrdB C97G, Rv0338c C2478T, folP2 C153A, gap G654T, ideR G57A, etgB G1122T, C578A, ctpJ G676C, and Rv3818 G373A were positively correlated with transmission clusters within M. tuberculosis isolates of lineage 4.
Relationship between iron-related gene mutations and cross-country transmission
After excluding sites with a mutation frequency below 0.01, we identified and included a total of 90 SNPs in iron-related genes that were analyzed to assess their relationship with cross-country transmission clades. The GLMM showed that 20 SNPs were found to be statistically significant for transmission clades of cross-country ( P < 0.05) (Table 3 ), among which five nonsynonymous SNPs and two synonymous SNPs were positively correlated with transmission clades, including Rv0104 (T167G, T478G), Rv0211 ( pckA , A302C), Rv0283 ( eccB3 , C423T), Rv1436 ( gap , G654T), Rv1469 ( ctpD , C758T), Rv3703c ( etgB , C578A). Two prediction models were established using random forest and gradient boosting decision tree (Additional file 1: Table S7, Table S12 and Additional file 2: Fig. S4 ), we found that Rv0104 (T167G, T478G), pckA A302C, eccB3 C423T, gap G654T, ctpD C758T, etgB C578A also contributed most to the random forest and gradient boosting decision tree. Overall, our results showed that Rv0104 (T167G, T478G), pckA A302C, eccB3 C423T, gap G654T, ctpD C758T, and etgB C578A were positively correlated with transmission clades across different countries.
Relationship between iron-related gene mutations and cross-regional transmission
After excluding sites with a mutation frequency below 0.01, we identified and included a total of 90 SNPs of iron-related genes. The GLMM showed that 12 SNPs were found to be statistically significant for cross-regional transmission clades ( P < 0.05) (Additional file 1: Table S3 ), among which four nonsynonymous SNPs and a synonymous SNP were positively correlated with cross-regional transmission clades, including Rv0104 T167G, Rv0211 ( pckA , A302C), Rv0283 ( eccB3 , C423T), Rv1469 ( ctpD , C758T), Rv3703c ( etgB , C578A). Two prediction models, random forest and gradient boosting decision tree, were established (Additional file 1: Table S8, Table S13 and Additional file 2: Fig. S5 ). The results demonstrated that Rv0104 T167G, pckA A302C, eccB3 C423T, ctpD C758T, and etgB C578A contributed significantly to both the random forest and gradient boosting decision tree models. Overall, our findings indicated that Rv0104 T167G, pckA A302C, eccB3 C423T, ctpD C758T, etgB C578A were positively correlated with transmission clades across different regions.
Relationship between iron-related gene mutations and clade size
After excluding sites with a mutation frequency less than 0.01, we identified and included a total of 90 iron-related gene SNPs. The results showed that 78 SNPs were significantly associated with clade size ( P < 0.05), among which 22 nonsynonymous SNPs and 11 synonymous SNPs were positively correlated with clade size, including eccB3 C423T, ctpD C758T, etgB C578A, rip C957T, etgB G1122T, and ctpJ G676C. For further details refer to Fig. 3 .
Correlation analysis of iron-related gene mutations and clade size
We have identified a relationship between iron-related gene mutations and the transmission of M. tuberculosis in this study. This included cluster transmission, characterized by 12 SNPs, and clade transmission, characterized by 25 SNPs. Our research findings indicated that globally, lineage 2 and lineage 4 dominate among M. tuberculosis isolates. Specifically, within the clusters defined by 12 SNPs, lineage 4 ( n = 3528, 57.80%) and lineage 2 ( n = 2131, 34.91%) were the primary contributors. Similarly, within the clades defined by 25 SNPs, lineage 4 ( n = 4577, 54.52%) and lineage 2 ( n = 2999, 35.72%) constitute the majority. This suggested that the transmission of M. tuberculosis was primarily driven by lineage 2 and lineage 4. Moreover, our findings also revealed 176 cross-country transmission clades. Among these, eight transmission clades involved three countries, while the transmission clade 254 extended across four nations: Peru, South Africa, India, and Thailand (see Fig. 4 ). These patterns of cross-continental transmission transcended the typical spread observed between neighboring countries. The distributional tendencies were likely intertwined with the prevalence of modern-day social activities, such as international trade, travel, and other forms of social interaction.
Distribution of cross-country transmission clades of Mycobacterium tuberculosis involves three or more countries
According to our study, two nonsynonymous SNPs of G344T and T2247G in Rv0197 increased the risk of transmission clusters. We also noticed the SNP of T2247G in Rv0197 was positively associated with transmission clusters of lineage2 and lineage4, which has previously been shown to be associated with enhanced transmissibility in vivo [ 26 ]. In addition, the frequent and independent occurrence in Lineage4.3/ Latin American and Mediterranean sub-lineage clonal complex (TUN4.3_CC1) of the in vivo enhanced transmission-associated mutation in Rv0197 T2247G, could have contributed to its evolutionary success. We understand that the protein encoded by the Rv0197 gene plays a critical role in bacterial metabolism and respiration, particularly as a putative iron oxidoreductase enzyme. We understand that the protein encoded by the Rv0197 gene plays a critical role in bacterial metabolism and respiration, particularly as a putative iron oxidoreductase enzyme. Therefore, these SNP variations could potentially lead to structural or functional changes in the protein, thus influencing bacterial physiology [ 27 ]. Furthermore, we speculate that these SNP variations might help the bacteria adapt and survive in specific environments or hosts, possibly through alterations in host immune evasion, growth regulation, or metabolic pathways. Rv2869c is a mechanism of transmembrane signal transduction that functions through intramembrane proteolysis of substrates [ 28 ]. Our research revealed that the nonsynonymous SNP G775T and synonymous SNP C957T in Rv2869c were positively associated with transmission clusters, especially those belonging to lineage 2. Further supporting evidence from Hideki Makinoshima et al. demonstrated that Rv2869c played a regulatory role in cell envelope composition, in vivo growth, and in vivo persistence of M. tuberculosis , while also controlling multiple cell envelope-based virulence determinants [ 29 ]. Based on these collective findings, we hypothesized that these two mutations (G775T and C957T) potentially induced functional changes in the Rv2869c protein, impacting the formation and structure of the bacterial cell envelope, thereby influencing the transmission potential of M. tuberculosis. The gene Rv0338c encoded IspQ, a membrane-bound protein containing 2Fe-2 S and 4Fe-4 S centers, which was believed to serve as an iron-sulfur binding oxidoreductase. Given its essential role in the β-oxidation process of M. tuberculosis , mutations in Rv0338c had the potential to affect oxygen reduction reactions involved in bacterial metabolism and respiration. Further research was needed to fully comprehend the specific consequences of this synonymous mutation on the functionality of Rv0338c and its impact on bacterial physiology. Our study findings demonstrated a positive correlation between the synonymous SNP C2478T in Rv0338c and transmission clusters, particularly within lineage 4. This indicated that this specific SNP variation may have contributed to the adaptation and transmission dynamics within distinct lineages. Notably, studies had shown that mutants lacking the etfD gene, which interacted with Rv0338c , exhibited impaired growth on fatty acids or cholesterol, as well as reduced survival and growth in murine infection models [ 30 , 31 ]. Our findings further underscored the significance of Rv0338c and its associated genes in mycobacterial physiology and pathogenesis. In our study, we also discovered that the nonsynonymous SNP G676C in Rv3743c , the nonsynonymous SNP G1122T in Rv3703c , and the nonsynonymous SNP C578A in Rv3703c were positively associated with transmission clusters, specifically those associated with lineage 4 isolates. Rv3743c is known as a cation transporter/ATPase, while Rv3703c is classified as an iron (II)-dependent oxidoreductase. However, the precise functional roles of these genes in the context of M. tuberculosis are not yet fully understood and require further investigation.
In our analysis of transmission clades, which includes cross-regional, cross-country, and clade size, we found a positive correlation between the nonsynonymous SNP C758T in Rv1469 and cross-regional transmission, cross-country transmission, and clade size. Additionally, the nonsynonymous SNP T980G and synonymous SNP C1350T in Rv1469 were positively associated with clade size. Rv1469 is one of the coding genes for homologous P1B4-ATPase [ 32 ]. It belongs to the ATPase superfamily and functions as a transmembrane protein involved in the transport and regulation of metal ions. The Rv1469 gene encodes a membrane protein annotated as the M. tuberculosis paralog of Rv1469 , a member of the metal cation-transporting P1B4-ATPase subgroup. It plays an essential role in M. tuberculosis survival within the host. Specifically, Rv1469 acts as a high-affinity Fe 2+ exporter required for overcoming redox stress and adapting to the host environment [ 32 , 33 ]. the nonsynonymous SNPs T167G in Rv0104 , A302C in Rv0211 , and C578A in Rv3703c were positively correlated with cross-regional transmission, cross-country transmission, and clade size. However, the specific functions of Rv0104 , Rv0211 , and Rv3703c remain unclear. It is worth noting that these associations suggest a potential link between these genetic variations and the sspread of tuberculosis across different regions and countries. However, without a clear understanding of the functions of these genes, it is difficult to determine the exact mechanisms underlying this correlation.
Additionally, our study also elucidated the association between SNPs in other iron-related genes and the transmission of M. tuberculosis . These genetic mutations have the potential to alter diverse physiological functions of the bacterium that are intricately linked to its transmission. By altering these iron-related pathways, SNPs in these genes may impact the fitness, virulence, or adaptive capabilities of the bacterium. This, in turn, could influence its ability to establish infections, replicate, evade host immune responses, and transmit to new hosts. Furthermore, our findings provided confirmation that both synonymous and non-synonymous mutations can impact the transmission of M. tuberculosis . This indicates that synonymous mutations in iron-related genes are not universally neutral, which aligns with previous studies by Xukang Shen suggesting that synonymous mutations in yeast genes are predominantly strong non-neutral mutations [ 34 ].
In this study, we have identified correlations between mutations in iron-related genes and the transmission of M. tuberculosis . However, it is important to acknowledge several limitations and shortcomings of our research. Firstly, although we have established these correlations, the specific impact of these mutations on the transmission dynamics of M. tuberculosis lacks experimental validation. Further research is needed to investigate the functional significance of these mutations and their direct influence on the transmission of the bacteria. Moreover, it is worth noting that mutations in iron-related genes may also affect other factors related to pathogenesis, such as bacterial virulence and immune response. These potential influences warrant further in-depth investigations. Understanding the broader implications of these mutations requires additional studies aimed at exploring their effects on various aspects of TB pathogenesis.
The findings of this study indicate that mutations in iron-related genes could potentially elevate the risk of M. tuberculosis transmission, underscoring the importance of conducting additional research to explore the impact of these mutations on the control and dissemination of M. tuberculosis . These results offer significant insights that can inform the development of therapeutic interventions for tuberculosis.
Data availability
The whole genome sequences have been submitted to the NCBI under the accession number PRJNA1002108.
Abbreviations
- Mycobacterium tuberculosis
- Whole-genome sequencing
Shandong Public Health Clinical Research Center
Weifang Respiratory Clinical Hospital
Cetyltrimethylammonium Bromide
Quality control
Single nucleotide polymorphism
Single nucleotide polymorphisms
National Center for Biotechnology Information
World Health Organization. Global tuberculosis report 2023. Geneva: World Health Organization; 2023.
Google Scholar
Lieu PT, Heiskala M, Peterson PA, Yang Y. The roles of iron in health and disease. Mol Aspects Med. 2001;22:1–87.
Article CAS PubMed Google Scholar
Sharma AK, Naithani R, Kumar V, Sandhu SS. Iron Regulation in Tuberculosis Research: Promise and challenges. CMC. 2011;18:1723–31.
Article CAS Google Scholar
Rodriguez GM. Control of iron metabolism in Mycobacterium tuberculosis. Trends Microbiol. 2006;14:320–7.
Gobin J, Moore CH, Reeve JR, Wong DK, Gibson BW, Horwitz MA. Iron acquisition by Mycobacterium tuberculosis: isolation and characterization of a family of iron-binding exochelins. Proc Natl Acad Sci U S A. 1995;92:5189–93.
Article CAS PubMed PubMed Central ADS Google Scholar
Hoffmann C, Leis A, Niederweis M, Plitzko JM, Engelhardt H. Disclosure of the mycobacterial outer membrane: Cryo-electron tomography and vitreous sections reveal the lipid bilayer structure. Proc Natl Acad Sci U S A. 2008;105:3963–7.
Chen X, He G, Wang S, Lin S, Chen J, Zhang W. Evaluation of whole-genome sequence method to Diagnose Resistance of 13 anti-tuberculosis drugs and characterize resistance genes in clinical Multi-drug Resistance Mycobacterium tuberculosis isolates from China. Front Microbiol. 2019;10:1741.
Article PubMed PubMed Central Google Scholar
Yang C, Luo T, Shen X, Wu J, Gan M, Xu P, et al. Transmission of multidrug-resistant Mycobacterium tuberculosis in Shanghai, China: a retrospective observational study using whole-genome sequencing and epidemiological investigation. Lancet Infect Dis. 2017;17:275–84.
Koster KJ, Largen A, Foster JT, Drees KP, Qian L, Desmond E, et al. Genomic sequencing is required for identification of tuberculosis transmission in Hawaii. BMC Infect Dis. 2018;18:608.
Article CAS PubMed PubMed Central Google Scholar
Hicks ND, Yang J, Zhang X, Zhao B, Grad YH, Liu L, et al. Clinically prevalent mutations in Mycobacterium tuberculosis alter propionate metabolism and mediate multidrug tolerance. Nat Microbiol. 2018;3:1032–42.
Liu Q, Ma A, Wei L, Pang Y, Wu B, Luo T, et al. China’s tuberculosis epidemic stems from historical expansion of four strains of Mycobacterium tuberculosis. Nat Ecol Evol. 2018;2:1982–92.
Huang H, Ding N, Yang T, Li C, Jia X, Wang G, et al. Cross-sectional whole-genome sequencing and epidemiological study of Multidrug-resistant Mycobacterium tuberculosis in China. Clin Infect Dis. 2019;69:405–13.
Luo T, Comas I, Luo D, Lu B, Wu J, Wei L, et al. Southern East Asian origin and coexpansion of Mycobacterium tuberculosis Beijing family with Han Chinese. Proc Natl Acad Sci USA. 2015;112:8136–41.
Jiang Q, Liu Q, Ji L, Li J, Zeng Y, Meng L, et al. Citywide transmission of Multidrug-resistant tuberculosis under China’s Rapid Urbanization: a Retrospective Population-based genomic spatial epidemiological study. Clin Infect Dis. 2020;71:142–51.
Article PubMed Google Scholar
Coll F, Phelan J, Hill-Cawthorne GA, Nair MB, Mallard K, Ali S, et al. Genome-wide analysis of multi- and extensively drug-resistant Mycobacterium tuberculosis. Nat Genet. 2018;50:307–16.
Jung Y, Han D. BWA-MEME: BWA-MEM emulated with a machine learning approach. Bioinformatics. 2022. btac137.
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, et al. The sequence Alignment/Map format and SAMtools. Bioinformatics. 2009;25:2078–9.
Liu F, Zhang Y, Zhang L, Li Z, Fang Q, Gao R, et al. Systematic comparative analysis of single-nucleotide variant detection methods from single-cell RNA sequencing data. Genome Biol. 2019;20:242.
Cingolani P, Platts A, Wang LL, Coon M, Nguyen T, Wang L, et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin). 2012;6:80–92.
Coll F, McNerney R, Guerra-Assunção JA, Glynn JR, Perdigão J, Viveiros M, et al. A robust SNP barcode for typing Mycobacterium tuberculosis complex strains. Nat Commun. 2014;5:4812.
Article CAS PubMed ADS Google Scholar
Nguyen L-T, Schmidt HA, von Haeseler A, Minh BQ. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol. 2015;32:268–74.
Seto J, Wada T, Suzuki Y, Ikeda T, Mizuta K, Yamamoto T, et al. Mycobacterium tuberculosis Transmission among Elderly persons, Yamagata Prefecture, Japan, 2009–2015. Emerg Infect Dis. 2017;23:448–55.
Walker TM, Ip CL, Harrell RH, Evans JT, Kapatai G, Dedicoat MJ, et al. Whole-genome sequencing to delineate Mycobacterium tuberculosis outbreaks: a retrospective observational study. Lancet Infect Dis. 2013;13:137–46.
Chiner-Oms Á, Sánchez-Busó L, Corander J, Gagneux S, Harris SR, Young D et al. Genomic determinants of speciation and spread of the Mycobacterium tuberculosis complex. Sci Adv. 2019.
Farhat MR, Freschi L, Calderon R, Ioerger T, Snyder M, Meehan CJ, et al. GWAS for quantitative resistance phenotypes in Mycobacterium tuberculosis reveals resistance genes and regulatory regions. Nat Commun. 2019;10:2128.
H N-G A, van Mr L, Vacm F, Jj K. M, A Z, Transmissible Mycobacterium tuberculosis strains share genetic markers and Immune phenotypes. Am J Respir Crit Care Med. 2017;195.
Dekhil N, Mardassi H. Genomic changes underpinning the emergence of a successful Mycobacterium tuberculosis Latin American and Mediterranean clonal complex. Front Microbiol. 2023;14:1159994.
Sklar JG, Makinoshima H, Schneider J, Glickman MS. M. Tuberculosis intramembrane protease Rip1 controls transcription through three anti-sigma factor substrates. Mol Microbiol. 2010;77:605–17.
Makinoshima H, Glickman MS. Regulation of M. Tuberculosis cell envelope composition and virulence by regulated Intramembrane Proteolysis. Nature. 2005;436:406.
Székely R, Rengifo-Gonzalez M, Singh V, Riabova O, Benjak A, Piton J, et al. 6,11-Dioxobenzo[f]pyrido[1,2-a]indoles kill Mycobacterium tuberculosis by Targeting Iron-Sulfur protein Rv0338c (IspQ), a putative Redox Sensor. ACS Infect Dis. 2020;6:3015–25.
Beites T, Jansen RS, Wang R, Jinich A, Rhee KY, Schnappinger D, et al. Multiple acyl-CoA dehydrogenase deficiency kills Mycobacterium tuberculosis in vitro and during infection. Nat Commun. 2021;12:6593.
Raimunda D, Long JE, Padilla-Benavides T, Sassetti CM, Argüello JM. Differential roles for the Co2+/Ni2 + transporting ATPases, CtpD and CtpJ, in Mycobacterium tuberculosis virulence. Mol Microbiol. 2014;91. https://doi.org/10.1111/mmi.12454 .
Patel SJ, Lewis BE, Long JE, Nambi S, Sassetti CM, Stemmler TL, et al. Fine-tuning of substrate Affinity leads to alternative roles of Mycobacterium tuberculosis Fe2+-ATPases. J Biol Chem. 2016;291:11529–39.
Shen X, Song S, Li C, Zhang J. Synonymous mutations in representative yeast genes are mostly strongly non-neutral. Nature. 2022;606:725–31.
Download references
Acknowledgements
We thank Shandong Public Health Clinical Research Center and Weifang Respiratory Clinical Hospital for providing us with the clinical sample data. Additionally, we extend our thanks to all the authors who have shared their sequence datasets on NCBI.
This research was supported by the Natural Science Foundation of Shandong Provincial.
(No. ZR2020KH013; No. ZR2021MH006; No. ZR2022QH259), the Department of Science & Technology of Shandong Province (CN) (No. 2007GG30002033; No. 2017GSF218052), and the Jinan Science and Technology Bureau (CN) (No. 201704100).
Author information
Yameng Li and Yifan Li contributed equally to this work.
Authors and Affiliations
Clinical Department of Integrated Traditional Chinese and Western Medicine , The First Clinical Medical College of Shandong University of Traditional Chinese Medicine, 250014, Jinan, Shandong, People’s Republic of China
Yameng Li, Tingting Wang & Huaichen Li
Department of Pulmonary and Critical Care Medicine, The Third Affiliated Hospital of Shandong First Medical University (Affiliated Hospital of Shandong Academy of Medical Sciences), 250031, Jinan, Shandong, People’s Republic of China
Yifan Li & Fei Long
Department of Pulmonary and Critical Care Medicine, Shandong Provincial Hospital Affiliated to Shandong University, Shandong Provincial Hospital Affiliated to Shandong First Medical University, 250021, Jinan, Shandong, People’s Republic of China
Yao Liu, Ningning Tao & Huaichen Li
Artificial Intelligence Institute, Qilu University of Technology (Shandong Academy of Sciences), 250011, Jinan, Shandong, People’s Republic of China
Xianglong Kong
Institute of Chinese Medical Literature and Culture of Shandong University of Traditional Chinese Medicine, 250355, Jinan, Shandong, People’s Republic of China
Shandong First Medical University & Shandong Academy of Medical Sciences, 250117, Jinan, Shandong, People’s Republic of China
Qilin Han & Yuzhen Zhang
You can also search for this author in PubMed Google Scholar
Contributions
HCL, FL, YML, and YFL participated in the study design. FL, YL, HCL, YML, XLK, NNT, and YFL performed data collection and statistical analyses. YL, TTW, YZZ, and YWH helped draft the manuscript. YWH, QLH, and YZZ overviewed and supervised the project. All authors read and approved the final manuscript.
Corresponding authors
Correspondence to Fei Long or Huaichen Li .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Ethics approval and consent to participate
This study complies with the Declaration of Helsinki, and was approved by the Ethics Committee of Shandong Provincial Hospital, affiliated with Shandong University (SPH), the Ethics Weifang Respiratory Clinical Hospital (WRCH) and the Ethics Committee of Shandong Provincial Chest Hospital (SPCH), which waived informed patient consent because all patient records and information were anonymized and deidentified before the analysis.
Consent for publication
Not applicable.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12864_2024_10152_MOESM1_ESM.docx
Supplementary Material 1: Additional file 1 Table S1 - S8
12864_2024_10152_MOESM2_ESM.xlsx
Supplementary Material 2: Additional file 1 Table S9 - S13
12864_2024_10152_MOESM3_ESM.xlsx
Supplementary Material 3: Additional file 1 Table S14
12864_2024_10152_MOESM4_ESM.xlsx
Supplementary Material 4: Additional file 1 Table S15
12864_2024_10152_MOESM5_ESM.xlsx
Supplementary Material 5: Additional file 1 Table S16
12864_2024_10152_MOESM6_ESM.docx
Supplementary Material 6: Additional file 2 Fig. S1 - S5
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Li, Y., Li, Y., Liu, Y. et al. Iron-related gene mutations driving global Mycobacterium tuberculosis transmission revealed by whole-genome sequencing. BMC Genomics 25 , 249 (2024). https://doi.org/10.1186/s12864-024-10152-1
Download citation
Received : 29 November 2023
Accepted : 21 February 2024
Published : 06 March 2024
DOI : https://doi.org/10.1186/s12864-024-10152-1
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Iron-related gene mutations
- Transmission
BMC Genomics
ISSN: 1471-2164
- Submission enquiries: [email protected]
- General enquiries: [email protected]
- Research article
- Open access
- Published: 17 November 2021
The impact of behavioural risk factors on communicable diseases: a systematic review of reviews
- Sara Wood 1 ,
- Sophie E. Harrison 2 , 3 ,
- Natasha Judd 1 , 2 ,
- Mark A. Bellis ORCID: orcid.org/0000-0001-6980-1963 1 , 2 ,
- Karen Hughes 1 , 2 &
- Andrew Jones 4
BMC Public Health volume 21 , Article number: 2110 ( 2021 ) Cite this article
11k Accesses
11 Citations
25 Altmetric
Metrics details
The coronavirus (COVID-19) pandemic has highlighted that individuals with behavioural risk factors commonly associated with non-communicable diseases (NCDs), such as smoking, harmful alcohol use, obesity, and physical inactivity, are more likely to experience severe symptoms from COVID-19. These risk factors have been shown to increase the risk of NCDs, but less is known about their broader influence on communicable diseases. Taking a wide focus on a range of common communicable diseases, this review aimed to synthesise research examining the impact of behavioural risk factors commonly associated with NCDs on risks of contracting, or having more severe outcomes from, communicable diseases.
Literature searches identified systematic reviews and meta-analyses that examined the association between behavioural risk factors (alcohol, smoking, illicit drug use, physical inactivity, obesity and poor diet) and the contraction/severity of common communicable diseases, including infection or associated pathogens. An a priori, prospectively registered protocol was followed (PROSPERO; registration number CRD42020223890).
Fifty-three systematic reviews were included, of which 36 were also meta-analyses. Reviews focused on: tuberculosis, human immunodeficiency virus, hepatitis C virus, hepatitis B virus, invasive bacterial diseases, pneumonia, influenza, and COVID-19. Twenty-one reviews examined the association between behavioural risk factors and communicable disease contraction and 35 examined their association with communicable disease outcomes (three examined their association with both contraction and outcomes). Fifty out of 53 reviews (94%) concluded that at least one of the behavioural risk factors studied increased the risk of contracting or experiencing worse health outcomes from a communicable disease. Across all reviews, effect sizes, where calculated, ranged from 0.83 to 8.22.
Conclusions
Behavioural risk factors play a significant role in the risk of contracting and experiencing more severe outcomes from communicable diseases. Prevention of communicable diseases is likely to be most successful if it involves the prevention of behavioural risk factors commonly associated with NCDs. These findings are important for understanding risks associated with communicable disease, and timely, given the COVID-19 pandemic and the need for improvements in future pandemic preparedness. Addressing behavioural risk factors should be an important part of work to build resilience against any emerging and future epidemics and pandemics.
Peer Review reports
The recent coronavirus (COVID-19) pandemic has highlighted that individuals with potentially modifiable behavioural risk factors that are commonly associated with non-communicable diseases (NCDs), such as smoking, harmful alcohol use, obesity and physical inactivity, are more likely to experience severe symptoms from COVID-19 infection [ 1 ], resulting in greater risk of hospitalisation [ 2 ]. With these behavioural risk factors often having higher prevalence in the poorest communities, COVID-19 has disproportionately impacted those already suffering the greatest risks of ill health, thereby widening health and social inequalities [ 3 ]. Indeed, due to its associations with existing health and social risk factors, COVID-19 has been referred to as a syndemic; one in which existing health and social challenges increase an individual’s susceptibility to disease [ 4 ]. However, whilst addressing behavioural risk factors is routinely considered in the prevention of NCDs, their role in the contraction of communicable disease, and severity of symptoms in those who are infected, has had a lower public health prominence.
Many modifiable behavioural risk factors are highly prevalent among adults and adolescents in both higher (HICs) and lower and middle income countries (LMICs) [ 5 , 6 ], with levels increasing in many LMICs (e.g. obesity, alcohol) [ 7 , 8 ]. As a result, NCDs, such as cancer, respiratory disease and cardiovascular disease are the highest cause of mortality and morbidity in HICs and account for a rapidly increasing proportion of both in LMICs [ 9 ]. Across countries globally, the burden of NCDs has been found to correlate with levels of COVID-19 cases and deaths [ 10 ].
With both international commerce and tourism connecting populations globally, it is highly likely that COVID-19 is only one in a series of existing and emerging infectious diseases likely to impact, to different extents, health and well-being on a global scale [ 11 ]. Although the exact nature or source of any future epidemic or pandemic threat is speculative, behavioural risk factors have also been found to increase the risk of infection and subsequent poorer outcomes across a range of other communicable diseases [ 12 , 13 , 14 ]. Understanding which factors may increase or reduce risk of contraction and severity of disease can provide important intelligence, both in increasing a population’s resilience to infectious disease, and in identifying which communities and individuals may be most at risk from the spread of different types of disease. Although previous research has explored links between behavioural risk factors and individual communicable diseases, few studies have synthesised information across a wider range of communicable diseases and their relationships with behavioural risks. Indeed, such relationships may elucidate how future pandemics threats will exploit behavioural risk factors.
Intending to explore whether communicable diseases and NCDs share a common set of behavioural risk factors, the aim of this review was to provide a synthesis of existing research examining the impact of behavioural risk factors commonly associated with NCDs on the risk of people (adults or children) contracting, or experiencing more severe outcomes from, common communicable diseases. With the breadth of communicable diseases requiring limitation, the focus of this review was on diseases common to high income countries. With an intentionally wide focus on a range of communicable diseases, the review focused specifically on systematic reviews and meta-analyses, clarifying existing knowledge and highlighting gaps in evidence to inform priority areas for future research.
This review was carried out in adherence to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. An a priori protocol was followed and prospectively registered at the National Institute for Health Research international prospective register of systematic reviews (PROSPERO) (registration number CRD42020223890). The focus of this review was limited to behavioural risk factors and communicable diseases common in HICs, regardless of the geographical location of the review. Those more specific to LMICs or certain regions of the world (e.g. tropical diseases) were considered best examined in a separate study.
Search strategy
A systematic search was performed across multiple databases through ProQuest covering the 10-year period 28th October 2010 to 28th October 2020. Preliminary scans of the literature reviews and discussion between members of the research team were used to aid selection of common behavioural risk factors and communicable diseases. Thus, alcohol use, smoking, physical inactivity, obesity, illicit drug use and poor diet were chosen as behavioural risk factors, covering some of the most common behavioural contributors to NCDs [ 15 ]. The same process identified: Tuberculosis (TB), acquired immune deficiency syndrome (AIDS), human immunodeficiency virus (HIV), viral hepatitis, COVID-19, severe acute respiratory syndrome (SARS), middle-east respiratory syndrome (MERS), pneumonia, influenza, and meningitis as communicable diseases (including infections and pathogens) feasible for review. These communicable diseases were broadly consistent with some of the most prevalent disease/infection categories reported in the global burden of disease study for HICs (excluding those categories predominantly affecting specific groups (e.g. maternal, neonatal) or where a component may be associated with non-infectious causes (e.g. diarrheal) [ 9 ]) and with previous outbreaks or epidemics involving HICs [ 16 ]. In addition, it was intended to include diseases that arose from both bacterial and viral pathogens, with a range of transmission types, e.g. airborne, droplet, fomite, blood-borne and contact. Combinations of search terms were developed based on these key risk factors and diseases. Search results were restricted to English language and peer reviewed systematic reviews and meta-analyses. Whilst this strategy restricted literature to that which qualified for inclusion in systematic reviews, it allowed for the inclusion of multiple behavioural risk factors and communicable diseases at the same time. The search was restricted to a 10-year period, allowing coverage of a broad range of behavioural risk factors and communicable diseases, yet limiting the literature to a manageable volume. The full search strategy is available in Supplementary file 1 . Searches also included poor housing conditions as a risk factor given the impact of housing conditions on respiratory disease [ 17 ], but this study focuses specifically on behavioural risk factors.
Study selection and eligibility criteria
To identify eligible studies, the titles and abstracts of studies retrieved were screened by two reviewers, with a sample of 15% screened independently by both reviewers and achieving 98.5% agreement (SH, NJ). Discrepancies were resolved between reviewers. For full text screening, ten reviews (7%) were initially screened by three reviewers (SH, NJ, SW), with results later discussed and discrepancies resolved. Following this, the full text screening was divided across reviewers and any reviews that were not a clear exclude/include (20%) were discussed and agreed between reviewers. This meant that, across all reviews screened by full text, 27% were discussed and agreed by more than one reviewer. Where full texts could not be accessed, authors were contacted to request the full text.
Systematic reviews and meta-analyses of observational studies (including cohort and case-control studies) that examined the association between an identified behavioural risk factor and the contraction or outcomes of an identified communicable disease (including infection or related pathogens) were included in the review. Since there are no single definitions of the selected risk factors across the literature, all reviews that focused on an identified risk factor were included regardless of the definition used in the review (definitions are provided in Tables 1 and 2 and in the results section). An aim of the review was to explore risks associated with illicit drug use in general. However, it was recognised that injection drug use can be a mechanism of transmission for some pathogens relating to included communicable diseases (e.g. HIV, HCV). Studies that focused specifically on injection drug use were therefore included alongside those focusing on drug use more generally. Studies were excluded if they: were not a systematic review or meta-analysis; did not examine the association between an selected behavioural risk factor and the contraction or outcomes of an selected communicable disease; included only selected specialist sub-populations (e.g. sex workers, prisoners), or included sub-populations relating to a risk factor (e.g. people who inject drugs) without a general population comparison; or included behavioural risk factors or communicable diseases not relevant to HICs (see above). No restrictions were made for the age of participants included. A flow chart demonstrating the selection process is presented in Fig. 1 .
PRISMA flow diagram of study identification, inclusion and exclusion
Data extraction and synthesis
Data were extracted by three reviewers (SH, NJ, SW) into a standardised, pre-piloted form. Each extraction was duplicated across reviewers and discrepancies resolved through discussion. Information extracted from the studies included: title, authors, abstract, behavioural risk factor(s) studied, communicable disease(s) studied (including infection or related pathogens), research question, geographical restrictions, population characteristics, number of reviews included in the systematic review or meta-analysis, main findings (including odds ratios (OR), relative risks (RR) or rate ratios where available), proposed mechanisms of association and conclusions related to identified behavioural risk factor(s) and communicable disease(s). Where information on the number of reviews included for each risk factor was not reported, the corresponding author of the paper was contacted for additional information.
Due to the variety of different communicable diseases, risk factor definitions, outcome measures, and methods of reporting in the included studies, as well as the challenges of conducting meta-analysis for observational studies [ 71 ], findings were not combined statistically through meta-analysis. Instead, a narrative synthesis of the findings was constructed [ 72 ], and effect size ranges reported for each behavioural risk factor. Key information that would have enabled calculation of a common effect size was often not available. To calculate these effect size ranges, it was assumed that ORs, RRs and rate ratios were approximately equivalent, a method suggested for umbrella reviews in these circumstances [ 73 ]. In addition, where studies reported reduced risk of a communicable disease with a health behaviour (e.g. physical activity, no alcohol drinking), an inverse OR (1/OR) for the corresponding risk behaviour was reported. Findings were structured according to the identified behavioural risk factors and their association with a) contraction of the identified communicable diseases, and b) experiencing more severe outcomes from these communicable diseases. Some study conclusions were amended for readability, to aid understanding. Further, where study conclusions were not relevant to the current research question, information was extracted from results sections and amended for readability (see Supplementary file 2 ).
Methodological quality of studies
The methodological quality of included studies was assessed using the Overview Quality Assessment Questionnaire (OQAQ); a frequently used, validated tool for assessing the methodological quality of systematic reviews [ 74 ]. Methodological quality assessment was carried out by three researchers (NJ, SH, SW), with any discrepancies resolved through discussion. Assessment ratings are available in Supplementary file 2 .
The database search yielded 1806 citations, of which 53 were included (Fig. 1 ). Research relating to the following communicable diseases (including infection and pathogens) was identified: TB, HIV, hepatitis C virus (HCV), hepatitis B virus (HBV), invasive bacterial disease (IBD), pneumonia, influenza, and COVID-19. No studies relating to SARS, MERS or meningitis were identified. Thirty-six of the identified systematic reviews also conducted meta-analyses. Reviews used a range of definitions of behavioural risk factors (e.g. current or former smoker, any alcohol use or heavy alcohol use). All definitions were included in the synthesis, and are presented for clarity in each section of the results and in the results tables (Tables 1 and 2 ). Eighteen reviews examined the association between behavioural risk factors and the contraction of a communicable disease only, 32 reviews examined the association between behavioural risk factors and the outcomes from communicable diseases only, and three reviews examined associations with both contraction of and outcomes from communicable diseases. Characteristics of all included reviews and their conclusions can be found in Supplementary file 2 . No systematic review had extensive or major flaws, with most reviews having only minimal or minor flaws (Supplementary file 2 ). Consequently, no reviews were excluded based on methodological quality. A breakdown of reviews by disease and risk factors is provided in Supplementary file 3 .
Behavioural risk factors for communicable diseases
Overall, 50 out of 53 reviews (94%) concluded that at least one of the behavioural risk factors studied increased the risk of contracting or having more severe outcomes of a communicable disease. Across all reviews, effect sizes, where calculated, ranged from 0.83 to 8.22 (Figs. 2 and 3 ; Tables 1 and 2 ). Nineteen out of 21 reviews (90%) concluded that at least one of the behavioural risk factors studied increased the risk of contracting a communicable disease (Table 1 ). Across all contraction reviews, effect sizes, where calculated, ranged from 1.03 to 8.22 (Fig. 2 ). Thirty-two out of 35 reviews (91%) concluded that at least one of the behavioural risk factors studied increased the likelihood of having more severe outcomes from a communicable disease (Table 2 ). Across all outcome reviews, effect sizes, where calculated, ranged from 0.83 to 3.96 (Fig. 3 ).
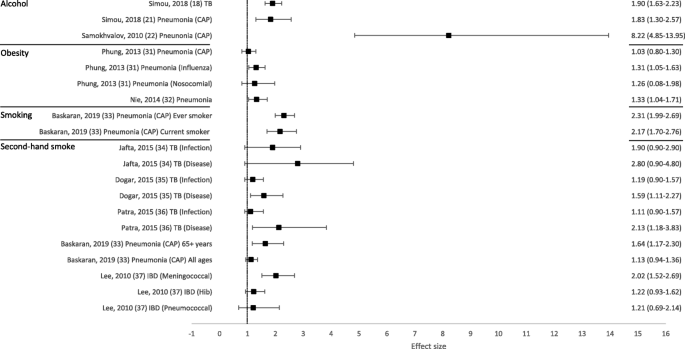
Forest plot of meta-analysis effect sizes: contraction of a communicable disease. Effect sizes refer to odds ratios and relative risks, see Table 1 for more information. CAP = community acquired pneumonia; TB = tuberculosis; IBD = invasive bacterial disease
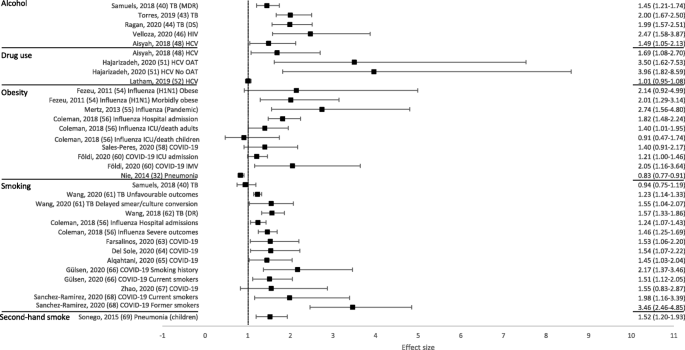
Forest plot of meta-analysis effect sizes: more severe communicable disease outcomes. Effect sizes refer to odds ratios, relative risks and rate ratios, see Table 2 for more information. MDR = multi-drug resistant; TB = tuberculosis; DS = drug-susceptible; HIV = human immunodeficiency virus; HCV = hepatitis C virus; ICU = intensive care unit; IMV = invasive mechanical ventilation; OAT = opioid agonist therapy
Alcohol as a risk factor
Seventeen reviews included alcohol as a risk factor for a communicable disease, with a range of definitions used: any alcohol consumption [ 19 , 23 , 41 , 43 , 44 , 46 , 47 , 49 ]; any alcohol use or higher amounts [ 18 , 21 ]; binge drinking or alcohol misuse [ 20 ]; alcohol misuse [ 40 ]; alcohol use disorder (AUD) [ 22 , 45 ]; alcoholism [ 39 , 42 ]; or current/history of excess use [ 48 ]. The majority of reviews reported an increased risk of contraction (5/6 reviews; Table 1 ) and more severe outcomes (10/12 reviews; Table 2 ). Across all alcohol reviews, effect sizes, where calculated, ranged from 1.83–8.22 for contraction (Fig. 2 ) and 1.45–2.47 for severe outcomes (Fig. 3 ). Alcohol use (any use, higher amounts, binge drinking or AUD) was reported to increase the risk of contracting TB [ 18 ], HIV [ 20 ], pneumonia [ 21 , 22 ] and invasive pneumococcal diseases (IPD) [ 23 ]. One review did not draw a conclusion, but reported mixed findings for the association between alcohol consumption and contraction of TB [ 19 ]. Alcohol use (any use, misuse, current/history of excess use, alcoholism or AUD) was reported to increase the risk of having more severe outcomes from TB [ 39 , 40 , 41 , 42 , 43 , 44 ], HIV [ 45 , 46 ], and HCV [ 48 , 49 ]. One review reported mixed findings and made no clear conclusion about the association of alcohol consumption and TB outcomes [ 19 ], and one review reported variable results among studies examining the association between alcohol consumption and the progression of HIV [ 47 ].
Illicit drug use as a risk factor
Fifteen reviews examined the association between illicit drug use and communicable disease contraction or outcomes, with a wide range of definitions used: drug abuse [ 19 , 41 ]; illicit drug use [ 25 ]; regular/problem cocaine use [ 50 ]; recent drug use [ 51 ]; and injection drug use [ 20 , 24 , 26 , 27 , 28 , 29 , 39 , 48 , 52 , 53 ]. The majority of reviews reported an increased risk of contraction (8/9 reviews; Table 1 ) and more severe outcomes (6/8 reviews; Table 2 ). Across all drug use reviews, effect sizes, where calculated (for more severe outcomes only), ranged from 1.01–3.96 (Fig. 3 ). Both injection drug use and illicit drug use were reported to increase the risk of contracting TB [ 24 ] and HIV [ 20 , 24 , 25 ], whilst the prevalence of HCV was found to be higher among people who inject drugs (PWID) compared to general population or community groups [ 26 , 27 , 28 , 29 ]. One review did not draw a conclusion but reported mixed findings for the association between drug abuse and TB contraction [ 19 ]. Both injecting drug use and drug use/abuse were reported to increase the risk of having more severe outcomes from TB [ 39 , 41 ], HIV [ 50 ] and HCV [ 48 , 51 ]. Furthermore, among those with HBV, the prevalence of hepatitis D (HDV; co-infection with HDV is considered a more severe form of viral hepatitis) was substantially higher for PWID compared to a mixed population with no risk factors [ 53 ]. One review did not draw a conclusion but reported mixed findings for the association between drug abuse and TB outcomes [ 19 ], and one review concluded that treatment outcomes for HCV were similar between people who currently were and were not injecting drugs [ 52 ].
Physical inactivity as a risk factor
One systematic review was identified examining the association between physical activity and communicable disease contraction or outcomes. This study reported an association between increased prolonged, moderate aerobic exercise and reduced influenza-related mortality, and improved immunocompetence [ 38 ].
Obesity as a risk factor
Ten reviews focused on the relationship between obesity [ 30 , 31 , 54 , 55 , 56 , 57 , 58 , 60 ], or overweight and obesity [ 32 , 59 ], and communicable disease risk. The majority of reviews reported an increased risk of contraction (3/3 reviews; Table 1 ) and more severe outcomes (8/9 reviews; Table 2 ). Across all obesity reviews, effect sizes, where calculated, ranged from 1.03–1.33 for contraction (Fig. 2 ) and 0.83–2.74 for severe outcomes (Fig. 3 ). Obesity was reported to increase the risk of contracting influenza [ 30 ] and pneumonia [ 31 , 32 ]. Obesity was reported to increase the risk of having more severe outcomes from influenza [ 30 , 54 , 55 , 56 ] and COVID-19 [ 58 , 59 , 60 ]. One review concluded that most studies showed some degree of association between higher body mass index (BMI) and a worse clinical presentation of COVID-19 and the need for hospitalisation. This review suggested that obesity seemed to predict poor clinical evolution in patients with COVID-19, but that studies in the review had limited methodological quality [ 57 ]. However, one review, which concluded that obesity increased the risk of contracting pneumonia, also found that obese individuals had a lower mortality risk from pneumonia [ 32 ].
Smoking as a risk factor
Eighteen reviews examined the association between smoking (current, past or both) and communicable disease contraction or outcomes. The majority of reviews reported some evidence of an increased risk of contraction (3/3 reviews; Table 1 ) and more severe outcomes (13/15 reviews; Table 2 ). Across all smoking reviews, effect sizes, where calculated, ranged from 2.17–2.31 for contraction (Fig. 2 ) and 0.94–3.46 for severe outcomes (Fig. 3 ). Smoking was reported to increase the risk of contracting HIV [ 20 ], pneumonia [ 33 ] and invasive pneumococcal disease (IPD) [ 23 ]. Further, smoking was reported to increase the risk of having more severe outcomes from TB [ 19 , 41 , 42 , 43 , 61 , 62 ], influenza [ 56 ], and COVID-19 [ 63 , 64 , 65 , 66 , 68 ]. Two reviews reported no associations between smoking and more severe outcomes from communicable diseases, including death from TB [ 39 ] and TB treatment outcomes [ 40 ]. One review reported that active smoking may increase the risk of severe COVID-19, but found the result was heavily influenced by one study [ 67 ].

Second-hand smoke as a risk factor
Six reviews focused on second-hand smoke as a risk factor for a communicable disease. The majority of reviews reported some evidence of an increased risk of contraction (4/5 reviews; Table 1 ) and more severe outcomes (1/1 review; Table 2 ). Across all second-hand smoking reviews, effect sizes, where calculated, ranged from 1.11 to 2.80 for contraction (Fig. 2 ), and the one effect size calculated for severe outcomes was 1.52 (Fig. 3 ). One review suggested that second-hand smoke exposure increased the risk of TB infection and disease [ 34 ]. The remaining four reviews reported at least some evidence of second-hand smoke exposure increasing the risk of contracting a communicable disease, including TB [ 35 , 36 ], pneumonia (among those aged 65+ only) [ 33 ], and IBD (invasive meningococcal disease [IMD] only) [ 37 ]. Second-hand smoke exposure was reported to increase the risk of severe outcomes from acute lower respiratory infections (ALRIs), including pneumonia [ 69 ].
Poor diet as a risk factor
Only two reviews were identified that examined the association between poor diet and communicable disease outcomes, and no reviews examining the association between poor diet and communicable disease contraction were identified. One review found that vitamin D status may influence the course of HIV disease [ 70 ]. The second review reported that a high intake of polyunsaturated fatty acids was associated with non-response to HCV antiviral therapy [ 49 ].
The key finding of this systematic review is that behavioural risk factors play a significant role in the risk of contracting, and having more severe outcomes from, common communicable diseases. To the authors’ knowledge, this is the first time that a review has brought together studies exploring the impact of behavioural risk factors on a range of communicable diseases. Whilst the focus on selected communicable diseases and use of systematic reviews has led to inevitable gaps, the findings nevertheless provide strong evidence that both NCDs and communicable diseases share a common set of behavioural risk factors. This work indicates that the prevention of communicable disease is likely to be most successful if it involves the prevention of behavioural risk factors. These findings are timely, in light of the COVID-19 pandemic, and highlight potential additional benefits of addressing behavioural risk factors ahead of any future epidemics or pandemics. While the specific diseases that may be involved can only be speculated, they are likely to share at least some characteristics with diseases in this review.
Although this review has not examined the mechanisms connecting behavioural risk factors and communicable diseases, there are likely to be multiple mechanisms. Behavioural factors, such as alcohol use, smoking, obesity, and illicit drug use, are well documented to impair the immune system. For instance, smoking is known to influence both innate and adaptive immunity [ 75 ]. Impairments to the immune system can make individuals more susceptible to communicable diseases and less able to control or recover from infection, leading to worse outcomes [ 76 , 77 , 78 , 79 , 80 , 81 ]. Use of alcohol/drugs may also reduce the efficacy of treatment for communicable diseases [ 82 ]. The presence of comorbidities, such as diabetes and cardiovascular disease, in individuals with behavioural risk factors has also been implicated in the increased risk of communicable diseases [ 83 , 84 ]. However, behavioural risk factors, such as obesity, are also reported to independently influence communicable diseases, after adjusting for comorbidities [ 55 ]. Behavioural mechanisms may also be important, particularly for alcohol and drug use, which may reduce risk perception [ 78 ], interfere with the uptake of services, or lead to poorer treatment adherence [ 85 ]. Additionally, behavioural risk factors may be likely to appear in combination, for example combined alcohol use and smoking [ 86 ], to further increase influences on communicable diseases. Furthermore, behaviours associated with drug use, such as injecting drugs, have a high efficiency of transmission of communicable diseases and reinfection with communicable diseases [ 87 ]. Having a communicable disease could also lead to the presence of behavioural risk factors (e.g. alcohol may be used as a way of coping with the emotional distress of diseases such as HIV and HCV [ 88 ]). Finally, there may be social mechanisms, such as the social marginalisation of heavy drinkers that affects health service use or treatment [ 89 ], or social issues such as homelessness, incarceration and poverty, which may increase the risk of both behavioural risk factors and communicable diseases [ 90 , 91 ]. It is likely that there are multiple ways in which these different physiological, behavioural and social factors come together to affect the likelihood of transmission and severity of communicable disease, which require further investigation.
With behavioural risk factors influencing the contraction and severity of communicable diseases, their prevention is likely to play a role in addressing future communicable disease burden, potentially through improvements in the immune system, bodily functioning and risk behaviours. As the recent COVID-19 pandemic has highlighted, their prevention is also likely to impact on communicable disease burden through the potential reduction of NCDs commonly associated with behavioural risk factors, which can also alter immune system function [ 92 ] and increase the risk of communicable disease complications and death [ 10 , 93 ]. The review findings are important in understanding communicable disease risk, and timely, in light of COVID-19. They suggest that improvements in the prevention of behavioural risk factors may serve to reduce the negative impacts of future epidemics or pandemics, building resilience and helping to address the pressing need for greater investment in pandemic preparedness [ 94 ]. Indeed, COVID-19 should not only be a reminder that good communicable disease control is necessary, but that the more successful we are in addressing behavioural risk factors, the better we will also be at reducing the burden of communicable disease, including future epidemics or pandemics. The finding that both communicable diseases and NCDs share a common set of behavioural risk factors also lends support for a more holistic understanding of these two disease categories. For instance, research suggests that NCDs and communicable diseases can interact; whilst NCDs can increase the risk and severity of communicable diseases (e.g. individuals with diabetes, hypertension and respiratory illnesses are more likely to affected by COVID-19 [ 95 ]), at least some diseases previously considered NCDs are now known to have an infectious origin (e.g. HBV is a cause of heptatocellular carcinoma [ 96 ]).
Although the focus of this review is on HICs, findings will be of importance to LMICs, which often experience a much higher burden of communicable disease [ 9 ] and where, for many countries, the prevalence of behavioural risk factors is increasing [ 7 , 8 ]. Due to ageing populations, the negative impacts of globalisation, and ill-equipped health systems, these countries are also facing a rapidly growing burden of NCDs [ 97 , 98 , 99 ], which may reduce resistance to infection, increase communicable disease complications, or interfere with its treatment [ 100 , 101 ]. In the current global society, any negative effects of rising behavioural risk factors and related NCDs on communicable disease transmission have the potential to affect not only LMICs, but health and well-being globally.
Across both HICs and LMICs, behavioural risk factors and related NCDs are known to cluster in disadvantaged populations [ 97 , 102 , 103 , 104 ], with poverty contributing to behavioural risk factors and NCDs, and vice versa [ 97 ]. Disadvantaged communities are more likely, therefore, to experience dual burdens of NCDs and communicable disease, contributing to social and economic health inequalities. In the UK for instance, people living in the most disadvantaged communities have been over twice as likely to die from COVID-19 as those in the least disadvantaged areas [ 105 ]. Preventing behavioural risk factors, particularly among disadvantaged populations, is likely to play an important role in reducing future global and national health inequalities, as well as the unequal burden of future pandemics.
There are some limitations to this work. The wide-ranging nature of the research allowed for a broad view of the links between behavioural risk factors and communicable diseases. However, this did not allow for the exploration of causal pathways of specific associations. Further research exploring these pathways would aid understanding and inform prevention. The use of systematic reviews to achieve a broader range of information also meant that newer empirical research may have been missed, only more widely researched topics for which there is enough information to conduct a systematic review would have been included, and more in-depth information such as potential interactions between risk factors could not be included. With no single definitions of behavioural risk factors agreed across the literature, it was not possible to standardise the definitions of risk factors in this review, meaning that there was often variation in the definitions included in each risk factor category, hampering discussion of relationships. Low socio-economic status (SES) and other factors associated with low SES, such as poor housing, are likely to be an important element in the link between behavioural risk factors and communicable diseases, although little is currently known about the influence of low SES and associated factors. It was not possible to explore the role of low SES within the review since many of the reviews included did not explore low SES in their analyses. Many of the included studies are global syntheses, however, the relationships between behavioural risk factors and communicable diseases may vary between countries. Only papers written in English were included, meaning that research in other languages may have been missed. Finally, conclusions should be considered with publication bias in mind; papers are more likely to be published if they reveal significant effects rather than null findings [ 106 ], so those reporting that behavioural risk factors are associated with communicable diseases are more likely to be identified.
This work identified several gaps in the current systematic review literature relating to specific behavioural risk factors and common communicable diseases, including studies examining the association of physical inactivity and poor dietary habits with communicable diseases, which warrant urgent further exploration. For instance, recently published literature has indeed highlighted the important role of physical inactivity in severe COVID-19 risk [ 2 , 107 ]. Due to the study being limited to systematic reviews only, a comprehensive comparison of behavioural risk factors across different disease types could not be provided; although future reviews could provide such comparisons. However, it was noted that reviews examining the association of communicable diseases with alcohol and illicit drug use largely focused on TB, HIV and hepatitis, whereas reviews examining association with obesity largely focused on pneumonia, influenza and COVID-19 (see Supplementary file 3 ). Further research understanding the more intricate ways in which individual behavioural risk factors are linked to specific types of disease, and the mechanisms by which they are linked, would provide a valuable framework for understanding how current and future communicable diseases may affect different population groups. Finally, findings highlight an opportunity for future research to examine the efficacy of behavioural risk factor prevention efforts in reducing communicable disease burden.
Behavioural risk factors play a significant role in the risk of contracting, and having more severe outcomes from, common communicable diseases. These risk factors are largely modifiable or preventable. Prevention of communicable diseases is likely to be most successful if it involves the prevention of behavioural risk factors that are commonly associated with NCDs, particularly among disadvantaged populations. These findings are important for understanding risks associated with communicable disease, and timely, given the current COVID-19 pandemic and need for improvements in future pandemic preparedness. Addressing behavioural risk factors should be an important part of work to build resilience against any emerging and future epidemics and pandemics. Furthermore, the pandemic can offer a timely, teachable moment for the public on how improvements to general health, through addressing risk behaviours commonly associated with NCDs, may help protect them from infections like COVID-19 in the future.
Availability of data and materials
Not applicable.
Abbreviations
Acquired immune deficiency syndrome
Acute lower respiratory infections
Body mass index
Hepatitis C virus
Hepatitis D virus
High-income country
Human immunodeficiency virus
Invasive bacterial disease
Invasive pneumococcal disease
Invasive meningococcal disease
Lower-middle income country
Middle east respiratory syndrome
Non-communicable disease
Overview quality assessment questionnaire
Preferred reporting items for systematic reviews and meta-analyses
International prospective register of systematic reviews
Relative risk
Severe acute respiratory syndrome
Tuberculosis
World Health Organization and the United Nations Development Programme. Responding to non-communicable diseases during and beyond the COVID-19 pandemic. 2020. Available from: https://www.who.int/publications/i/item/WHO-2019-nCoV-Non-communicable_diseases-Evidence-2020.1 . Accessed 24 June 2021.
Google Scholar
Hamer M, Kivimäki M, Gale CR, Batty GD. Lifestyle risk factors, inflammatory mechanisms, and COVID-19 hospitalization: a community-based cohort study of 387,109 adults in UK. Brain Behav Immun. 2020;87:184–7. https://doi.org/10.1016/j.bbi.2020.05.059 .
Article CAS PubMed PubMed Central Google Scholar
Bambra C, Riordan R, Ford J, Matthews F. The COVID-19 pandemic and health inequalities. J Epidemiol Community Health. 2020;74(11):964–8. https://doi.org/10.1136/jech-2020-214401 .
Article PubMed Google Scholar
Horton R. Offline: COVID-19 is not a pandemic. Lancet. 2020;396(10255):874. https://doi.org/10.1016/S0140-6736(20)32000-6 .
World Health Organization. The Global Health Observatory. Available from: https://www.who.int/data/gho . Accessed 8 Nov 2021.
Global Burden of Disease Collaborative Network. GBD Results Tool. 2021. Available from: http://ghdx.healthdata.org/gbd-results-tool . Accessed 24 June 2021.
Uddin R, Lee E-Y, Khan SR, Tremblay MS, Khan A. Clustering of lifestyle risk factors for non-communicable diseases in 304,779 adolescents from 89 countries: a global perspective. Prev Med An Int J Devoted to Pract Theory. 2020;131:8. https://doi.org/10.1016/j.ypmed.2019.105955 .
Article Google Scholar
Ford ND, Patel SA, Narayan KMV. Obesity in low- and middle-income countries: burden, drivers, and emerging challenges. Annu Rev Public Health. 2017;38(1):145–64. https://doi.org/10.1146/annurev-publhealth-031816-044604 .
World Health Organization. Global status report on alcohol and health 2018. Geneva: World Health Organization; 2018.
Azarpazhooh MR, Morovatdar N, Avan A, Phan TG, Divani AA, Yassi N, et al. COVID-19 pandemic and burden of non-communicable diseases: an ecological study on data of 185 countries. J Stroke Cerebrovasc Dis. 2020;29(9):1–9. https://doi.org/10.1016/j.jstrokecerebrovasdis.2020.105089 .
Bloom DE, Cadarette D. Infectious disease threats in the twenty-first century: strengthening the global response. Front Immunol. 2019;10:549. https://doi.org/10.3389/fimmu.2019.00549 .
Article PubMed PubMed Central Google Scholar
Hamer M, O’Donovan G, Stamatakis E. Lifestyle risk factors, obesity and infectious disease mortality in the general population: linkage study of 97,844 adults from England and Scotland. Prev Med (Baltim). 2019;123:65–70. https://doi.org/10.1016/j.ypmed.2019.03.002 .
Baik I, Curhan GC, Rimm EB, Bendich A, Willett WC, Fawzi WW. A prospective study of age and lifestyle factors in relation to community-acquired pneumonia in US men and women. Arch Intern Med. 2000;160(20):3082–8. https://doi.org/10.1001/archinte.160.20.3082 .
Article CAS PubMed Google Scholar
Creswell J, Raviglione M, Ottmani S, Migliori GB, Uplekar M, Blanc L, et al. Tuberculosis and noncommunicable diseases: neglected links and missed opportunities. Eur Respir J. 2011;37(5):1269–82. https://doi.org/10.1183/09031936.00084310 .
Majid E, Elio R. Behavioral and dietary risk factors for noncommunicable diseases. N Engl J Med. 2013;369(10):954–64. https://doi.org/10.1056/NEJMra1203528 .
Article CAS Google Scholar
World Health Organization. Disease outbreaks. 2021. Available from: https://www.who.int/emergencies/diseases/en/ . Accessed 24 June 2021.
Wimalasena NN, Chang-Richards A, Wang KI-K, Dirks KN. Housing risk factors associated with respiratory disease: a systematic review. Int J Environ Res Public Health. 2021;18(6):2815. https://doi.org/10.3390/ijerph18062815 .
Simou E, Britton J, Leonardi-Bee J. Alcohol consumption and risk of tuberculosis: a systematic review and meta-analysis. Int J Tuberc Lung Dis. 2018;22(11):1277–85. https://doi.org/10.5588/ijtld.18.0092 .
Mohidem NA, Hashim Z, Osman M, Shaharudin R, Farrah MM, Makeswaran P. Demographic, socio-economic and behavior as risk factors of tuberculosis in Malaysia: a systematic review of the literature. Rev Environ Health. 2018;33(4):407–21. https://doi.org/10.1515/reveh-2018-0026 .
Rumbwere Dube BN, Marshall TP, Ryan RP, Omonijo M. Predictors of human immunodeficiency virus (HIV) infection in primary care among adults living in developed countries: a systematic review. Syst Rev. 2018;7(1):82. https://doi.org/10.1186/s13643-018-0744-3 .
Simou E, Britton J, Leonardi-Bee J. Alcohol and the risk of pneumonia: a systematic review and meta-analysis. BMJ Open. 2018;8(8):e022344. https://doi.org/10.1136/bmjopen-2018-022344 .
Samokhvalov AV, Irving HM, Rehm J. Alcohol consumption as a risk factor for pneumonia: a systematic review and meta-analysis. Epidemiol Infect. 2010;138(12):1789–95. https://doi.org/10.1017/S0950268810000774 .
Cruickshank HC, Jefferies JM, Clarke SC. Lifestyle risk factors for invasive pneumococcal disease: a systematic review. BMJ Open. 2014;4(6):e005224. https://doi.org/10.1136/bmjopen-2014-005224 .
Pimpin L, Drumright LN, Kruijshaar ME, Abubakar I, Rice B, Delpech V, et al. Tuberculosis and HIV co-infection in European Union and European economic area countries. Eur Respir J. 2011;38(6):1382–92. https://doi.org/10.1183/09031936.00198410 .
Zhao Y, Luo T, Tucker JD, Wong WCW. Risk factors of HIV and other sexually transmitted infections in China: a systematic review of reviews. PLoS One. 2015;10(10):e0140426. https://doi.org/10.1371/journal.pone.0140426 .
Goel A, Seguy N, Aggarwal R. Burden of hepatitis C virus infection in India: a systematic review and meta-analysis. J Gastroenterol Hepatol. 2019;34(2):321–9. https://doi.org/10.1111/jgh.14466 .
Mahmud S, Akbarzadeh V, Abu-Raddad LJ. The epidemiology of hepatitis C virus in Iran: systematic review and meta-analyses. Sci Rep. 2018;8(1):1. https://doi.org/10.1038/s41598-017-18296-9 .
Al Kanaani Z, Mahmud S, Kouyoumjian SP, Abu-Raddad LJ. The epidemiology of hepatitis C virus in Pakistan: systematic review and meta-analyses. R Soc Open Sci. 2018;5(4):180257. https://doi.org/10.1098/rsos.180257 .
Fadlalla FA, Mohamoud YA, Mumtaz GR, Abu-Raddad LJ. The epidemiology of hepatitis C virus in the Maghreb region: systematic review and meta-analyses. PLoS One. 2015;10(3):e0121873. https://doi.org/10.1371/journal.pone.0121873 .
Falagas M, Koletsi PK, Baskouta E, Rafailidis PI, Dimopoulos G, Karageorgopoulos DE. Pandemic a(H1N1) 2009 influenza: review of the southern hemisphere experience. Epidemiol Infect. 2011;139(1):27–40. https://doi.org/10.1017/S0950268810002037 .
Phung DT, Wang Z, Rutherford S, Huang C, Chu C. Body mass index and risk of pneumonia: a systematic review and meta-analysis. Obes Rev. 2013;14(10):839–57. https://doi.org/10.1111/obr.12055 .
Nie W, Zhang Y, Jee SH, Jung KJ, Li B, Xiu Q. Obesity survival paradox in pneumonia: a meta-analysis. BMC Med. 2014;12(1):61. https://doi.org/10.1186/1741-7015-12-61 .
Baskaran V, Murray RL, Hunter A, Lim WS, McKeever TM. Effect of tobacco smoking on the risk of developing community acquired pneumonia: a systematic review and meta-analysis. PLoS One. 2019;14(7):e0220204. https://doi.org/10.1371/journal.pone.0220204 .
Jafta N, Jeena PM, Barregard L, Naidoo RN. Childhood tuberculosis and exposure to indoor air pollution: a systematic review and meta-analysis. Int J Tuberc Lung Dis. 2015;19(5):596–602. https://doi.org/10.5588/ijtld.14.0686 .
Dogar OF, Pillai N, Safdar N, Shah SK, Zahid R, Siddiqi K. Second-hand smoke and the risk of tuberculosis: a systematic review and a meta-analysis. Epidemiol Infect. 2015;143(15):3158–72. https://doi.org/10.1017/S0950268815001235 .
Patra J, Bhatia M, Suraweera W, Morris SK, Patra C, Gupta PC, et al. Exposure to second-hand smoke and the risk of tuberculosis in children and adults: a systematic review and meta-analysis of 18 observational studies. PLoS Med. 2015;12(6):e1001835. https://doi.org/10.1371/journal.pmed.1001835 .
Lee C-C, Middaugh NA, Howie SRC, Ezzati M. Association of secondhand smoke exposure with pediatric invasive bacterial disease and bacterial carriage: a systematic review and meta-analysis. PLoS Med. 2010;7(12):e1000374. https://doi.org/10.1371/journal.pmed.1000374 .
Song Y, Ren F, Sun D, Wang M, Baker JS, István B, et al. Benefits of exercise on influenza or pneumonia in older adults: a systematic review. Int J Environ Res Public Health. 2020;17(8):2655. https://doi.org/10.3390/ijerph17082655 .
Article CAS PubMed Central Google Scholar
Waitt CJ, Squire SB. A systematic review of risk factors for death in adults during and after tuberculosis treatment. Int J Tuberc Lung Dis. 2011;15(7):871–85. https://doi.org/10.5588/ijtld.10.0352 .
Samuels JP, Sood A, Campbell JR, Khan FA, Johnston JC. Comorbidities and treatment outcomes in multidrug resistant tuberculosis: a systematic review and meta-analysis. Sci Rep. 2018;8(1):1–13. https://doi.org/10.1038/s41598-018-23344-z .
Rajendran M, Zaki RA, Aghamohammadi N. Contributing risk factors towards the prevalence of multidrug-resistant tuberculosis in Malaysia: a systematic review. Tuberculosis. 2020;122:1. https://doi.org/10.1016/j.tube.2020.101925 .
Azhar G. DOTS for TB relapse in India: a systematic review. Lung India. 2012;29(2):147–53. https://doi.org/10.4103/0970-2113.95320 .
Torres NMC, Rodríguez JJQ, Andrade PSP, Arriaga MB, Netto EM. Factors predictive of the success of tuberculosis treatment: a systematic review with meta-analysis. PLoS One. 2019;14(12):e0226507. https://doi.org/10.1371/journal.pone.0226507 .
Ragan EJ, Kleinman MB, Sweigart B, Gnatienko N, Parry CD, Horsburgh CR, et al. The impact of alcohol use on tuberculosis treatment outcomes: a systematic review and meta-analysis. Int J Tuberc Lung Dis. 2020;24(1):73–82. https://doi.org/10.5588/ijtld.19.0080 .
Azar MM, Springer SA, Meyer JP, Altice FL. A systematic review of the impact of alcohol use disorders on HIV treatment outcomes, adherence to antiretroviral therapy and health care utilization. Drug Alcohol Depend. 2010;112(3):178–93. https://doi.org/10.1016/j.drugalcdep.2010.06.014 .
Velloza J, Kemp CG, Aunon FM, Ramaiya MK, Creegan E, Simoni JM. Alcohol use and antiretroviral therapy non-adherence among adults living with HIV/AIDS in sub-saharan Africa: a systematic review and meta-analysis. AIDS Behav. 2020;24(6):1727–42. https://doi.org/10.1007/s10461-019-02716-0 .
Ge S, Sanchez M, Nolan M, Liu T, Savage CL. Is alcohol use associated with increased risk of developing adverse health outcomes among adults living with human immunodeficiency virus: a systematic review. J Addict Nurs. 2018;29(2):96–118. https://doi.org/10.1097/JAN.0000000000000220 .
Aisyah DN, Shallcross L, Hully AJ, O’Brien A, Hayward A. Assessing hepatitis C spontaneous clearance and understanding associated factors—a systematic review and meta-analysis. J Viral Hepat. 2018;25(6):680–98. https://doi.org/10.1111/jvh.12866 .
Sublette VA, Douglas MW, McCaffery K, George J, Perry KN. Psychological, lifestyle and social predictors of hepatitis C treatment response: a systematic review. Liver Int. 2013;33(6):894–903. https://doi.org/10.1111/liv.12138 .
Peacock A, Tran LT, Larney S, Stockings E, Santo T, Jones H, et al. All-cause and cause-specific mortality among people with regular or problematic cocaine use: a systematic review and meta-analysis. Addiction. 2021;116(4):725–42. https://doi.org/10.1111/add.15239 .
Hajarizadeh B, Cunningham EB, Valerio H, Martinello M, Law M, Janjua NZ, et al. Hepatitis C reinfection after successful antiviral treatment among people who inject drugs: a meta-analysis. J Hepatol. 2020;72(4):643–57. https://doi.org/10.1016/j.jhep.2019.11.012 .
Latham NH, Doyle JS, Palmer AY, Vanhommerig JW, Agius P, Goutzamanis S, et al. Staying hepatitis C negative: a systematic review and meta-analysis of cure and reinfection in people who inject drugs. Liver Int. 2019;39(12):2244–60. https://doi.org/10.1111/liv.14152 .
Chen HY, Shen DT, Ji DZ, Han PC, Zhang WM, Ma JF, et al. Prevalence and burden of hepatitis D virus infection in the global population: a systematic review and meta-analysis. Gut. 2019;68(3):512–21. https://doi.org/10.1136/gutjnl-2018-316601 .
Fezeu L, Julia C, Henegar A, Bitu J, Hu FB, Grobbee DE, et al. Obesity is associated with higher risk of intensive care unit admission and death in influenza a (H1N1) patients: a systematic review and meta-analysis. Obes Rev. 2011;12(8):653–9. https://doi.org/10.1111/j.1467-789X.2011.00864.x .
Mertz D, Kim TH, Johnstone J, Lam P-P, Science M, Kuster SP, et al. Populations at risk for severe or complicated influenza illness: systematic review and meta-analysis. BMJ. 2013;347(aug23 1):f5061. https://doi.org/10.1136/bmj.f5061 .
Coleman BL, Fadel SA, Fitzpatrick T, Thomas S. Risk factors for serious outcomes associated with influenza illness in high- versus low- and middle-income countries: systematic literature review and meta-analysis. Influenza Other Respir Viruses. 2018;12(1):22–9. https://doi.org/10.1111/irv.12504 .
Peres KC, Riera R, Martimbianco ALC, Ward LS, Cunha LL. Body Mass Index and Prognosis of COVID-19 Infection. A Systematic Review. Front Endocrinol (Lausanne). 2020;11:562.
de Carvalho Sales-Peres SH, de Azevedo-Silva LJ, RCS B, de Carvalho Sales-Peres M, da Silvia Pinto AC, Santiago Junior JF. Coronavirus (SARS-CoV-2) and the risk of obesity for critically illness and ICU admitted: Meta-analysis of the epidemiological evidence. Obes Res Clin Pract. 2020;14(5):389–97. https://doi.org/10.1016/j.orcp.2020.07.007 .
de Siqueira JVV, Almeida LG, Zica BO, Brum IB, Barceló A, de Siqueira Galil AG. Impact of obesity on hospitalizations and mortality, due to COVID-19: a systematic review. Obes Res Clin Pract. 2020;14(5):398–403. https://doi.org/10.1016/j.orcp.2020.07.005 .
Földi M, Farkas N, Kiss S, Zádori N, Váncsa S, Szakó L, et al. Obesity is a risk factor for developing critical condition in COVID-19 patients: a systematic review and meta-analysis. Obes Rev. 2020;21(10):e13095. https://doi.org/10.1111/obr.13095 .
Wang EY, Arrazola RA, Mathema B, Ahluwalia IB, Mase SR. The impact of smoking on tuberculosis treatment outcomes: a meta-analysis. Int J Tuberc Lung Dis. 2020;24(2):170–5. https://doi.org/10.5588/ijtld.19.0002 .
Wang M-G, Huang W-W, Wang Y, Zhang Y-X, Zhang M-M, Wu S-Q, et al. Association between tobacco smoking and drug-resistant tuberculosis. Infect Drug Resist. 2018;11:873–87. https://doi.org/10.2147/IDR.S164596 .
Farsalinos K, Barbouni A, Poulas K, Polosa R, Caponnetto P, Niaura R, et al. Current smoking, former smoking, and adverse outcome among hospitalized COVID-19 patients: a systematic review and meta-analysis. Ther Adv Chronic Dis. 2020;11:1–14. https://doi.org/10.1177/2040622320935765 .
Del Sole F, Farcomeni A, Loffredo L, Carnevale R, Menichelli D, Vicario T, et al. Features of severe COVID-19: a systematic review and meta-analysis. Eur J Clin Investig. 2020;50(10):1–7. https://doi.org/10.1111/eci.13378 .
Alqahtani JS, Oyelade T, Aldhahir AM, Alghamdi SM, Almehmadi M, Alqahtani AS, et al. Prevalence, severity and mortality associated with COPD and smoking in patients with COVID-19: a rapid systematic review and meta-analysis. PLoS One. 2020;15(5):e0233147. https://doi.org/10.1371/journal.pone.0233147 .
Gülsen A, Yigitbas BA, Uslu B, Drömann D, Kilinc O. The effect of smoking on COVID-19 symptom severity: systematic review and meta-analysis. Pulm Med. 2020;2020:7590207–11. https://doi.org/10.1155/2020/7590207 .
Zhao Q, Meng M, Kumar R, Wu Y, Huang J, Lian N, et al. The impact of COPD and smoking history on the severity of COVID-19: a systemic review and meta-analysis. J Med Virol. 2020;92(10):1915–21. https://doi.org/10.1002/jmv.25889 .
Sanchez-Ramirez DC, Mackey D. Underlying respiratory diseases, specifically COPD, and smoking are associated with severe COVID-19 outcomes: a systematic review and meta-analysis. Respir Med. 2020;171:106096. https://doi.org/10.1016/j.rmed.2020.106096 .
Sonego M, Pellegrin MC, Becker G, Lazzerini M. Risk factors for mortality from acute lower respiratory infections (ALRI) in children under five years of age in low and middle-income countries: a systematic review and meta-analysis of observational studies. PLoS One. 2015;10(1):17. https://doi.org/10.1371/journal.pone.0116380 .
Giusti A, Penco G, Pioli G. Vitamin D deficiency in HIV-infected patients: a systematic review. Nutr Diet Suppl. 2011;3:101–11. https://doi.org/10.2147/NDS.S6921 .
Metelli S, Chaimani A. Challenges in meta-analyses with observational studies. Evid Based Ment Heal. 2020;23(2):83–7 Available from: http://ebmh.bmj.com/content/23/2/83.abstract .
Popay J, Roberts H, Sowden A, Petticrew M, Arai L, Rodgers M, et al. Guidance on the conduct of narrative synthesis in systematic Reviews. A Product from the ESRC Methods Programme. Version 1. 2006. Available from: https://www.lancaster.ac.uk/media/lancaster-university/content-assets/documents/fhm/dhr/chir/NSsynthesisguidanceVersion1-April2006.pdf . Accessed 24 June 2021.
Fusar-Poli P, Radua J. Ten simple rules for conducting umbrella reviews. Evid Based Ment Health. 2018;21(3):95–100. https://doi.org/10.1136/ebmental-2018-300014 .
Pussegoda K, Turner L, Garritty C, Mayhew A, Skidmore B, Stevens A, et al. Identifying approaches for assessing methodological and reporting quality of systematic reviews: a descriptive study. Syst Rev. 2017;6(1):117. https://doi.org/10.1186/s13643-017-0507-6 .
Qiu F, Liang C-L, Liu H, Zeng Y-Q, Hou S, Huang S, et al. Impacts of cigarette smoking on immune responsiveness: up and down or upside down? Oncotarget. 2017;8(1):268–84. https://doi.org/10.18632/oncotarget.13613 .
Díaz LE, Montero A, González-Gross M, Vallejo AI, Romeo J, Marcos A. Influence of alcohol consumption on immunological status: a review. Eur J Clin Nutr. 2002;56(S3):S50–3. https://doi.org/10.1038/sj.ejcn.1601486 .
Sattar N, McInnes IB, McMurray JJV. Obesity is a risk factor for severe COVID-19 infection: multiple potential mechanisms. Circulation. 2020;142(1):4–6. https://doi.org/10.1161/CIRCULATIONAHA.120.047659 .
Andreasson S, Chikritzhs T, Dangardt F, Holder H, Naimi T, Sherk A, et al. Alcohol and society 2021: alcohol and the coronavirus pandemic: individual, societal and policy perspectives. Stockholm: Medicine, SIGHT, Movendi International & IOGT-NTO; 2021.
Trivedi GY, Saboo B. The risk factors for immune system impairment and the need for lifestyle changes. J Soc Heal Diabetes. 2020;8(01):025–8. https://doi.org/10.1055/s-0040-1715778 .
Pacifici R, Zuccaro P, Pichini S, Roset PN, Poudevida S, Farré M, et al. Modulation of the immune system in cannabis users. JAMA. 2003;289(15):1929–31. https://doi.org/10.1001/jama.289.15.1929-a .
Friedman H, Eisenstein TK. Neurological basis of drug dependence and its effects on the immune system. J Neuroimmunol. 2004;147(1-2):106–8. https://doi.org/10.1016/j.jneuroim.2003.10.022 .
Pandrea I, Happel KI, Amedee AM, Bagby GJ, Nelson S. Alcohol’s role in HIV transmission and disease progression. Alcohol Res Heal. 2010;33:203–18.
Vas P, Hopkins D, Feher M, Rubino F, Whyte M. Diabetes, obesity and COVID −19: a complex interplay. Diabetes Obes Metab. 2020;22(10):1892–6. https://doi.org/10.1111/dom.14134 .
Poirier P, Alpert MA, Fleisher LA, Thompson PD, Sugerman HJ, Burke LE, et al. Cardiovascular evaluation and management of severely obese patients undergoing surgery. Circulation. 2009;120(1):86–95. https://doi.org/10.1161/CIRCULATIONAHA.109.192575 .
Gonzalez A, Barinas J, O’Cleirigh C. Substance use: impact on adherence and HIV medical treatment. Curr HIV/AIDS Rep. 2011;8(4):223–34. https://doi.org/10.1007/s11904-011-0093-5 .
Poortinga W. The prevalence and clustering of four major lifestyle risk factors in an English adult population. Prev Med (Baltim). 2007;44(2):124–8. https://doi.org/10.1016/j.ypmed.2006.10.006 .
Maher L, Jalaludin B, Chant KG, Jayasuriya R, Sladden T, Kaldor JM, et al. Incidence and risk factors for hepatitis C seroconversion in injecting drug users in Australia. Addiction. 2006;101(10):1499–508. https://doi.org/10.1111/j.1360-0443.2006.01543.x .
Moitra E, Anderson BJ, Herman DS, Hayaki J, Pinkston MM, Kim HN, et al. Examination of using alcohol to cope, depressive symptoms, and perceived social support in persons with HIV and Hepatitis C. AIDS Care. 2020 Oct;32(10):1238–45. https://doi.org/10.1080/09540121.2020.1734177 .
Room R. Stigma, social inequality and alcohol and drug use. Drug Alcohol Rev. 2005;24(2):143–55. https://doi.org/10.1080/09595230500102434 .
Fazel S, Bains P, Doll H. Substance abuse and dependence in prisoners: a systematic review. Addiction. 2006;101(2):181–91. https://doi.org/10.1111/j.1360-0443.2006.01316.x .
Story A, Murad S, Roberts W, Verheyen M, Hayward AC. Tuberculosis in London: the importance of homelessness. Problem drug use and prison. Thorax. 2007;62(8):667–71. https://doi.org/10.1136/thx.2006.065409 .
Berbudi A, Rahmadika N, Tjahjadi AI, Ruslami R. Type 2 diabetes and its impact on the immune system. Curr Diabetes Rev. 2020;16(5):442–9. https://doi.org/10.2174/1573399815666191024085838 .
Clark A, Jit M, Warren-Gash C, Guthrie B, Wang HHX, Mercer SW, et al. Global, regional, and national estimates of the population at increased risk of severe COVID-19 due to underlying health conditions in 2020: a modelling study. Lancet Glob Heal. 2020;8(8):e1003–17. https://doi.org/10.1016/S2214-109X(20)30264-3 .
Fan VY, Jamison DT, Summers LH. Pandemic risk: how large are the expected losses? Bull World Health Organ. 2018;96(2):129–34. https://doi.org/10.2471/BLT.17.199588 .
Nikoloski Z, Alqunaibet AM, Alfawaz RA, Almudarra SS, Herbst CH, El-Saharty S, et al. Covid-19 and non-communicable diseases: evidence from a systematic literature review. BMC Public Health. 2021;21(1):1068. Available from. https://doi.org/10.1186/s12889-021-11116-w .
Xie Y, Hepatitis B. Virus-associated hepatocellular carcinoma. Adv Exp Med Biol. 2017;1018:11–21. https://doi.org/10.1007/978-981-10-5765-6_2 .
World Health Organization. Global status report on noncommunicable diseases 2010. Geneva: World Health Organization; 2011.
Islam SMS, Purnat TD, Phuong NTA, Mwingira U, Schacht K, Fröschl G. Non-Communicable Diseases (NCDs) in developing countries: a symposium report. Global Health. 2014;10:81.
Kostova D, Chaloupka FJ, Frieden TR, Henning K, Paul J, Osewe PL, et al. Noncommunicable disease risk factors in developing countries: policy perspectives. Prev Med (Baltim). 2017;105:S1–3. https://doi.org/10.1016/j.ypmed.2017.09.027 .
Kostova D, Husain M, Sugerman D, Hong Y, Saraiya M, Keltz J, et al. Synergies between communicable and noncommunicable disease programs to enhance global health security. Emerg Infect Dis J. 2017;23(13):S40–467. https://doi.org/10.3201/eid2313.170581 .
Remais JV, Zeng G, Li G, Tian L, Engelgau MM. Convergence of non-communicable and infectious diseases in low- and middle-income countries. Int J Epidemiol. 2013;42(1):221–7. https://doi.org/10.1093/ije/dys135 .
Marmot M, Bell R. Social determinants and non-communicable diseases: time for integrated action. BMJ. 2019;364:10–2. https://doi.org/10.1136/bmj.l251 .
Hosseinpoor AR, Bergen N, Kunst A, Harper S, Guthold R, Rekve D, et al. Socioeconomic inequalities in risk factors for non communicable diseases in low-income and middle-income countries: results from the world health survey. BMC Public Health. 2012;12(1):912. https://doi.org/10.1186/1471-2458-12-912 .
Allen L, Williams J, Townsend N, Mikkelsen B, Roberts N, Foster C, et al. Socioeconomic status and non-communicable disease behavioural risk factors in low-income and lower-middle-income countries: a systematic review. Lancet Glob Heal. 2017;5(3):e277–89. https://doi.org/10.1016/S2214-109X(17)30058-X .
Caul S. Deaths involving COVID-19 by local area and socioeconomic deprivation : deaths occurring between 1 March and 31 July 2020. 2020. Available from: https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/deaths/bulletins/deathsinvolvingcovid19bylocalareasanddeprivation/deathsoccurringbetween1marchand31july2020 . Accessed 24 June 2021.
Knobloch K, Yoon U, Vogt PM. Preferred reporting items for systematic reviews and meta-analyses (PRISMA) statement and publication bias. J Cranio-Maxillofacial Surg. 2011;39(2):91–2. https://doi.org/10.1016/j.jcms.2010.11.001 .
Sallis R, Young DR, Tartof SY, Sallis JF, Sall J, Li Q, et al. Physical inactivity is associated with a higher risk for severe COVID-19 outcomes: a study in 48 440 adult patients. Br J Sports Med. 2021;0:1–8.
Download references
Acknowledgements
We would like to thank Rebecca Hill and Nel Griffith, Public Health Wales, for their assistance in reviewing this manuscript.
This project was funded by Public Health Wales.
Author information
Authors and affiliations.
Policy and International Health, World Health Organization Collaborating Centre on Investment for Health and Well-being, Public Health Wales, Wrexham, UK
Sara Wood, Natasha Judd, Mark A. Bellis & Karen Hughes
Public Health Collaborating Unit, School of Medical and Health Sciences, Bangor University, Wrexham, UK
Sophie E. Harrison, Natasha Judd, Mark A. Bellis & Karen Hughes
Institute for Applied Human Physiology, School of Human and Behavioural Sciences, Bangor University, Bangor, UK
Sophie E. Harrison
Health Protection and Screening Services, Public Health Wales, Cardiff, UK
Andrew Jones
You can also search for this author in PubMed Google Scholar
Contributions
MB and KH contributed to the conception, design and direction of the project. SW managed the project with SH and NJ. NJ and SH undertook the literature searches and conducted the initial data screening. NJ, SH and SW conducted the full text screening, data extraction and quality assessment. SH synthesised the findings. AJ and all other authors contributed to the manuscript write up / revisions. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Mark A. Bellis .
Ethics declarations
Ethics approval and consent to participate, consent for publication, competing interests.
The authors declare that they have no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1., additional file 2., additional file 3., rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Wood, S., Harrison, S.E., Judd, N. et al. The impact of behavioural risk factors on communicable diseases: a systematic review of reviews. BMC Public Health 21 , 2110 (2021). https://doi.org/10.1186/s12889-021-12148-y
Download citation
Received : 07 October 2021
Accepted : 22 October 2021
Published : 17 November 2021
DOI : https://doi.org/10.1186/s12889-021-12148-y
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Infectious disease
- Risk factors
BMC Public Health
ISSN: 1471-2458
- Submission enquiries: [email protected]
- General enquiries: [email protected]
- Open access
- Published: 08 March 2024
Mid-level managers’ perspectives on implementing isoniazid preventive therapy for people living with HIV in Ugandan health districts: a qualitative study
- Canice Christian 1 ,
- Elijah Kakande 2 ,
- Violah Nahurira 2 ,
- Cecilia Akatukwasa 2 ,
- Fredrick Atwine 2 ,
- Robert Bakanoma 2 ,
- Harriet Itiakorit 2 ,
- Asiphas Owaraganise 2 ,
- William DiIeso 3 ,
- Derek Rast 3 ,
- Jane Kabami 2 ,
- Jason Johnson Peretz 1 ,
- Starley B. Shade 1 ,
- Moses R. Kamya 4 ,
- Diane V. Havlir 1 ,
- Gabriel Chamie 1 &
- Carol S. Camlin 1
BMC Health Services Research volume 24 , Article number: 313 ( 2024 ) Cite this article
113 Accesses
10 Altmetric
Metrics details
Isoniazid preventive therapy (IPT) works to prevent tuberculosis (TB) among people living with HIV (PLHIV), but uptake remains low in Sub-Saharan Africa. In this analysis, we sought to identify barriers mid-level managers face in scaling IPT in Uganda and the mechanisms by which the SEARCH-IPT trial intervention influenced their abilities to increase IPT uptake.
The SEARCH-IPT study was a cluster randomized trial conducted from 2017–2021. The SEARCH-IPT intervention created collaborative groups of district health managers, facilitated by local HIV and TB experts, and provided leadership and management training over 3-years to increase IPT uptake in Uganda. In this qualitative study we analyzed transcripts of annual Focus Group Discussions and Key Informant Interviews, from a subset of SEARCH-IPT participants from intervention and control groups, and participant observation field notes. We conducted the analysis using inductive and deductive coding (with a priori codes and those derived from analysis) and a framework approach for data synthesis.
When discussing factors that enabled positive outcomes, intervention managers described feeling ownership over interventions, supported by the leadership and management training they received in the SEARCH-IPT study, and the importance of collaboration between districts facilitated by the intervention. In contrast, when discussing factors that impeded their ability to make changes, intervention and control managers described external funders setting agendas, lack of collaboration in meetings that operated with more of a ‘top-down’ approach, inadequate supplies and staffing, and lack of motivation among frontline providers. Intervention group managers mentioned redistribution of available stock within districts as well as between districts, reflecting efforts of the SEARCH-IPT intervention to promote between-district collaboration, whereas control group managers mentioned redistribution within their districts to maximize the use of available IPT stock.
Conclusions
In Uganda, mid-level managers’ perceptions of barriers to scaling IPT included limited power to set agendas and control over funding, inadequate resources, lack of motivation of frontline providers, and lack of political prioritization. We found that the SEARCH-IPT intervention supported managers to design and implement strategies to improve IPT uptake and collaborate between districts. This may have contributed to the overall intervention effect in increasing the uptake of IPT among PLHIV compared to standard practice.
Trial registration
ClinicalTrials.gov, NCT03315962 , Registered 20 October 2017.
Peer Review reports
Introduction
Tuberculosis (TB) is a leading cause of death among people living with HIV (PLHIV) globally [ 1 ]. Isoniazid Preventive Therapy (IPT), a form of TB preventive treatment (TPT), reduces TB cases and death, even among PLHIV on antiretroviral therapy (ART) with suppressed HIV viral load [ 2 , 3 ]. However, IPT uptake has remained suboptimal in Sub-Saharan Africa and barriers, including supply issues and knowledge gaps, persist [ 4 , 5 ].
Multiple studies have focused on and identified provider and patient-level barriers to IPT uptake among PLHIV. Provider-level barriers have included staff shortages, lack of policy-maker support, knowledge gaps, communication challenges with patients, and negative attitude/fears surrounding IPT efficacy, side effects and drug resistance [ 6 , 7 , 8 , 9 ]. For example, frontline providers in Eritrea described barriers to IPT for PLHIV, including knowledge gaps among providers, particularly around fear of adverse effects of IPT, and inadequate systems for laboratory testing [ 10 ]. Patient-level barriers have included knowledge gaps about the benefits of IPT, fear of side effects, pill burden, HIV/TB related stigma, socio-demographic characteristics (i.e. age, educational level, and sex), lack of motivation, and financial burden, including transportation costs and lost time at work due to time spent in clinic [ 6 , 9 ]. A study in Kampala, Uganda similarly identified insufficient understanding of IPT and fear of side effects as patient-level barriers to IPT [ 11 ]. Less is known about district level health system and mid-level management barriers to scaling IPT for PLHIV.
The SEARCH-IPT cluster randomized trial intervened at the level of district health officers: mid-level managers in the health system of Uganda. The SEARCH-IPT intervention included multiple components including (1) bi-annual collaborative meetings with district managers to address challenges to scaling IPT; (2) district-level data feedback at each meeting; (3) leadership and management training for mid-level health managers annually; and (4) an SMS platform to ease communication over a 3-year study period. Mid-level managers in the health system in Uganda oversee health care delivery in their districts. These mid-level managers bridge the gap between Ministry of Health (MoH) guidelines and practical implementation at the district level. Mid-level managers have been shown to play an important role in implementing evidence-based practices globally [ 12 ]. The SEARCH-IPT trial (the primary results of which have been previously published) compared an intervention among mid-level managers to standard practice across multiple regions of Uganda. The SEARCH-IPT trial found that the mid-level manager intervention led to an increase in the number of patients started on IPT compared to standard practice, after taking into account national secular trends that influenced IPT implementation [ 13 ].
In this analysis, we sought to identify and describe the mechanisms by which the SEARCH-IPT intervention influenced the abilities of mid-level managers to increase IPT uptake in their districts. We also sought to describe shared barriers faced by both intervention and control managers that were not addressed by the SEARCH-IPT intervention.
Study design
The SEARCH-IPT study was a cluster randomized trial, where the unit of randomization was a cluster of districts in Uganda. A cluster randomized trial design was used because the intervention was provided to groups of managers, with one district health manager enrolled per district. The primary results of the trial and further description of the intervention have been published elsewhere [ 13 ]. In brief, we enrolled mid-level health managers beginning in November 2017 and follow-up ended in September 2021. The primary outcome of interest was the initiation of IPT among adults with HIV. The intervention was based on the PRECEDE model for behavioral change [ 14 ]. Predisposing and reinforcing factors of the intervention were informed by the PRECEDE model for health promotion strategies and included collaborative groups where leadership and management skills were taught through a ‘Mini-MBA’ course over a 3-year period. The trial intervention’s leadership and management training curriculum provided several tools, taught in interactive sessions with coaching by international business professionals and Uganda HIV/TB expert clinicians. These tools were designed to identify challenges managers faced and to develop strategies to overcome these challenges and accomplish their goals. During the meetings, managers were trained on tools for setting short- and long-term objectives and key results (OKRs) which allowed them to set goals and monitor progress and discuss their goals as a group. Study staff shared data on IPT uptake, and other district-level TB metrics, throughout the follow-up period to enable the tracking of progress overtime and comparisons between groups. Mini-collaboratives met every 6-months, with new leadership and management training material taught annually and reinforced at intervening collaborative meetings.
Data for this analysis were derived from transcripts of annual Focus Group Discussions (FGDs), Key Informant Interviews (KIIs), and participant observation field notes. FGDs and KIIs were conducted annually during the SEARCH-IPT intervention, starting at baseline, through three years of follow-up. Mid-level managers in intervention districts participated in FGDs, whereas a sample of managers in control districts participated in KIIs. KIIs were conducted among control districts instead of FGDs to avoid the creation of collaborative groups (similar to the intervention) among control managers. Study staff conducted participant observations at the majority of mini-collaborative meetings during the SEARCH-IPT intervention. There was no patient or public involvement in the design of this study.
Study population
The study population included all mid-level managers (district health officers and district TB supervisors) who participated in the SEARCH-IPT trial. Details on sample selection in the SEARCH-IPT trial have been published elsewhere [ 13 ]. Managers from 25 districts in the Southwest (12 control and 13 intervention districts), 23 districts in the East (11 control and 12 intervention districts), and 34 districts in the East Central (16 control and 18 intervention districts) enrolled. All FGDs included two cadres of mid-level managers from the SEARCH-IPT trial (district health officers [lead manager] and district-level TB supervisors [who report to the district health officer]), except the baseline FGDs in the Eastern region, when a separate FGD was conducted among district health officers and district TB supervisors due to the number of FGD participants. Study staff conducted participant observations at 12 meetings throughout the 3-year follow-up in the southwestern and eastern regions.
Data collection
FGDs and KIIs were conducted annually. At the FGDs, the number of participants ranged from 7 to 12. A team of trained qualitative researchers collected the data. The qualitative research team was trained on the study material at the beginning of the study and a second refresher training occurred midway through the study. Researchers conducted all FGDs and KIIs in English. All meetings where participant observations occurred were conducted in English; field notes were taken to capture discussions. FGDs and KIIs were audio recorded and transcribed verbatim for analysis.
Topic areas of the FGDs included general feedback on the mini-collaborative meetings, how mini-collaborative meetings compared to other meetings supervisors had attended, the most interesting and challenging components of the meetings and cross-district collaboration, the effects of seeing IPT and TB data compared between districts, challenges with IPT delivery and motivations for making change in their districts. KIIs consisted of semi-structured interviews using open-ended questions. Topics covered in the KIIs included discussion of the burden of TB in their districts, standard TB control practices, challenges related to IPT delivery, changes to TB prevention policy, and motivations for change in their district. Focus groups lasted between 71 – 147 min (average = 111 min) and the KIIs lasted 17 – 76 min (average = 37 min); in the results below, individual participants in FGDs are denoted (‘P1’, ‘P2’, etc.). Study staff developed participant observation field notes from attending and observing mini-collaborative meetings, including notes on general observations of the conduct of the meeting, interactions between participants and study staff, as well as some direct quotes from the participants.
Data analysis
Dedoose qualitative software was used to analyze data from FGDs, KIIs, and participant observation reports [ 15 ]. A framework analysis approach, which falls within the qualitative research tradition of thematic analysis, was used for this study [ 16 ]. We used a hybrid approach to code the data, involving both a priori codes that were informed by theory, and focused inductive coding, in which the categories of meaning were derived directly from review of the empirical data. This hybrid approach to coding included the use of established theories for behavior change as well as allowed for the data to speak for itself which allowed for a comprehensive, adaptive approach to analyzing the data. For the development of the initial codes, the first author conducted focused inductive coding on the sample of transcripts (approximately 20% of the data), developed the initial coding framework, then applied the framework to the remainder of the data [ 17 , 18 , 19 ]. The a priori codes were informed by a set of theories, including diffusion of innovation theory [ 20 ] and social learning theory [ 21 ]. Following the inductive coding phase, the first author and last authors developed an analytical framework based on the initial set of transcripts, the sample of data, and categories of grouped codes. The first author then applied the codes to the remaining transcript data using the identified codes and categories. The first and last author developed framework tables for reducing and synthesizing data, as described by Gale (2013), and with subsequent interpretation, including across-case analysis to identify the emergent themes presented here [ 22 ].
Overall, 163 mid-level managers were enrolled from 82 districts, representing 61% of the districts in Uganda. We conducted a total of 6 FGDs and 23 KIIs over the 3-year study period. Four overarching themes emerged from the data when mid-level managers discussed factors that either enabled or impeded them from making positive changes in their districts: agenda setting, collaboration, availability of resources, and motivations (Table 1 ). When discussing factors that enabled positive outcomes, intervention managers described feeling ownership over interventions, supported by the leadership and management training they had received, and the importance of collaboration. In contrast, when discussing factors that impeded their ability to make changes, intervention and control managers described external funders setting agendas, lack of collaboration in some meetings, inadequate supplies and staffing, and lack of motivation among frontline providers. Quotes and elaboration of these themes are below.
Agenda Setting
Managers feeling of ownership over local strategies to promote ipt.
During all FGDs with managers in the intervention group (6 of 6 FGDs), managers mentioned the feeling of ownership over local strategies to promote IPT as motivation for scaling IPT in their districts. Many of these local strategies were developed by managers during collaborative meetings as part of the trial intervention. This manifested in a sense of independence in deciding how change was enacted in one’s district to realize a goal: in this case, the scaling of IPT.
P8: To me I can say that it is motivational because you can identify the problem yourself. It motivates you to actually look at the problem yourself, and put in place strategies. P7: And you take the charge, you become responsible, you feel it is yours. – FGD at 2 Year Follow-up with trial intervention district health officers in the Southwest
Managers discussed the expected long-term sustainability of the SEARCH-IPT intervention after trial completion, in part because the intervention promoted the use of available resources and the mid-level managers’ self-motivation to take ownership of local strategies they developed to promote IPT. For example, an intervention manager in the East said, “This has made us use the resources that we have, which means if [the SEARCH-IPT study] is not to be there, the implementation with IPT would go on.”
Power to set agendas impeded by external funders and their implementing partners
However, managers also felt that the role of external funders and implementing partners counteracted their sense of control and ownership. During 4 of 6 FGDs (67%) and 11 of 23 KIIs (48%), managers discussed the roles that implementing partners play in scaling IPT in their districts, including implementing strategies to improve health, allocating funding, and establishing trainings. This finding contrasted with the intervention managers' discussion of the benefits of independence in deciding how change is enacted in one’s district to realize the goal of scaling IPT. Intervention and control managers described frustration with implementing partners setting the agenda and determining what steps to prioritize to improve IPT implementation, particularly concerning funding allocation and local intervention strategies.
Some [Implementing Partners] come to support activities, say for TB programs, when as a district, you have other areas that need support because at the district, you know your problems more than these [Implementing Partners] do. But when they come at the district, they tell us that they are going to support this and this program, but we tell them ‘please, me I want support in this and this’. - FGD at 3 Year Follow-up with trial intervention district health officers and district TB supervisors in Southwest
An intervention manager argued that managers should not always be dependent on the implementing partners: “If you have issues in your house, you do not expect another man to keep coming to solve them; implementing partners should not be depended on.” – Participant observation field note at the trial intervention baseline meeting in Southwest Uganda.
Intervention managers also reported challenges associated with losing funding for a particular project or support for activities and how that made it difficult to plan and execute TB prevention and TB treatment.
You find that when the [Implementing Partner] moves, they move with the program or projects... They tell you that in the next two months, they will not be supporting you in this program because the donors have withdrawn the money and the district does not have the money to take over the program that this [Implementing Partner] has been managing and you find yourself in a mix. – FGD at 2 Year Follow-up with trial intervention district health officers in Southwest
Collaboration
Collaborating with other districts enabled sharing of best practices.
Managers discussed ways in which the intervention helped them to cope with or devise strategies to address challenges. Intervention managers mentioned collaboration with other districts as an enabler to IPT uptake in all FGDs (6 of 6, 100%). This included the discussion of how mini-collaborative meetings in the SEARCH-IPT trial helped with troubleshooting challenges and developing implementation strategies to address IPT uptake.
You find that your colleagues share best experiences of how they have carried out an activity… It motivates you to say ‘I can also do it.’ If a certain district can perform to this level, then I can also improve. - FGD at 2 Year Follow-up with trial intervention district health officers in Southwest
Positive effect of the pressure of social comparison and recognition
Intervention managers discussed being motivated to improve IPT uptake in their district by comparisons to other districts during the study intervention. During the mini-collaborative meetings, study staff presented dashboards to each district that included quarterly data from the Ministry of Health on TB cases and the number of people with HIV started on IPT. Researchers tracked progress over time and presented an anonymized summary of progress to the group during the meeting, which enabled each district to see how they compared to other districts without sharing which districts fell on the ranking. A manager in an intervention district in the Southwest said, “If you are there alone, you may think that you are doing very well or you may think that this is not important. But after sitting as a team, you see how [a different district] is doing and you realize that ‘I am sleeping.’”.
Similarly, control managers discussed wanting feedback to be able to make these comparisons so they could better understand how other districts are faring in the scaling of IPT. A manager in a control district in the Southwest said, “I would like to have frequent feedback, so that we can be able to know how we stand, because internally I might be thinking that I am doing well in my district when actually my neighbors are doing better, and so eventually I could learn from them.”
Both intervention and control managers reported being motivated by accountability and recognition as a key driver to scaling IPT. For example, a manager from an intervention district in the Southwest said, “There is a way you can feel motivated, and you will say, yes I can continue doing this because you are being appreciated; your achievement is being noticed somewhere.”
Collaboration within districts between cadres of mid-level managers (district health officers and district TB supervisors) enabled scaling of IPT
One positive effect of the trial intervention was improved collaboration and alignment of priorities between district health officers (i.e., lead manager in each district) and district TB supervisors (i.e., TB-specific managers that report to lead managers), supported by the leadership and management training provided in the SEARCH-IPT intervention. This improved within-district collaboration and generated buy-in and support from the district health officer for TB-specific manager activities. Participants discussed the importance of this support, resulting in greater ease in addressing inadequate IPT uptake.
For example, I am a [district TB supervisor]. However, the people that I supervise, I do not have absolute power or authority towards them and say ‘you have not done the work’. But… when a big drum like the [district health officer] talks about something people tend to pick it up... Now that would also give us power as the [district TB supervisors]; you also stand firm because people will hear you because of your boss. - FGD at 1 Year Follow-up with trial intervention district TB supervisors in East
Control managers also mentioned that the involvement of the district health officer in their district may help make changes in their district. A manager in the East said, “I feel if the [district health officer] is involved much, the other frontline health workers will be taking my words a bit more seriously.”
Ministry of Health meetings are top-down and did not facilitate collaboration or district input
Additionally, intervention managers discussed the benefits of collaborating with neighboring districts and taking leadership roles at the mini-collaborative meetings. Participants compared the intervention meetings to meetings held by the Ministry of Health and other entities where they perceived meetings to be more directive or “top-down”. An intervention manager in the Southwest said, “[Ministry of Health meetings], it is do this, do this; it is like a directive. But here [at the collaborative meetings] we look at what had been done and we suggest on how to improve, on how to go and implement.”
Availability of resources
Inadequate inh stocks and inconsistent supplies was a barrier to ipt uptake.
Managers in intervention and control districts frequently mentioned inadequate resources as barriers to IPT uptake, including insufficient drug stock. For example, an intervention manager in the Southwest said, “ The truth is that we created demand. But over time, the supplies we had for adults were nowhere to be seen.” There was also a discussion about how inadequate stock not only interrupts the prescription of IPT in the near term but also affects the long-term uptake of IPT. When frontline providers remove IPT from regular prescribing practices due to frequent stockouts, they are slow to prescribe it when IPT does become available because it is not what they have been doing routinely.
If you really know that this [IPT prescribing] is ‘what I am supposed to do’ and you are not interrupted by [INH] stock outs… but when there is a stock out, people [front line providers] again forget that ‘this is what I am supposed to do routinely’. Then the medicine[s] come, [but] people again relax and they are not taking it as part of what they are supposed to do. So, that makes it difficult. - FGD at 1 Year Follow-up with trial intervention district TB supervisors in East
There were also discussions of challenges with requesting stock from Uganda’s centralized National Medical Stores (NMS) as a barrier to IPT uptake. Clinic staff may not request stock because they are used to a ‘push’ system for certain drugs where the NMS allots drugs to clinics without clinics placing orders. However, the system for IPT is different, as it requires the clinic to order medication (i.e., a “pull” system). Managers suggested training on proper ordering may be beneficial to mitigating this barrier.
Delivery of [IPT] depends on facilities because it’s a ‘pull’ system with TB drugs and [IPT]. If a facility doesn’t make an order in two months, it will not get the supply… This has come maybe from knowledge gap in ordering. And some facilities are used to the ‘push’ system, like essential drugs, and that’s where the problem is. - KII at 2 Year Follow-up with a control TB-specific Manger in East
Intervention and control managers also mentioned inadequate funding limited their ability to access clinics due to a lack of transportation and lack of fuel. One intervention manager in the Southwest said, “Initially we had some programs that were giving out motorcycles, but then the challenge was that when they give you a motorcycle, then fuel becomes an issue.”
Intervention and control managers also perceived insufficient staffing as a barrier to IPT. One manager in a control district in the East said, “ the challenge in handling TB/HIV is really understaffing.”
Collaboration within districts and with other districts to redistribute available supplies
Intervention and control group managers discussed resourcefulness in redistributing IPT stock to maximize the use of available stock in their district and region. Intervention group managers mentioned redistribution within districts (i.e., between health centers) as well as between districts, reflecting efforts of the SEARCH-IPT trial intervention to promote between-district collaboration, whereas control group managers mentioned redistribution within their districts.
We also discovered that some facilities with [IPT] were over stocked and this made us to do some re-distribution and take [IPT] to facilities where the consumption was high. – FGD at 1 Year Follow-up with trial intervention district health officers in East
Frontline provider turnover and knowledge gaps was a barrier for scaling IPT
Another perceived resource-related barrier to IPT uptake included gaps in frontline providers’ knowledge of IPT. This included providers lacking confidence in prescribing IPT due to inadequate training or supervision. For example, a control manager in the East said, “Some health workers didn’t have the knowledge [of IPT] so they would even fear to initiate clients for fear of the side effects.”
Managers also perceived the turnover of frontline providers, resulting in new replacement frontline providers with inadequate training in prescribing IPT, as a barrier.
The main challenges we have are experience in the sites where IPT is given, for instance you go to such a site and mentor a number of staff who are working in that clinic, and maybe two, three months down the road, transfers happen and a particular provider has been moved to another facility, and most times you find it is from high to low volume site, meaning that the capacity we will have built in that person goes to another site that is actually not offering this service. So, we tend to have issues of knowledge gaps created by transfers, whereby the trained person has been moved, and the one coming in is not well oriented, meaning that we have to continuously do mentorships in such clinics if we are to keep the standards. – KII at 2 Year Follow-up with control district TB supervisors in Southwest
Managers motivated by improving the health of their constituents
Many intervention and control managers reported being motivated by positive health outcomes in their districts. Participants discussed seeing improvements in patients’ health as a motivation to work hard in their positions. A manager in an intervention district in the Southwest said, “They come to you in pain, they are crying but by the time they leave you, they are smiling and you see their quality of life improving day by day. That is good enough to make you say that I can even take an extra mile to do this other thing.”
Lack of motivation of frontline providers was a barrier to IPT uptake
In contrast, participants perceived frontline providers’ negative attitudes as a challenge in scaling IPT. Managers said frontline providers are reluctant to be close to contacts of known TB cases and those who may be eligible for IPT due to fear of being infected with TB. A manager from an intervention district in the East said, “ The issues of attitude in case the frontline health workers, [when they] hear about TB, they may not even wish to go near the patient because they think they should first have a mask before they treat, so even if we say this is a TB contact, they even want to disassociate themselves with the contact. ”
Lack of political leadership and prioritization of IPT scale up
Lastly, another motivational barrier was a perceived lack of political leadership to scaling IPT. Intervention and control managers discussed the importance of galvanizing politicians, community leaders, and clinic-in-charges to scale IPT. They described the lack of political will as demotivating in their work to improve health outcomes because of feeling unsupported.
If you have supervisor, in this case I am talking about the political leadership of the local government and they are blinded, they cannot hear. Even if you play the best tune, they cannot dance to it because they are deaf and blind, they do not see what you are seeing and yet they are your supervisors. - FGD at 1 Year Follow-up with trial intervention district health officers in Southwest
Both intervention and control managers mentioned politics influencing the uptake of IPT, including agenda setting and political will for the scaling of IPT. One control manager in the East said, “We have a lot of political interference and that is quite discouraging. You want to do things in a certain way and without any reasonable reason, somebody who doesn’t even know what he is talking about wants you to do things just to suit his political interests.”
This qualitative study provides insights into the perceptions of mid-level managers in the Ugandan health system on the barriers and facilitators to increasing IPT uptake. This study also highlights the mechanisms by which the SEARCH-IPT trial’s intervention may have impacted IPT uptake among PLHIV (the trial’s primary outcome), by comparing perspectives of intervention to control managers who participated in the trial. We found that the SEARCH-IPT intervention, which included leadership and management training, allowed managers to design and implement strategies to improve IPT uptake in their districts, in contrast to the ‘top-down’ approach perceived by managers to be typical of external funders and implementing partners. This leadership and management training may have changed knowledge and behavior among the mid-level managers, similar to other studies that have used the PRECEDE model to improve knowledge and change health outcomes [ 23 ]. Other features of the SEARCH-IPT intervention that enabled IPT uptake included collaboration between districts and the positive impact of accountability through comparing performance with other districts at the meetings. In addition, resourcefulness through the strategic redistribution of resources was employed by both intervention and control managers to scale IPT while dealing with inadequate supplies, though intervention managers relied on the cross-district collaboratives created by the SEARCH-IPT intervention, rather than just within-district redistribution as reported by control managers, to address this challenge.
Health managers’ ownership of locally generated implementation strategies was a motivational enabler to scaling IPT, in contrast to a top-down approach of agenda setting. While the evidence of the effects of decentralization on health systems is mixed [ 24 , 25 ], strengthening district governance and decision-making have shown promise [ 26 , 27 , 28 ]. In Uganda, research has demonstrated that mid-level health managers have felt limited in their abilities to make changes [ 29 ]. In contrast, the SEARCH-IPT intervention allowed managers to have greater ownership and creativity in developing local strategies to promote IPT, supported by leadership and management training, by using available resources to maximize uptake. Managers discussed that this approach was likely to have lasting effects and potential sustainability, compared to “top down” strategies.
Intervention managers were also motivated by accountability and comparisons between districts at the collaborative meetings. This is consistent with findings from other studies which focus on decentralization and the shifting of accountability and monitoring progress toward mid-level managers [ 27 ]. The Community and District-management Empowerment for Scale-up (CODES) study in Uganda focused on aiding districts in identifying health system bottlenecks for child health. The CODES intervention provided data to managers in participating districts to better monitor and address health system challenges and showed positive effects of the intervention on child health outcomes [ 27 ]. Accountability and monitoring of data were likely to be key factors in the success of the SEARCH-IPT and CODES studies in Uganda [ 13 , 27 ].
Managers frequently mentioned the lack of sufficient supplies, including IPT stock, funding for transportation, and inadequate staffing as barriers to scaling IPT in Uganda. These findings are consistent with other initiatives focused on improving district management of health interventions in Uganda [ 30 ]. Despite implementing partners providing funding and resources, the managers described frustration in not having control over which implementation strategies were funded. In contrast, the SEARCH-IPT study approach focused on shifting decision-making power to the district managers who were able to develop their own strategies to reach their goals, supported by management training and tools. Managers discussed being aware of deficits in resources available in their districts and felt that having more input into spending would help in addressing these gaps. In addition, the SEARCH-IPT intervention allowed for collaboration among managers from other neighboring districts, aiding managers in identifying solutions to shared challenges, and within shared contexts, among peers.
This study has strengths and limitations. Strengths of this study include the cluster randomized design, breadth of managers enrolled from the majority of districts in Uganda, and duration of follow-up of 3 years. For limitations, this study did not involve stakeholders outside district health officers or district TB supervisors, such as decision-makers from implementing partners, frontline providers or other members of the Ministry of Health outside of mid-level managers. However, we collected data from district health officers and district TB supervisors over three years, providing a breadth of experience working in this space. Additionally, while this study included managers across three regions of Uganda, the results may not be generalizable to other regions in Uganda, or other national health systems. Additionally, there may be limited generalizability of the study results due to the purposeful nature of this qualitative analysis. However, the perspectives of mid-level managers have not been fully captured when evaluating barriers and enablers to IPT in the past [ 6 ] and this study is among the first to provide insight into their experiences.
Our findings suggest that involving mid-level health managers in decision-making, supported by leadership and management training, may allow managers to have a greater sense of ownership in achieving goals in their districts. Creating a space for regional collaboration and comparison may galvanize managers to improve outcomes and allow for sharing of successful interventions.
In Uganda, mid-level managers’ perceptions of barriers to scaling IPT include limited power to set agendas and control funding, inadequate resources, lack of motivation of frontline providers, and lack of political prioritization. We found that the SEARCH-IPT intervention, composed of leadership and management training and inter-district collaborative groups, enabled managers to design and implement strategies to improve IPT uptake, which may have contributed to the overall positive intervention effect in increasing the uptake of IPT among people with HIV.
Availability of data and materials
Data will be made available upon request to corresponding author.
World Health Organization. Global tuberculosis report 2022. Geneva; Licence: CC BY-NC-SA 3.0 IGO. Geneva; 2022.
Ayele HT, Mourik MS, Debray TP, Bonten MJ. Isoniazid prophylactic therapy for the prevention of tuberculosis in HIV infected adults: a systematic review and meta-analysis of randomized trials. PLoS One. 2015;10(11):e0142290.
Article PubMed PubMed Central Google Scholar
Ross JM, Badje A, Rangaka MX, Walker AS, Shapiro AE, Thomas KK, et al. Isoniazid preventive therapy plus antiretroviral therapy for the prevention of tuberculosis: a systematic review and meta-analysis of individual participant data. Lancet HIV. 2021;8(1):e8–15.
Article CAS PubMed PubMed Central Google Scholar
World Health Organization (WHO): Global tuberculosis report. Geneva. Licence: CC BY-NC-SA 3.0 IGO.2020.
Zhu J, Lyatuu G, Sudfeld CR, Kiravu A, Sando D, Machumi L, et al. Re-evaluating the health impact and cost-effectiveness of tuberculosis preventive treatment for modern HIV cohorts on antiretroviral therapy: a modelling analysis using data from Tanzania. Lancet Glob Health. 2022;10(11):e1646–54.
Müller P, Velez LL. Mixed methods systematic review and metasummary about barriers and facilitators for the implementation of cotrimoxazole and isoniazid—Preventive therapies for people living with HIV. PLoS ONE. 2022;17(3):e0251612.
Lai J, Dememew Z, Jerene D, Abashawl A, Feleke B, Teklu AM, et al. Provider barriers to the uptake of isoniazid preventive therapy among people living with HIV in Ethiopia. Int J Tuberc Lung Dis. 2019;23(3):371–7.
Article CAS PubMed Google Scholar
Elvis Omondi Achach W, Martin A, Ejemai E, Latifat I. Factors affecting the acceptability of isoniazid preventive therapy among healthcare providers in selected HIV clinics in Nairobi County, Kenya: a qualitative study. BMJ Open. 2018;8(12):e024286.
Article Google Scholar
Komba FF, Frumence G. Facility and patient barriers in the implementation of isoniazid preventive therapy for people living with HIV attending care and treatment centers, Songea Municipality, Tanzania. Pan Afr Med J. 2021;38:197.
Russom M, Jeannetot DYB, Tesfamariam S, Stricker BH, Verhamme K. Perspectives of healthcare professionals on factors limiting implementation of isoniazid preventive therapy in people living with HIV in Eritrea: a qualitative study. Risk Manag Healthc Policy. 2022;15:1407–19.
Semitala FC, Musinguzi A, Ssemata J, Welishe F, Nabunje J, Kadota JL, et al. Acceptance and completion of rifapentine-based TB preventive therapy (3HP) among people living with HIV (PLHIV) in Kampala, Uganda—patient and health worker perspectives. Implement Sci Commun. 2021;2(1):71.
Birken S, Clary A, Tabriz AA, Turner K, Meza R, Zizzi A, et al. Middle managers’ role in implementing evidence-based practices in healthcare: a systematic review. Implement Sci. 2018;13(1):149.
Kakande E, Christian C, Balzer LB, Owaraganise A, Nugent JR, DiIeso W, et al. A mid-level health manager intervention to promote uptake of isoniazid preventive therapy among people with HIV in Uganda: a cluster randomised trial. Lancet HIV. 2022;9(9):e607–16.
Green L, Kreuter M. Health promotion today and a framework for planning. Health promotion planning: an educational and environmental approach. Palo Alto: Mayfield Publishers; 1991. p. 1–42.
Google Scholar
Dedoose Version 9.0.17, cloud application for managing, analyzing, and presenting qualitative and mixed method research data (2021). Los Angeles: SocioCultural Research Consultants, LLC www.dedoose.com .
Gale NK, Heath G, Cameron E, Rashid S, Redwood S. Using the framework method for the analysis of qualitative data in multi-disciplinary health research. BMC Med Res Methodol. 2013;13(1):117.
Charmaz K. Constructing grounded theory. 2nd ed. London: SAGE Publications; 2014.
Timmermans S, Tavory I. Theory construction in qualitative research: from grounded theory to abductive analysis. Sociol Theory. 2012;30(3):167–86.
Thomas DR. A general inductive approach for analyzing qualitative evaluation data. Am J Eval. 2006;27(2):237–46.
Rogers EM. Diffusion of innovations. 5th ed. New York: Free P; 2003.
Bandura A. Social learning theory. Englewood Cliffs: Prentice Hall; 1977.
Kim J, Jang J, Kim B, Lee KH. Effect of the PRECEDE-PROCEED model on health programs: a systematic review and meta-analysis. Syst Rev. 2022;11(1):213.
Abimbola S, Baatiema L, Bigdeli M. The impacts of decentralization on health system equity, efficiency and resilience: a realist synthesis of the evidence. Health Policy Plan. 2019;34(8):605–17.
Sumah AM, Baatiema L, Abimbola S. The impacts of decentralisation on health-related equity: a systematic review of the evidence. Health Policy. 2016;120(10):1183–92.
Article PubMed Google Scholar
Schneider H, George A, Mukinda F, Tabana H. District governance and improved maternal, neonatal and child health in South Africa: Pathways of Change. Health Syst Reform. 2020;6(1):e1669943.
Waiswa P, Mpanga F, Bagenda D, Kananura RM, O’Connell T, Henriksson DK, et al. Child health and the implementation of Community and District-management Empowerment for Scale-up (CODES) in Uganda: a randomised controlled trial. BMJ Glob Health. 2021;6(6):e006084.
Henriksson DK, Ayebare F, Waiswa P, Peterson SS, Tumushabe EK, Fredriksson M. Enablers and barriers to evidence based planning in the district health system in Uganda; perceptions of district health managers. BMC Health Serv Res. 2017;17(1):103.
Bulthuis SE, Kok MC, Amon S, Agyemang SA, Nsabagasani X, Sanudi L, et al. How district health decision-making is shaped within decentralised contexts: A qualitative research in Malawi, Uganda and Ghana. Glob Public Health. 2021;16(1):120–35.
Katahoire AR, Henriksson DK, Ssegujja E, Waiswa P, Ayebare F, Bagenda D, et al. Improving child survival through a district management strengthening and community empowerment intervention: early implementation experiences from Uganda. BMC Public Health. 2015;15(1):797.
Download references
Acknowledgements
We thank the Uganda Ministry of Health and the participating District Health Officers and District TB and Leprosy Supervisors for their time participating in the SEARCH-IPT trial. We also acknowledge the staff at IDRC for their work on this project.
This work was supported by a grant from the National Institute of Allergy and Infectious Diseases (NIAID R01AI125000). The funder had no role in the study design, manuscript development, or decision to submit for publication.
Author information
Authors and affiliations.
University of California San Francisco, San Francisco, CA, USA
Canice Christian, Jason Johnson Peretz, Starley B. Shade, Diane V. Havlir, Gabriel Chamie & Carol S. Camlin
Infectious Diseases Research Collaboration, Kampala, Uganda
Elijah Kakande, Violah Nahurira, Cecilia Akatukwasa, Fredrick Atwine, Robert Bakanoma, Harriet Itiakorit, Asiphas Owaraganise & Jane Kabami
Sustainable East Africa Research Collaboration (SEARCH)-IPT Trial, Mbarara, Uganda
William DiIeso & Derek Rast
Makerere University, Kampala, Uganda
Moses R. Kamya
You can also search for this author in PubMed Google Scholar
Contributions
CSC, GC, DVH, MRK, and SBS contributed to the writing and design of the study protocol. CC, EK, VN, CA, FA, RB, HI, AO, WD, DR, and JK assisted in executing study activities. CA, FA, RB, and HI conducted data collection activities. CC, JJP, and CSC assisted in data analysis. CC wrote the first draft of this manuscript. All authors contributed to writing and editing this manuscript.
Corresponding author
Correspondence to Canice Christian .
Ethics declarations
Ethics approval and consent to participate.
This study and all study activities were conducted in accordance with the Declaration of Helsinki. Approval was obtained by the Institutional Review Boards at the School of Medicine Research and Ethics Committee at Makerere University School of Medicine (2017–116), the University of California, San Francisco (17–22136), and the Uganda National Council for Science and Technology (HS 2331). Written, informed consent was obtained by all study participants prior to study activities. A written summary of the study was provided to participants prior to consent being obtained.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Christian, C., Kakande, E., Nahurira, V. et al. Mid-level managers’ perspectives on implementing isoniazid preventive therapy for people living with HIV in Ugandan health districts: a qualitative study. BMC Health Serv Res 24 , 313 (2024). https://doi.org/10.1186/s12913-024-10803-9
Download citation
Received : 15 March 2023
Accepted : 28 February 2024
Published : 08 March 2024
DOI : https://doi.org/10.1186/s12913-024-10803-9
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- TB preventive therapy
- Mid-level managers
- Health systems
BMC Health Services Research
ISSN: 1472-6963
- General enquiries: [email protected]
EDITORIAL article
Editorial: the evolution in pharmacology of infectious diseases: 2022.

- 1 UCL School of Pharmacy, University College London, London, United Kingdom
- 2 Department of Clinical Pharmacy, School of Pharmacy, Shahid Beheshti University of Medical Sciences, Tehran, Iran
- 3 School of Health and Life Sciences, Teesside University, Middlesbrough, United Kingdom
Editorial on the Research Topic The evolution in pharmacology of infectious diseases: 2022
1 The landscape of infectious diseases post-pandemic: a pharmacological perspective
The era post-2020 has marked a defining moment in the evolution of pharmacology, with a particular emphasis on infectious diseases ( Baker et al., 2022 ). This period has been characterized by the relentless global campaign against COVID-19 and the emergence of a spectrum of other infectious pathogens ( Tabish, 2020 ). It was during this time that the field of pharmacology experienced an unprecedented amalgamation of global research endeavors and the development of innovative therapeutic solutions. The research conducted in these years has not only paved the way for groundbreaking therapeutic strategies but has also illustrated the extraordinary capacity for resilience and innovation within the scientific community ( Rijs and Fenter, 2020 ). These advancements, born out of necessity in the face of global health crises ( Betz et al., 2023 ), have reshaped the landscape of infectious disease treatment and prevention.
This editorial offers an insightful examination and analysis of the significant advancements in the field of infectious diseases from 2022 to 2023, specifically focusing on how these developments have transformed our approaches to combating such diseases. At the heart of this exploration is an overview of 15 pivotal articles featured in our Research Topic . These articles, contributed by 120 distinguished experts worldwide, represent a critical blend of knowledge, combining a range of expert insights and discoveries. This collective wisdom is crucial for developing innovative strategies to treat infectious diseases and significantly enhances our methods for addressing global health challenges. Additionally, this editorial delves into the interplay between pathogens and recent advances in drug development, highlighting how this period has served as both a rigorous test of scientific resolve and a catalyst for notable progress in medical science. By exploring the dynamic relationship between emerging diseases and pharmacological innovations, it underscores how this era has been challenging yet instrumental in driving substantial advancements in the evolution of the medical field.
2 COVID-19 therapeutic developments: key research findings and innovations
In the ongoing struggle against COVID-19, the scientific community has been unwavering in its quest to uncover new therapeutic avenues. Umakanthan et al. made a significant contribution with their study on the unexpected benefits of Statins, commonly used for lipid management, in reducing mortality among hospitalized COVID-19 patients. This groundbreaking finding not only highlights the anti-inflammatory and immunomodulatory properties of statins but also opens new avenues for COVID-19 treatment, suggesting a broader application of these drugs beyond their traditional use. Complementing this, Barati et al. ’s research on the combination of Noscapine and Licorice for cough relief in COVID-19 outpatients indicated a slight superiority over traditional treatments. The study is particularly pertinent as it addresses the symptomatic burden experienced by COVID-19 patients and emphasizes the importance of effective symptom management strategies in viral infections. It signifies the potential of integrating traditional remedies with modern pharmacological practices, a theme that resonates throughout our Research Topic. For instance, Maen et al. ’s study on Thymoquinone formulation (NP-101) against SARS-CoV-2 revealed its potential as a novel treatment, pointing to new directions in antiviral therapy. This finding not only contributes to the ongoing efforts against COVID-19 but also underscores the importance of exploring natural compounds in drug development.
The exploration of combination therapy as a strategy for treating COVID-19 has gained traction, as highlighted by Akinbodale et al. (2022) . This approach involves the concurrent use of multiple medications, targeting the virus from various perspectives to potentially enhance patient outcomes. A notable study by Dastan et al. examined the combined use of Tocilizumab, a biologic drug approved for treating moderate to severe rheumatoid arthritis in adults, and Baricitinib, an immunomodulatory medication for rheumatoid arthritis. Their research, focusing on severe COVID-19 cases, revealed that while this combination did not significantly reduce mortality rates, it was associated with a reduced necessity for Intensive Care Unit (ICU) admission. This outcome underscores the potential benefits of such combination therapies in certain patient groups and clinical settings. It also emphasizes the growing importance of personalized medicine in effectively managing COVID-19.
In a real-world study, Zhong et al. evaluated the efficacy and safety of Nirmatrelvir/Ritonavir co-administration in patients with rheumatic disease infected with SARS-CoV-2. This study found that using Nirmatrelvir/Ritonavir as part of standard or early treatment regimens led to a shorter time for symptom resolution compared to a control group. Even when this combination of drugs was administered after 5 days of symptom onset, it still offered benefits for rheumatic patients. The study highlighted the importance of early Nirmatrelvir/Ritonavir utilization and following the recommended regimen, showing favorable outcomes and an acceptable safety profile for immunosuppressed rheumatic patients. In addition, Wei et al. ’s comparative analysis of Azvudine, originally used for HIV-1, and Nirmatrelvir/Ritonavir concluded that Azvudine offered similar safety but slightly better clinical benefits in hospitalized patients. This finding adds to the growing body of literature on effective COVID-19 treatments and highlights the importance of continual evaluation and comparison of therapeutic options in the rapidly evolving landscape of the pandemic. In a similar vein, Zhu ’s perspective on Azvudine, showcased its effectiveness in treating moderate COVID-19 cases. This work exemplifies the potential of drug repurposing in the pandemic era, providing a cost-effective solution to the global health crisis. It also underscores the importance of adaptability in pharmaceutical research, as existing drugs can be re-evaluated and repurposed to meet emerging health challenges.
Finally, the investigation into the cardiac impacts of Remdesivir by Hajimoradi et al. shed light on the complexities of COVID-19 treatment, particularly the incidence of sinus bradycardia in patients treated with this antiviral drug. This finding highlights the need for comprehensive monitoring during treatment, underscoring the multifaceted nature of COVID-19 and the importance of considering the broader implications of antiviral therapies.
3 Innovations beyond COVID-19
Moving beyond COVID-19, these years also witnessed significant advancements in the treatment of other infectious diseases. For instance, El-Mahdy et al. ’s study on chronic hepatitis C patients treated with direct-acting antivirals showed marked improvements in liver function and antioxidant profiles. This work marks a milestone in the management of this chronic infection and highlights the progress made in understanding and treating hepatitis C.
In the realm of cystic fibrosis, a debilitating genetic disease, Rakhshan et al. ’s evaluation of inhaled Amikacin as an adjunct therapy in treating Pseudomonas aeruginosa exacerbations represents a significant step forward. This clinical trial not only highlights innovation in treatment strategies but also emphasizes the importance of addressing the specific needs of patients with cystic fibrosis.
Drug repurposing continued to be a key theme in the research landscape, as exemplified by Porta et al. ’s review on Chagas disease. This neglected tropical disease, caused by the protozoan parasite Trypanosoma cruzi , has been a longstanding public health challenge, particularly in Latin America. The review emphasized the urgent need for new treatment strategies and highlighted the potential of repurposing existing drugs as a cost-effective and expedient approach to address this issue. In the field of cutaneous leishmaniasis, another neglected tropical disease, Hakamifard et al. ’s clinical trial on liposomal Clarithromycin, an antibiotic, combined with the antileishmanial agent Glucantime suggested a significant effect in reducing lesion size. This clinical trial is particularly relevant for regions where leishmaniasis remains a significant public health issue and where effective treatment options are sorely needed.
The challenge of drug resistance, a critical concern in modern medicine, was highlighted in Sichen et al. ’s analysis of multidrug resistance in tuberculosis patients in Northeast China. This report underscores the need for continued vigilance and innovation in the fight against drug-resistant infections, a challenge that continues to evolve and requires new strategies. Similarly, Wang et al. ’s review on the complexities of non-tuberculosis mycobacteria skin infections called for a more nuanced understanding and tailored therapeutic strategies. This review reflects the evolving nature of infectious diseases and the corresponding need for adaptive and targeted treatment approaches.
In veterinary medicine, Lee et al. ’s pharmacokinetic/pharmacodynamic study on Tylosin, a macrolide antibiotic, in pigs co-infected with Actinobacillus pleuropneumoniae and Pasteurella multocida provided valuable insights into effective dosing strategies. This study not only advances our understanding of antimicrobial therapy in animal health but also reflects the interconnectedness of human and animal health, a concept integral to the One Health approach ( Mackenzie and Jeggo, 2019 ).
4 Conclusion
The period from 2022 to 2023 in pharmacology has been characterized by a blend of innovation, repurposing, and deepened understanding of infectious diseases. The articles in our Research Topic have significantly contributed to this evolving landscape, where old and new therapies converge, providing hope and direction for future research and patient care. This era’s developments serve as a beacon of progress, illuminating the path towards more effective, safe, and accessible treatments for infectious diseases globally. As we move forward, the lessons learned, and strategies developed during this time will undoubtedly continue to influence the field of pharmacology and the broader scientific community’s approach to addressing health challenges.
Author contributions
EP: Conceptualization, Formal Analysis, Investigation, Supervision, Writing–original draft, Writing–review and editing. AS: Conceptualization, Formal Analysis, Investigation, Supervision, Writing–original draft, Writing–review and editing. KK: Conceptualization, Formal Analysis, Investigation, Supervision, Writing–original draft, Writing–review and editing.
Acknowledgments
We sincerely thank the authors, reviewers, and editors for their invaluable contributions and efforts that have greatly enriched this Research Topic.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Akinbolade, S., Coughlan, D., Fairbairn, R., McConkey, G., Powell, H., Ogunbayo, D., et al. (2022). Combination therapies for COVID-19: an overview of the clinical trials landscape. Br. J. Clin. Pharmacol. 88 (4), 1590–1597. doi:10.1111/bcp.15089
PubMed Abstract | CrossRef Full Text | Google Scholar
Baker, R. E., Mahmud, A. S., Miller, I. F., Rajeev, M., Rasambainarivo, F., Rice, B. L., et al. (2022). Metcalf CJE. Infectious disease in an era of global change. Nat. Rev. Microbiol. 20 (4), 193–205. doi:10.1038/s41579-021-00639-z
Betz, U. A. K., Arora, L., Assal, R. A., Azevedo, H., Baldwin, J., Becker, M. S., et al. (2023). Game changers in science and technology - now and beyond. Technol. Forecast. Soc. Change. 193, 122588. doi:10.1016/j.techfore.2023.122588
CrossRef Full Text | Google Scholar
Mackenzie, J. S., and Jeggo, M. (2019). The One health approach-why is it so important? Trop. Med. Infect. Dis. 4 (2), 88. doi:10.3390/tropicalmed4020088
Rijs, C., and Fenter, F. (2020). The academic response to COVID-19. Front. Public Health 8, 621563. doi:10.3389/fpubh.2020.621563
Tabish, S. A. (2020). COVID-19 pandemic: emerging perspectives and future trends. J. Public Health Res. 9 (1), 1786. doi:10.4081/jphr.2020.1786
Keywords: Chagas disease, COVID-19, cystic fibrosis, hepatitis C, infectious diseases, leishmaniasis, pharmacology, tuberculosis
Citation: Porta EOJ, Saffaei A and Kalesh K (2024) Editorial: The evolution in pharmacology of infectious diseases: 2022. Front. Pharmacol. 15:1386077. doi: 10.3389/fphar.2024.1386077
Received: 14 February 2024; Accepted: 06 March 2024; Published: 12 March 2024.
Edited and reviewed by:
Copyright © 2024 Porta, Saffaei and Kalesh. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Exequiel O. J. Porta, [email protected] ; Ali Saffaei, [email protected] ; Karunakaran Kalesh, [email protected]
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- News & Views
- Published: 08 March 2024
Infectious diseases
Restocking the tuberculosis drug arsenal
- Eric L. Nuermberger ORCID: orcid.org/0000-0003-1440-9889 1 &
- Richard E. Chaisson ORCID: orcid.org/0000-0003-4710-925X 1
Nature Medicine ( 2024 ) Cite this article
170 Accesses
10 Altmetric
Metrics details
- Tuberculosis
After many lean years, important progress has been made in updating the anti-tuberculosis drug armamentarium; a new drug that targets bacterial protein synthesis is one of several that could help transform the treatment of this neglected and deadly disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
24,99 € / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
195,33 € per year
only 16,28 € per issue
Rent or buy this article
Prices vary by article type
Prices may be subject to local taxes which are calculated during checkout
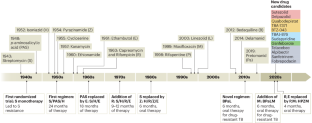
Chaisson, R. E., Frick, M. & Nahid, P. Int. J. Tuberc. Lung Dis. 26 , 186–189 (2022).
Article CAS PubMed PubMed Central Google Scholar
Dorman, S. E. et al. N. Engl. J. Med. 384 , 1705–1718 (2021).
Diacon, A. H. et al. Nat. Med. https://doi.org/10.1038/s41591-024-02829-7 (2024).
Article PubMed Google Scholar
Conradie, F. et al. N. Engl. J. Med. 382 , 893–902 (2020).
Gler, M. T. et al. N. Engl. J. Med. 366 , 2151–2160 (2012).
Article CAS PubMed Google Scholar
Conradie, F. et al. N. Engl. J. Med. 387 , 810–823 (2022).
Li, X. et al. J. Med. Chem. 60 , 8011–8026 (2017).
Dawson, R. et al. Antimicrob. Agents Chemother. 67 , e0147722 (2023).
Gupta, A. et al. Clin. Infect. Dis. 62 , 761–769 (2016).
Nogueira, B. M. F. et al. EMBO Mol. Med. 14 , e14088 (2022).
Xie, Y. L. et al. Sci. Transl. Med. 13 , eabd7618 (2021).
Ma, Z., Lienhardt, C., McIlleron, H., Nunn, A. J. & Wang, X. Lancet 375 , 2100–2109 (2010).
Download references
Author information
Authors and affiliations.
Center for Tuberculosis Research, Division of Infectious Diseases, Johns Hopkins University School of Medicine, Baltimore, MD, USA
Eric L. Nuermberger & Richard E. Chaisson
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Richard E. Chaisson .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Rights and permissions
Reprints and permissions
About this article
Cite this article.
Nuermberger, E.L., Chaisson, R.E. Restocking the tuberculosis drug arsenal. Nat Med (2024). https://doi.org/10.1038/s41591-024-02840-y
Download citation
Published : 08 March 2024
DOI : https://doi.org/10.1038/s41591-024-02840-y
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.
An official website of the United States government
The .gov means it's official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you're on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- Browse Titles
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-.

StatPearls [Internet].
Tuberculosis.
Rotimi Adigun ; Rahulkumar Singh .
Affiliations
Last Update: July 11, 2023 .
- Continuing Education Activity
Tuberculosis (TB) is a human disease caused by Mycobacterium tuberculosis. It mainly affects the lungs, making pulmonary disease the most common presentation. Other commonly affected organ systems include the respiratory system, the gastrointestinal (GI) system, the lymphoreticular system, the skin, the central nervous system, the musculoskeletal system, the reproductive system, and the liver. In the past few decades, there has been a concerted global effort to eradicate tuberculosis. Despite the gains in tuberculosis control and the decline in both new cases and mortality, it still accounts for a huge burden of morbidity and mortality worldwide. This activity reviews the evaluation and management of tuberculosis and highlights the role of interprofessional team members in collaborating to provide well-coordinated care and enhance outcomes for affected patients.
- Identify the epidemiology of tuberculosis.
- Review the presentation of a patient with tuberculosis.
- Outline the treatment and management options available for tuberculosis.
- Employ interprofessional team strategies for improving care and outcomes in patients with tuberculosis.
- Introduction
Tuberculosis (TB) is an ancient human disease caused by Mycobacterium tuberculosis which mainly affects the lungs, making pulmonary disease the most common presentation (K Zaman, 2010) [1] . However, TB is a multi-systemic disease with a protean presentation. The organ system most commonly affected includes the respiratory system, the gastrointestinal (GI) system, the lymphoreticular system, the skin, the central nervous system, the musculoskeletal system, the reproductive system, and the liver [2] [3] .
Evidence of TB has been reported in human remains dated thousands of years (Hershkovitz et al., 2017, K Zaman, 2010). For a human pathogen with no known environmental reservoir, Mycobacterium tuberculosis has honed the art of survival and has persisted in human communities from antiquity through modern times.
In the past few decades, there has been a concerted global effort to eradicate TB. These efforts had yielded some positive dividends, especially since 2000 when the World Health Organization (WHO, 2017) estimated that the global incidence rate for tuberculosis has fallen by 1.5% every year. Furthermore, mortality arising from tuberculosis has significantly and steadily declined. The World Health Organization (WHO, 2016) reports a 22% drop in global TB mortality from 2000 through 2015.
Despite the gains in tuberculosis control and the decline in both new cases and mortality, TB still accounts for a huge burden of morbidity and mortality worldwide. The bulk of the global burden of new infection and tuberculosis death is borne by developing countries, with 6 countries, India, Indonesia, China, Nigeria, Pakistan, and South Africa, accounting for 60% of TB death in 2015 (WHO, 2017). [4]
Tuberculosis remains a significant cause of both illness and death in developed countries, especially among individuals with a suppressed immune system [5] [6] . People with HIV are particularly vulnerable to death due to tuberculosis. Tuberculosis accounted for 35% of global mortality in individuals with HIV/AIDS in 2015. (W.H.O, 2017). Children are also vulnerable, and tuberculosis was responsible for one million illnesses in children in 2015, according to the WHO.
M. tuberculosis causes tuberculosis. M. tuberculosis is an alcohol and acid-fast bacillus. It is part of a group of organisms classified as the M. tuberculosis complex. Other members of this group are Mycobacterium africanum, Mycobacterium bovis, and Mycobacterium microti [1] . Most other mycobacteria organisms are classified as non-tuberculous or atypical mycobacterial organisms.
M. tuberculosis is a non-spore-forming, non-motile, obligate-aerobic, facultative, catalase-negative, intracellular bacteria. The organism is neither gram-positive nor gram-negative because of a very poor reaction with the Gram stain. Weakly positive cells can sometimes be demonstrated on Gram stain, a phenomenon known as "ghost cells."
The organism has several unique features compared to other bacteria, such as the presence of several lipids in the cell wall, including mycolic acid, cord factor, and Wax-D. The high lipid content of the cell wall is thought to contribute to the following properties of M. tuberculosis infection:
- Resistance to several antibiotics
- Difficulty staining with Gram stain and several other stains
- Ability to survive under extreme conditions such as extreme acidity or alkalinity, low oxygen situation, and intracellular survival(within the macrophage)
The Ziehl-Neelsen stain is one of the most commonly used stains to diagnose T.B. The sample is initially stained with carbol fuchsin (pink color stain), decolorized with acid-alcohol, and then counter-stained with another stain (usually, blue-colored methylene blue). A positive sample would retain the pink color of the original carbol fuchsin, hence the designation, alcohol, and acid-fast bacillus (AAFB).
- Epidemiology
Geographic Distribution
Tuberculosis is present globally [1] . However, developing countries account for a disproportionate share of tuberculosis disease burden. In addition to the six countries listed above, several countries in Asia, Africa, Eastern Europe, and Latin and Central America continue to have an unacceptably high burden of tuberculosis.
In more advanced countries, high-burden tuberculosis is seen among recent arrivals from tuberculosis-endemic zones, healthcare workers, and HIV-positive individuals. The use of immunosuppressive agents such as long-term corticosteroid therapy has also been associated with an increased risk.
More recently, the use of a monoclonal antibody targeting the inflammatory cytokine, tumor necrotic factor alpha (TNF-alpha), has been associated with an increased risk. Antagonists of this cytokine include several monoclonal antibodies (biologics) used for the treatment of inflammatory disorders. Drugs in this category include infliximab, adalimumab, etanercept, and golimumab. Patients using any of these medications should be monitored for tuberculosis before and during the period of drug treatment.
Other Major Risk Factors
- Socioeconomic factors: Poverty, malnutrition, wars
- Immunosuppression: HIV/AIDS, chronic immunosuppressive therapy (steroids, monoclonal antibodies against tumor necrotic factor), a poorly developed immune system (children, primary immunodeficiency disorders)
- Occupational: Mining, construction workers, pneumoconiosis (silicosis)
Multi-Drug Resistant Tuberculosis (MDR-TB) and Extremely Multi-Drug Resistant Tuberculosis (XDR-TB)
- This refers to tuberculosis with strains of Mycobacterium which have developed resistance to the classic anti-tuberculosis medications. TB is especially a problem among patients with HIV/AIDS. Resistance to multiple anti-tuberculosis medications, including at least the two standard anti-tuberculous medications, Rifampicin or Isoniazid, is required to make a diagnosis of MDR-TB.
- Seventy-five percent of MDR-TB is considered primary MDR-TB, caused by infection with MDR-TB pathogens. The remaining 25% are acquired and occur when a patient develops resistance to treatment for tuberculosis. Inappropriate treatment for tuberculosis because of several factors such as antibiotic abuse; inadequate dosage; incomplete treatment, is the number one cause of acquired MDR-TB.
- This is a more severe type of MDR-TB. Diagnosis requires resistance to at least four anti-tuberculous medications, including resistance to Rifampicin, Isoniazid, and resistance to any two of the newer anti-tuberculous medications. The newer medications implicated in XDR-TB are the fluoroquinolones (Levofloxacin and moxifloxacin) and the injectable second-line aminoglycosides, Kanamycin, Capreomycin, and amikacin.
- The mechanism of developing XDR-TB is similar to the mechanism for developing MDR-TB.
- XDR -TB is an uncommon occurrence.
- Pathophysiology
Although usually a lung infection, tuberculosis is a multi-system disease with protean manifestation. The principal mode of spread is through the inhalation of infected aerosolized droplets.
The body's ability to effectively limit or eliminate the infective inoculum is determined by the immune status of the individual, genetic factors, and whether it is a primary or secondary exposure to the organism. Additionally, M. tuberculosis possesses several virulence factors that make it difficult for alveolar macrophages to eliminate the organism from an infected individual. The virulence factors include the high mycolic acid content of the bacteria's outer capsule, which makes phagocytosis to be more difficult for alveolar macrophages. Furthermore, some of the other constituents of the cell wall, such as the cord factor, may directly damage alveolar macrophages. Several studies have shown that mycobacteria tuberculosis prevents the formation of an effective phagolysosome, hence, preventing or limiting the elimination of the organisms.
The first contact of the Mycobacterium organism with a host leads to manifestations known as primary tuberculosis. This primary TB is usually localized to the middle portion of the lungs, and this is known as the Ghon focus of primary TB. In most infected individuals, the Ghon focus enters a state of latency. This state is known as latent tuberculosis.
Latent tuberculosis is capable of being reactivated after immunosuppression in the host. A small proportion of people would develop an active disease following first exposure. Such cases are referred to as primary progressive tuberculosis. Primary progressive tuberculosis is seen in children, malnourished people, people with immunosuppression, and individuals on long-term steroid use.
Most people who develop tuberculosis do so after a long period of latency (usually several years after the initial primary infection). This is known as secondary tuberculosis. Secondary tuberculosis usually occurs because of the reactivation of latent tuberculosis infection. The lesions of secondary tuberculosis are in the lung apices. A smaller proportion of people who develop secondary tuberculosis do so after getting infected a second time (re-infection).
The lesions of secondary tuberculosis are similar for both reactivation and reinfection in terms of location (at the lung apices), and the presence of cavitation enables a distinction from primary progressive tuberculosis which tends to be in the middle lung zones and lacks marked tissue damage or cavitation.
Type-IV Hypersensitivity and Caseating Granuloma
Tuberculosis is a classic example of a cell-mediated delayed type IV hypersensitivity reaction.
Delayed Hypersensitivity Reaction: By stimulating the immune cells (the helper T-Lymphocyte, CD4+ cells), Mycobacterium tuberculosis induces the recruitment and activation of tissue macrophages. This process is enhanced and sustained by the production of cytokines, especially interferon-gamma.
Two main changes involving macrophages occur during this process, namely, the formation of multinucleated giant cells and the formation of epithelioid cells. Giant cells are aggregates of macrophages that are fused together and function to optimize phagocytosis. The aggregation of giant cells surrounding the Mycobacterium particle and the surrounding lymphocytes and other cells is known as a granuloma.
Epithelioid cells are macrophages that have undergone a change in shape and have developed the ability for cytokine synthesis. Epithelioid cells are modified macrophages and have a flattened (spindle-like shape) as opposed to the globular shape characteristic of normal macrophages. Epithelioid cells often coalesce together to form giant cells in a tuberculoid granuloma.
In addition to interferon-gamma (IFN-gamma), the following cytokines play important roles in the formation of a tuberculosis granuloma, Interleukin-4 (IL-4), Interleukin-6 (IL-6), and tumor necrotic factor-alpha (TNF-alpha).
The appearance of the granuloma in tuberculosis has been described as caseous or cheese-like on gross examination. This is principally explained by the rich mycolic acid content of the mycobacterium cell wall. Because of this unique quality, the term caseous or caseating necrosis has been used to describe granulomatous necrosis caused by mycobacteria tuberculosis.
Histologically, caseous necrosis would present as a central area of uniform eosinophilia on routine hematoxylin and eosin stain.
- Histopathology
The granuloma is the diagnostic histopathological hallmark of tuberculosis.
The defining features of the granuloma of tuberculosis are:
- Caseation or caseous necrosis is demonstrable as a region of central eosinophilia.
- Multinucleated giant cells
- History and Physical
A chronic cough, hemoptysis, weight loss, low-grade fever, and night sweats are some of the most common physical findings in pulmonary tuberculosis.
Secondary tuberculosis differs in clinical presentation from primary progressive disease. In secondary disease, the tissue reaction and hypersensitivity are more severe, and patients usually form cavities in the upper portion of the lungs.
Pulmonary or systemic dissemination of the tubercles may be seen in active disease, and this may manifest as miliary tuberculosis characterized by millet-shaped lesions on chest x-ray. Disseminated tuberculosis may also be seen in the spine, the central nervous system, or the bowel.
Screening Tests
Tuberculin skin testing: Mantoux test (skin testing with PP D)
The Mantoux reaction following the injection of a dose of PPD (purified protein derivative) is the traditional screening test for exposure to Tuberculosis. The result is interpreted taking into consideration the patient's overall risk of exposure. Patients are classified into 3 groups based on the risk of exposure with three corresponding cut-off points. The 3 major groups used are discussed below.
Low Risk
- Individuals with minimal probability of exposure are considered to have a positive Mantoux test only if there is very significant induration following intradermal injection of PPD. The cut-off point for this group of people (with minimal risk of exposure) is taken to be 15 mm.
Intermediate Risk
- Individuals with intermediate probability are considered positive if the induration is greater than 10 mm.
- Individuals with a high risk of a probability of exposure are considered positive if the induration is greater than 5 mm.
Examples of Patients in the Different Risk Categories
- Low Risk/Low Probability: Patients with no known risk of exposure to TB. Example: No history of travel, military service, HIV-negative, no contact with a chronic cough patient, no occupational exposure, no history of steroids. Not a resident of a TB-endemic region.
- Intermediate Risk/Probability: Residents of TB-endemic countries (Latin America, Sub -Sahara Africa, Asia), workers or residents of shelters, Medical or microbiology department personnel.
- High Risk/Probability: HIV-positive patient, a patient with evidence of the previous TB such as the healed scar on an x-ray), contact with chronic cough patients.
Note that a Mantoux test indicates exposure or latent tuberculosis. However, this test lacks specificity, and patients would require subsequent visits for interpreting the results as well as chest x-ray for confirmation. Although relatively sensitive, the Mantoux reaction is not very specific and may give false-positive reactions in individuals who have been exposed to the BCG vaccine.
Interferon release assays (IGRA, Quantiferon Assays)
This is a tuberculosis screening test that is more specific and equally as sensitive as the Mantoux test. This test assays for the level of the inflammatory cytokine, especially interferon-gamma.
The advantages of antigen-specific stimulation of IFN-γ release, especially in those with prior vaccination with BCG vaccine, include the test requires a single blood draw, obviating the need for repeat visits to interpret results. Furthermore, additional investigations, such as HIV screening, could be performed (after patient consent) on the same blood draw.
Quantiferon's disadvantages include cost and the technical expertise required to perform the test.
Screening in Immunocompromised Patients
Immunocompromised patients may show lower levels of reaction to PPD or false-negative Mantoux because of cutaneous anergy.
A high level of suspicion should be entertained when reviewing negative screening tests for tuberculosis in HIV-positive individuals.
The Significance of Screening
A positive screening test indicates exposure to tuberculosis and a high chance of developing active tuberculosis in the future. Tuberculosis incidence in patients with positive Mantoux test averages between 2% to 10% without treatment.
Patients with a positive test should have a chest x-ray as a minimum diagnostic test. In some cases, these patients should have additional tests. Patients meeting the criteria for latent tuberculosis should receive prophylaxis with isoniazid.
Screening Questionnaires for Resource-Poo r Settings
Several screening questionnaires have been validated to enable healthcare workers working in remote and resource-poor environments to screen for tuberculosis.
These questionnaires make use of an algorithm that combines several clinical signs and symptoms of tuberculosis. Some of the commonly used symptoms are:
- Chronic cough
- Weight loss
- Fever and night sweats
- History of contact
- Blood in sputum
Several studies have confirmed the utility of using several criteria rather than a focus on only chronic cough or weight loss.
Confirmatory and Diagnostic Tests
- A chest x-ray is indicated to rule out or rule in the presence of active disease in all screening test-positive cases.
- Acid Fast Staining-Ziehl-Neelsen
- Nuclear Amplification and Gene-Based Tests: These represent a new generation of diagnostic tools for tuberculosis. These tests enable the identification of bacteria or bacteria particles by making use of DNA-based molecular techniques.
The new molecular-based techniques are faster and enable rapid diagnosis with high precision. Confirmation of TB could be made in hours rather than the days or weeks it takes to wait for a standard culture. This is very important, especially among immunocompromised hosts where there is a high rate of false-negative results. Some molecular-based tests also allow for the identification of multidrug-resistant tuberculosis.
- Treatment / Management
Latent Tuberculosis
2020 LTBI treatment guidelines include the NTCA- and CDC-recommended treatment regimens that comprise three preferred rifamycin-based regimens and two alternative monotherapy regimens with daily isoniazid. These are only recommended for persons infected with Mycobacterium tuberculosis that is presumed to be susceptible to isoniazid or rifampin. A regimen of 3 months of once-weekly isoniazid plus rifapentine is a preferred regimen that is strongly recommended for children aged more than 2 years and adults. Another option is 4 months of daily rifampin for HIV-negative adults and children of all ages. Three months of daily isoniazid plus rifampin is a preferred treatment that is conditionally recommended for adults and children of all ages and for patients with HIV. Regimens of 6 or 9 months of daily isoniazid are alternative recommended regimens.
Treatment of Active Infection
Treatment of confirmed TB requires a combination of drugs. Combination therapy is always indicated, and monotherapy should never be used for tuberculosis. The most common regimen for TB includes the following anti-TB medications:
First-Line Medications, Group 1
- Isoniazid - Adults (maximum): 5 mg/kg (300 mg) daily; 15 mg/kg (900 mg) once, twice, or three times weekly.Children (maximum): 10-15 mg/kg (300 mg) daily; 20--30 mg/kg (900 mg) twice weekly (3).Preparations. Tablets (50 mg, 100 mg, 300 mg); syrup (50 mg/5 ml); aqueous solution (100 mg/ml) for IV or IM injection.
- Rifampicin - Adults (maximum): 10 mg/kg (600 mg) once daily, twice weekly, or three times weekly.Children (maximum): 10-20 mg/kg (600 mg) once daily or twice weekly.Preparations. Capsules (150 mg, 300 mg)
- Rifabutin- Adults (maximum): 5 mg/kg (300 mg) daily, twice, or three times weekly. When rifabutin is used with efavirenz the dose of rifabutin should be increased to 450--600 mg either daily or intermittently.Children (maximum): Appropriate dosing for children is unknown. Preparations: Capsules (150 mg) for oral administration.
- RIfapentine - Adults (maximum): 10 mg/kg (600 mg), once weekly (continuation phase of treatment)Children: The drug is not approved for use in children.Preparation. Tablet (150 mg, film-coated).
- Pyrazinamide - Adults: 20-25 mg/kg per day. Children (maximum): 15-30 mg/kg (2.0 g) daily; 50 mg/kg twice weekly (2.0 g).Preparations. Tablets (500 mg).
- Ethambutol - Adults: 15-20 mg/kg per day: Children (maximum): 15-20 mg/kg per day (2.5 g); 50 mg/kg twice weekly (2.5 g). The drug can be used safely in older children but should be used with caution in children in whom visual acuity cannot be monitored (generally less than 5 years of age) (66). In younger children, EMB can be used if there is a concern with resistance to INH or RIF.Preparations. Tablets (100 mg, 400 mg) for oral administration.
Isoniazid and Rifampicin follow a 4-drug regimen (usually including Isoniazid, Rifampicin, Ethambutol, and Pyrazinamide) for 2 months or six months. Vitamin B6 is always given with Isoniazid to prevent neural damage (neuropathies).
Several other antimicrobials are effective against tuberculosis, including the following categories:
Second-Line Anti-tuberculosis Drugs, Group 2
Injectable aminoglycosides and injectable polypeptides
Injectable aminoglycosides
- Streptomycin
Injectable polypeptides
- Capreomycin
- Viomycin
Second - Line Anti-Tuberculosis Drugs, Group 3, Oral and Injectable Fluoroquinolones
Fluoroquinolones
- Levofloxacin
- Moxifloxacin
- Gatifloxacin
Second-Line Anti-tuberculosis Drugs, Group 4
- Para-aminosalicylic acid
- Cycloserine
- Ethionamide
- Prothionamide
- Thioacetazone
Third- Line Anti-Tuberculosis Drugs, Group 5
These are medications with variable but unproven efficacy against TB. They are used for total drug-resistant TB as drugs of last resort.
- Clofazimine
- Amoxicillin/clavulanic acid
- Imipenem/cilastatin
- Clarithromycin
MDR-TB, XDR-TB
Multi-drug-resistant TB is becoming increasingly common.
The combination of first-line and second-line medications is used at high doses to treat this condition.
Bedaquiline
On December 28, 2012, the United States Food and Drug Administration Agency (FDA) approved Bedaquiline as a drug for treating MDR-TB. This is the first FDA approval for an anti-TB medication in 40 years. While showing remarkable promise in drug-resistant tuberculosis, cost remains a big obstacle to delivering this drug to the people most affected by MDR-TB.
Clinical and Laboratory Monitoring
Liver function tests are required for all patients taking isoniazid. Other monitoring in TB includes monitoring for retinopathies for patients on ethambutol.
Treatment of Patients with HIV
In patients with active TB and HIV with severe immunosuppression (CD4+ 60/microliter), the recommendations are to immediately start antituberculous therapy, followed by the initiation of anti-retroviral after 2 to 4 weeks. Delaying treatment with antiretroviral drugs prevents the development of immune reconstitution inflammatory syndrome (IRIS). This is a syndrome characterized by paradoxical worsening of symptoms of primary disease when treatment with antiretroviral agents is initiated. The presenting infection should be treated immediately, and retroviral should start no earlier than 2 weeks. The earlier the antiretroviral agents are initiated, the greater the likelihood of IRIS. Unnecessary delay of antiretroviral therapy leads to an increased risk of death from AIDS.
- Differential Diagnosis
Tuberculosis is a great mimic and should be considered in the differential diagnosis of several systemic disorders. The following is a non-exhaustive list of conditions to be strongly considered when evaluating the possibility of pulmonary tuberculosis.
- Non-tuberculous mycobacterium
- Fungal infection
- Histoplasmosis
- Sarcoidosis
- Toxicity and Adverse Effect Management
Side Effect associated with most commonly used anti-TB drugs [7]
1) Isoniazid- Asymptomatic elevation of aminotransferases (10-20%), clinical hepatitis (0.6%), peripheral neurotoxicity, hypersensitivity. [8]
2) Rifampin- Pruritis, nausea & vomiting, flulike symptoms, hepatotoxicity, orange discoloration of bodily fluid.
3) Rifabutin- Neutropenia, uveitis (0.01%), polyarthralgias, hepatotoxicity (1%))
4) Rifapentine- Similar to rifampin
5) Pyrazinamide- Hepatotoxicity (1%), nausea & vomiting, polyarthralgias (40%), acute gouty arthritis, rash, and photosensitive dermatitis
6) Ethambutol- Retrobulbar neuritis (18%)
One of the most important aspects of tuberculosis treatment is close follow-up and monitoring for these side effects. Most of these side effects can be managed by either close monitoring or adjusting the dose. In some cases, the medication needs to be discontinued, and second-line therapy should be considered if other alternatives are not available.
The majority of patients with a diagnosis of TB have a good outcome. This is mainly because of effective treatment. Without treatment mortality rate for tuberculosis is more than 50%.
The following group of patients is more susceptible to worse outcomes or death following TB infection:
- Extremes of age, elderly, infants, and young children
- Delay in receiving treatment
- Radiologic evidence of extensive spread.
- Severe respiratory compromise requiring mechanical ventilation
- Immunosuppression
- Multidrug resistance (MDR) tuberculosis
- Complications
Most patients have a relatively benign course. Complications are more frequently seen in patients with the risk factors mentioned above. Some of the complications associated with tuberculosis are:
- Extensive lung destruction
- Damage to cervical sympathetic ganglia leading to Horner's syndrome.
- Acute respiratory distress syndrome
- Milliary spread (disseminated tuberculosis), including TB meningitis.
- Pneumothorax
- Systemic amyloidosis
- Pearls and Other Issues
Tuberculosis is a preventable and treatable infectious disease. Having said that, it is still one of the major contributors to morbidity and mortality in developing countries where we are still struggling to provide adequate access to care. Other challenges include lack of awareness, delayed diagnosis, poor accessibility to medication and vaccination as well as medication adherence. DOTS (Direct Observed Therapy), proposed by WHO, has been very effective in recent years to improve adherence to treatment in tuberculosis patients. [9] [10] Also, vaccination drive in developing countries has played a bigger role in decreasing the prevalence of this infection. The preventive effect of BCG vaccination is controversial, but many studies have identified vaccination as a very important tool in the fight against tuberculosis, and we need to keep our focus on childhood vaccination, especially in developing countries. [11] WHO and other health organizations have to continue their investment in developing strategies and research until we eradicate this disease from the world map. New antituberculosis drugs need to be developed to shorten or otherwise simplify the treatment of tuberculosis caused by drug-susceptible organisms, to improve the treatment of drug-resistant tuberculosis, and to provide more efficient and effective treatment of latent tuberculosis infection.
- Enhancing Healthcare Team Outcomes
A team approach involving nurses, clinicians, and technicians will lead to the best outcomes in treating patients with tuberculosis. [Level 5]
- Review Questions
- Access free multiple choice questions on this topic.
- Comment on this article.
Granuloma of Tuberculosis. Arrows pointed at multi-nucleated giant cells. Contributed By Dr. Rotimi Adigun (with permission from Kingston General Hospital)
Disclosure: Rotimi Adigun declares no relevant financial relationships with ineligible companies.
Disclosure: Rahulkumar Singh declares no relevant financial relationships with ineligible companies.
This book is distributed under the terms of the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0) ( http://creativecommons.org/licenses/by-nc-nd/4.0/ ), which permits others to distribute the work, provided that the article is not altered or used commercially. You are not required to obtain permission to distribute this article, provided that you credit the author and journal.
- Cite this Page Adigun R, Singh R. Tuberculosis. [Updated 2023 Jul 11]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-.
In this Page
Bulk download.
- Bulk download StatPearls data from FTP
Related information
- PMC PubMed Central citations
- PubMed Links to PubMed
Similar articles in PubMed
- Review Tuberculosis. [Major Infectious Diseases. 2017] Review Tuberculosis. Bloom BR, Atun R, Cohen T, Dye C, Fraser H, Gomez GB, Knight G, Murray M, Nardell E, Rubin E, et al. Major Infectious Diseases. 2017 Nov 3
- Active Tuberculosis. [StatPearls. 2024] Active Tuberculosis. Jilani TN, Avula A, Zafar Gondal A, Siddiqui AH. StatPearls. 2024 Jan
- Review The global tuberculosis epidemic and progress in care, prevention, and research: an overview in year 3 of the End TB era. [Lancet Respir Med. 2018] Review The global tuberculosis epidemic and progress in care, prevention, and research: an overview in year 3 of the End TB era. Floyd K, Glaziou P, Zumla A, Raviglione M. Lancet Respir Med. 2018 Apr; 6(4):299-314.
- Fighting the tuberculosis epidemic in the Western Pacific region: current situation and challenges ahead. [Kekkaku. 2010] Fighting the tuberculosis epidemic in the Western Pacific region: current situation and challenges ahead. van Maaren PJ. Kekkaku. 2010 Jan; 85(1):9-16.
- Consensus statement. Global burden of tuberculosis: estimated incidence, prevalence, and mortality by country. WHO Global Surveillance and Monitoring Project. [JAMA. 1999] Consensus statement. Global burden of tuberculosis: estimated incidence, prevalence, and mortality by country. WHO Global Surveillance and Monitoring Project. Dye C, Scheele S, Dolin P, Pathania V, Raviglione MC. JAMA. 1999 Aug 18; 282(7):677-86.
Recent Activity
- Tuberculosis - StatPearls Tuberculosis - StatPearls
Your browsing activity is empty.
Activity recording is turned off.
Turn recording back on
Connect with NLM
National Library of Medicine 8600 Rockville Pike Bethesda, MD 20894
Web Policies FOIA HHS Vulnerability Disclosure
Help Accessibility Careers

IMAGES
VIDEO
COMMENTS
High-dose rifapentine with or without moxifloxacin for shortening treatment of pulmonary tuberculosis: Study protocol for TBTC study 31/ACTG A5349 phase 3 clinical trial. Contemp Clin Trials. 2020 Mar; 90:105938. doi: 10.1016/j.cct.2020.105938. Epub 2020 Jan 22.
Tuberculosis (TB) is an infectious disease caused by strains of bacteria known as mycobacteria. The disease most commonly affects the lungs and can be fatal if not treated. However, most infected ...
A.H. Diacon. N Engl J Med 2022;387:2380-2381. Tuberculosis is an important cause of death worldwide, and drug resistance is implicated in one third of the almost 2 million deaths from tuberculosis ...
Global TB burden. With 10.6 million estimated new cases, 1.6 million deaths and 1.7 billion latently infected in 2021, TB remains a major global health concern [].Although able to affect anyone anywhere, in low- and middle-income countries TB stands as the eighth and seventh most common cause of death, respectively, and these countries bear the burden of 80% of all TB cases [].
Despite ongoing research, treatment for DS-TB has remained unchanged for decades. This highly effective regimen is often poorly tolerated by patients, and "drug holidays" are frequent during treatment. This, of course, increases the likelihood of relapse and evolution of drug resistance. Moreover, patients with resistant TB have to endure ...
Guidelines on the treatment of tuberculosis (TB) have essentially remained the same for the past 35 years, but are now starting to change. Ongoing clinical trials will hopefully transform the landscape for treatment of drug sensitive TB, drug resistant TB, and latent TB infection. Multiple trials are evaluating novel agents, repurposed agents, adjunctive host directed therapies, and novel ...
The detection of multidrug-resistant (MDR) tuberculosis (resistance to rifampicin and isoniazid) or rifampicin-resistant tuberculosis has increased by about 20% annually over the past decade (2009-2018); drug-resistant tuberculosis underpins about 15-20% of global tuberculosis mortality.1,2 In some countries, such as Russia and Belarus, almost half of all patients with tuberculosis have ...
Research has shown that people with TB navigate long care-seeking pathways, with multiple visits to health providers before a diagnosis is made. Mystery client (standardized patient) ...
The World Health Organization (WHO) estimates that one-quarter of the world's population has been infected with M. tuberculosis and, in 2021, 10.6 million people had active TB with 1.6 million ...
1. Introduction. Tuberculosis (TB) is a leading global public health problem, with high morbidity and mortality in humans. Until the COVID-19 pandemic, TB was still the leading cause of death from a single infectious agent, ranking above HIV/acquired immune deficiency syndrome [].The number of people newly diagnosed with TB fell from 7.1 million in 2019 to 5.8 million in 2020, and reduced ...
In addition, articles that were excluded e.g., due to the absence of full text might have influenced the current results. Finally, to adjust the effect of potential confounders including the burden of TB in the countries of the included articles, sociodemographic and economic factors, a subgroup analysis should ideally be performed.
The current global TB strategy, formulated as the End TB Strategy, is the most comprehensive ever, with three major pillars: Integrated, patient-centered care and prevention. Social and political action to address determinants of disease. Recognition of the urgent need for research to provide new tools (WHO 2015a).
Read the full article. Tuberculosis Research and Treatment publishes original research articles and review articles related to all aspects of tuberculosis, from the immunological basis of disease to translational and clinical research.
According to the WHO, in 2014, an estimated 9.6 million people developed active TB disease, of whom 1.5 million died 1.The burden of TB is heterogeneously distributed ().For example, TB incidence ...
Tuberculosis is a speciality journal focusing on basic experimental research on tuberculosis. Tuberculosis aims to publish original research and reviews. It publishes articles on host response and immunology of tuberculosis and the molecular biology, genetics and physiology of the organism. View full aims & scope. $2490. Article publishing charge.
Abstract. Tuberculosis (TB) remains one of the deadliest infectious diseases responsible for millions of deaths annually across the world. In this paper we present a general overview of TB ...
A total of 7,163 TB cases were reported during 2020 (2.2 cases per 100,000 persons), 20% fewer than during 2019 (2.7 cases per 100,000 persons). Thirty-nine states and DC reported a decrease in cases, eight states reported an increase, and three reported no change. California reported the highest number of cases (1,703), and Alaska reported the ...
Iron plays a crucial role in the growth of Mycobacterium tuberculosis (M. tuberculosis). However, the precise regulatory mechanism governing this system requires further elucidation. Additionally, limited studies have examined the impact of gene mutations related to iron on the transmission of M. tuberculosis globally. This research aims to investigate the correlation between mutations in iron ...
The database search yielded 1806 citations, of which 53 were included (Fig. 1).Research relating to the following communicable diseases (including infection and pathogens) was identified: TB, HIV, hepatitis C virus (HCV), hepatitis B virus (HBV), invasive bacterial disease (IBD), pneumonia, influenza, and COVID-19.
Despite two decades of intensified research to understand and cure tuberculosis disease, biological uncertainties remain and hamper progress. However, owing to collaborative initiatives including ...
Abstract. Tuberculosis (TB) is one of the most ancient diseases of mankind, with molecular evidence going back to over 17,000 years. In spite of newer modalities for diagnosis and treatment of TB, unfortunately, people are still suffering, and worldwide it is among the top 10 killer infectious diseases, second only to HIV.
Background Isoniazid preventive therapy (IPT) works to prevent tuberculosis (TB) among people living with HIV (PLHIV), but uptake remains low in Sub-Saharan Africa. In this analysis, we sought to identify barriers mid-level managers face in scaling IPT in Uganda and the mechanisms by which the SEARCH-IPT trial intervention influenced their abilities to increase IPT uptake. Methods The SEARCH ...
4 Conclusion. The period from 2022 to 2023 in pharmacology has been characterized by a blend of innovation, repurposing, and deepened understanding of infectious diseases. The articles in our Research Topic have significantly contributed to this evolving landscape, where old and new therapies converge, providing hope and direction for future ...
After a golden age of discovery from the 1940s to the 1960s, research on drugs for tuberculosis essentially ceased (Fig. 1).Until recently, standard treatment regimens had not changed since the ...
Tuberculosis (TB) is an ancient human disease caused by Mycobacterium tuberculosis which mainly affects the lungs, making pulmonary disease the most common presentation (K Zaman, 2010) [1]. However, TB is a multi-systemic disease with a protean presentation. The organ system most commonly affected includes the respiratory system, the gastrointestinal (GI) system, the lymphoreticular system ...